+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 4a49 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
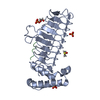
| Title | Structure of phosphoTyr371-c-Cbl-UbcH5B complex | |||||||||
 Components Components |
| |||||||||
 Keywords Keywords | LIGASE | |||||||||
| Function / homology |  Function and homology information Function and homology informationregulation of platelet-derived growth factor receptor-alpha signaling pathway / regulation of Rap protein signal transduction / phosphatidylinositol 3-kinase regulatory subunit binding / flotillin complex / (E3-independent) E2 ubiquitin-conjugating enzyme / Regulation of KIT signaling / ubiquitin-dependent endocytosis / Interleukin-6 signaling / E2 ubiquitin-conjugating enzyme / response to starvation ...regulation of platelet-derived growth factor receptor-alpha signaling pathway / regulation of Rap protein signal transduction / phosphatidylinositol 3-kinase regulatory subunit binding / flotillin complex / (E3-independent) E2 ubiquitin-conjugating enzyme / Regulation of KIT signaling / ubiquitin-dependent endocytosis / Interleukin-6 signaling / E2 ubiquitin-conjugating enzyme / response to starvation / mast cell degranulation / negative regulation of T cell activation / response to testosterone / TGF-beta receptor signaling activates SMADs / ubiquitin conjugating enzyme activity / negative regulation of epidermal growth factor receptor signaling pathway / negative regulation of T cell receptor signaling pathway / protein monoubiquitination / protein K63-linked ubiquitination / protein autoubiquitination / positive regulation of epidermal growth factor receptor signaling pathway / ephrin receptor binding / cellular response to platelet-derived growth factor stimulus / phosphotyrosine residue binding / protein K48-linked ubiquitination / FLT3 signaling by CBL mutants / Negative regulation of FLT3 / PTK6 Regulates RTKs and Their Effectors AKT1 and DOK1 / Synthesis of active ubiquitin: roles of E1 and E2 enzymes / protein modification process / response to activity / TICAM1, RIP1-mediated IKK complex recruitment / InlB-mediated entry of Listeria monocytogenes into host cell / IKK complex recruitment mediated by RIP1 / PINK1-PRKN Mediated Mitophagy / response to gamma radiation / Regulation of signaling by CBL / Negative regulators of DDX58/IFIH1 signaling / Negative regulation of FGFR3 signaling / Peroxisomal protein import / sperm end piece / Negative regulation of FGFR2 signaling / Negative regulation of FGFR4 signaling / Negative regulation of FGFR1 signaling / EGFR downregulation / Regulation of TNFR1 signaling / Constitutive Signaling by EGFRvIII / cellular response to nerve growth factor stimulus / Spry regulation of FGF signaling / Negative regulation of MET activity / RING-type E3 ubiquitin transferase / receptor tyrosine kinase binding / SH3 domain binding / Inactivation of CSF3 (G-CSF) signaling / positive regulation of receptor-mediated endocytosis / male gonad development / Oxygen-dependent proline hydroxylation of Hypoxia-inducible Factor Alpha / CLEC7A (Dectin-1) signaling / FCERI mediated NF-kB activation / cytokine-mediated signaling pathway / Signaling by CSF1 (M-CSF) in myeloid cells / protein polyubiquitination / ubiquitin-protein transferase activity / ubiquitin protein ligase activity / Downstream TCR signaling / Cargo recognition for clathrin-mediated endocytosis / Antigen processing: Ubiquitination & Proteasome degradation / Constitutive Signaling by Ligand-Responsive EGFR Cancer Variants / E3 ubiquitin ligases ubiquitinate target proteins / Clathrin-mediated endocytosis / growth cone / Neddylation / cellular response to hypoxia / ubiquitin-dependent protein catabolic process / response to ethanol / positive regulation of phosphatidylinositol 3-kinase/protein kinase B signal transduction / protein stabilization / cilium / protein ubiquitination / cadherin binding / membrane raft / focal adhesion / calcium ion binding / DNA damage response / symbiont entry into host cell / negative regulation of apoptotic process / perinuclear region of cytoplasm / Golgi apparatus / signal transduction / protein-containing complex / extracellular exosome / zinc ion binding / nucleoplasm / ATP binding / nucleus / plasma membrane / cytosol Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.214 Å MOLECULAR REPLACEMENT / Resolution: 2.214 Å | |||||||||
