[English] 日本語
 Yorodumi
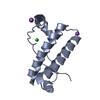
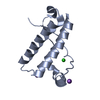
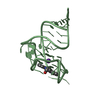
Yorodumi- PDB-2xb1: Crystal structure of the human Pygo2 PHD finger in complex with t... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 2xb1 | ||||||
|---|---|---|---|---|---|---|---|
| Title | Crystal structure of the human Pygo2 PHD finger in complex with the B9L HD1 domain | ||||||
 Components Components | PYGOPUS HOMOLOG 2, B-CELL CLL/LYMPHOMA 9-LIKE PROTEIN | ||||||
 Keywords Keywords | TRANSCRIPTION / FUSION PROTEIN / SIGNAL TRANSDUCTION / METAL BINDING / WNT PROTEINS | ||||||
| Function / homology |  Function and homology information Function and homology informationhistone acetyltransferase regulator activity / spermatid nucleus differentiation / regulation of mammary gland epithelial cell proliferation / beta-catenin-TCF complex / mammary gland development / myoblast differentiation / regulation of cell morphogenesis / lens development in camera-type eye / roof of mouth development / somatic stem cell population maintenance ...histone acetyltransferase regulator activity / spermatid nucleus differentiation / regulation of mammary gland epithelial cell proliferation / beta-catenin-TCF complex / mammary gland development / myoblast differentiation / regulation of cell morphogenesis / lens development in camera-type eye / roof of mouth development / somatic stem cell population maintenance / skeletal muscle cell differentiation / developmental growth / canonical Wnt signaling pathway / positive regulation of epithelial to mesenchymal transition / kidney development / Deactivation of the beta-catenin transactivating complex / negative regulation of transforming growth factor beta receptor signaling pathway / Formation of the beta-catenin:TCF transactivating complex / brain development / beta-catenin binding / fibrillar center / histone binding / transcription by RNA polymerase II / transcription coactivator activity / chromatin binding / positive regulation of transcription by RNA polymerase II / zinc ion binding / nucleoplasm / nucleus Similarity search - Function | ||||||
| Biological species |  HOMO SAPIENS (human) HOMO SAPIENS (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.9 Å MOLECULAR REPLACEMENT / Resolution: 1.9 Å | ||||||
 Authors Authors | Miller, T.C. / Rutherford, T.J. / Johnson, C.M. / Fiedler, M. / Bienz, M. | ||||||
 Citation Citation |  Journal: J.Mol.Biol. / Year: 2010 Journal: J.Mol.Biol. / Year: 2010Title: Allosteric Remodeling of the Histone H3 Binding Pocket in the Pygo2 Phd Finger Triggered by its Binding to the B9L/Bcl9 Co-Factor. Authors: Miller, T.C. / Rutherford, T.J. / Johnson, C.M. / Fiedler, M. / Bienz, M. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  2xb1.cif.gz 2xb1.cif.gz | 96.5 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb2xb1.ent.gz pdb2xb1.ent.gz | 74.4 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  2xb1.json.gz 2xb1.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/xb/2xb1 https://data.pdbj.org/pub/pdb/validation_reports/xb/2xb1 ftp://data.pdbj.org/pub/pdb/validation_reports/xb/2xb1 ftp://data.pdbj.org/pub/pdb/validation_reports/xb/2xb1 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  2vp7S S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||
| 2 | 
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 11267.499 Da / Num. of mol.: 2 Fragment: PHD FINGER AND HD1 DOMAIN, RESIDUES 325-387,232-266 Source method: isolated from a genetically manipulated source Details: ARTIFICIAL LINKER BETWEEN PHD FINGER AND HD1 DOMAIN (7AA LINKER-GSGSGSG) Source: (gene. exp.)  HOMO SAPIENS (human) / Plasmid: PETM41-HPHD2GSGHD1 / Production host: HOMO SAPIENS (human) / Plasmid: PETM41-HPHD2GSGHD1 / Production host:  #2: Chemical | ChemComp-ZN / #3: Chemical | #4: Chemical | ChemComp-PEG / | #5: Water | ChemComp-HOH / | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.3 Å3/Da / Density % sol: 46.12 % / Description: NONE |
|---|---|
| Crystal grow | Details: 6% PEG 1000, 1.6 M (NH4)2SO4, 100 MM HEPES PH 7.5, 45 MM HCL |
-Data collection
| Diffraction | Mean temperature: 77 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  ESRF ESRF  / Beamline: ID29 / Wavelength: 1.04 / Beamline: ID29 / Wavelength: 1.04 |
| Detector | Type: ADSC QUANTUM 315r / Detector: CCD / Date: Jun 29, 2009 / Details: UNDULATOR |
| Radiation | Monochromator: SI(311) / Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1.04 Å / Relative weight: 1 |
| Reflection | Resolution: 1.9→38.75 Å / Num. obs: 18032 / % possible obs: 99.5 % / Observed criterion σ(I): 1.5 / Redundancy: 3.4 % / Biso Wilson estimate: 17 Å2 / Rsym value: 0.08 / Net I/σ(I): 6.4 |
| Reflection shell | Resolution: 1.9→2 Å / Redundancy: 3.4 % / Rmerge(I) obs: 0.358 / Mean I/σ(I) obs: 1.6 / Rsym value: 0.358 / % possible all: 99.5 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: PDB ENTRY 2VP7 Resolution: 1.9→31.31 Å / Cor.coef. Fo:Fc: 0.963 / Cor.coef. Fo:Fc free: 0.945 / SU B: 4.873 / SU ML: 0.079 / Cross valid method: THROUGHOUT / ESU R: 0.139 / ESU R Free: 0.136 / Stereochemistry target values: MAXIMUM LIKELIHOOD Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS.U VALUES RESIDUAL ONLY ATOM RECORD CONTAINS SUM OF TLS AND RESIDUAL B FACTORS. ANISOU RECORD CONTAINS SUM OF TLS AND RESIDUAL U FACTORS.
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.4 Å / Solvent model: MASK | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 9.815 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.9→31.31 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller









 PDBj
PDBj