 Authors Authors | Dou, H. / Buetow, L. / Hock, A. / Sibbet, G.J. / Vousden, K.H. / Huang, D.T. | |||||||||
 Citation Citation |  Journal: Nat. Struct. Mol. Biol. / Year: 2012 Journal: Nat. Struct. Mol. Biol. / Year: 2012Title: Structural basis for autoinhibition and phosphorylation-dependent activation of c-Cbl. Authors: Dou, H. / Buetow, L. / Hock, A. / Sibbet, G.J. / Vousden, K.H. / Huang, D.T. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  4a49.cif.gz 4a49.cif.gz | 112 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb4a49.ent.gz pdb4a49.ent.gz | 87.6 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  4a49.json.gz 4a49.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/a4/4a49 https://data.pdbj.org/pub/pdb/validation_reports/a4/4a49 ftp://data.pdbj.org/pub/pdb/validation_reports/a4/4a49 ftp://data.pdbj.org/pub/pdb/validation_reports/a4/4a49 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  2y1mC  2y1nC  4a4bC  4a4cC  1fbvS  2eskS C: citing same article ( S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
| ||||||||
| Components on special symmetry positions |
|
- Components
Components
| #1: Protein | Mass: 9508.621 Da / Num. of mol.: 1 / Fragment: C-CBL RESIDUES 354-435 / Mutation: Y368F Source method: isolated from a genetically manipulated source Details: TYR371 IS PHOSPHORYLATED / Source: (gene. exp.)  Homo sapiens (human) / Gene: CBL, CBL2, RNF55 / Production host: Homo sapiens (human) / Gene: CBL, CBL2, RNF55 / Production host:  References: UniProt: P22681, RING-type E3 ubiquitin transferase | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| #2: Protein | Mass: 16755.227 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: UBE2D2, PUBC1, UBC4, UBC5B, UBCH4, UBCH5B / Production host: Homo sapiens (human) / Gene: UBE2D2, PUBC1, UBC4, UBC5B, UBCH4, UBCH5B / Production host:  References: UniProt: P62837, E2 ubiquitin-conjugating enzyme, (E3-independent) E2 ubiquitin-conjugating enzyme | ||||||||||
| #3: Chemical | | #4: Chemical | ChemComp-K / | #5: Water | ChemComp-HOH / | Compound details | ENGINEERED | Has protein modification | Y | Sequence details | Y368 IS MUTATED TO PHE. Y371 IS PHOSPHORYL | |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 3.8 Å3/Da / Density % sol: 67 % / Description: NONE |
|---|---|
| Crystal grow | Temperature: 291 K Details: 20% (W/V) PEG 3350 AND 0.2 M POTASSIUM THIOCYANATE AT 18 DEGREES CELSIUS. |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  Diamond Diamond  / Beamline: I02 / Wavelength: 0.9795 / Beamline: I02 / Wavelength: 0.9795 |
| Detector | Type: ADSC QUANTUM 315 / Detector: CCD / Date: Sep 19, 2011 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.9795 Å / Relative weight: 1 |
| Reflection | Resolution: 2.21→40 Å / Num. obs: 20424 / % possible obs: 99.6 % / Observed criterion σ(I): 2 / Redundancy: 6 % / Biso Wilson estimate: 44.92 Å2 / Rmerge(I) obs: 0.08 / Net I/σ(I): 11.6 |
| Reflection shell | Resolution: 2.21→2.27 Å / Redundancy: 6.1 % / Rmerge(I) obs: 0.64 / Mean I/σ(I) obs: 2.8 / % possible all: 100 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: PDB ENTRIES 2ESK AND 1FBV Resolution: 2.214→36.297 Å / SU ML: 0.57 / σ(F): 1.35 / Phase error: 19.67 / Stereochemistry target values: ML Details: CHAIN A RESIDUE 354-358 AND CHAIN B RESIDUE 1 ARE DISORDERED.
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.83 Å / VDW probe radii: 1.1 Å / Solvent model: FLAT BULK SOLVENT MODEL / Bsol: 45.77 Å2 / ksol: 0.314 e/Å3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 53.6 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.214→36.297 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS params. | Method: refined / Refine-ID: X-RAY DIFFRACTION
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS group |
|
 Movie
Movie Controller
Controller





















 PDBj
PDBj













