Entry Database : PDB / ID : 2p4rTitle Structural basis for a novel interaction between AIP4 and beta-PIX E3 ubiquitin-protein ligase Itchy homolog Rho guanine nucleotide exchange factor 7 Keywords / Function / homology Function Domain/homology Component
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / Biological species Rattus norvegicus (Norway rat)Method / / Resolution : 2.001 Å Authors Min, K.C. Journal : J.Biol.Chem. / Year : 2007Title : A novel interaction between atrophin-interacting protein 4 and beta-p21-activated kinase-interactive exchange factor is mediated by an SH3 domain.Authors : Janz, J.M. / Sakmar, T.P. / Min, K.C. History Deposition Mar 13, 2007 Deposition site / Processing site Revision 1.0 Jul 24, 2007 Provider / Type Revision 1.1 May 1, 2008 Group Revision 1.2 Jul 13, 2011 Group / Version format complianceRevision 1.3 Aug 30, 2023 Group Data collection / Database references ... Data collection / Database references / Derived calculations / Refinement description Category chem_comp_atom / chem_comp_bond ... chem_comp_atom / chem_comp_bond / database_2 / pdbx_initial_refinement_model / struct_ref_seq_dif / struct_site Item _database_2.pdbx_DOI / _database_2.pdbx_database_accession ... _database_2.pdbx_DOI / _database_2.pdbx_database_accession / _struct_ref_seq_dif.details / _struct_site.pdbx_auth_asym_id / _struct_site.pdbx_auth_comp_id / _struct_site.pdbx_auth_seq_id
Show all Show less
 Yorodumi
Yorodumi Open data
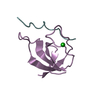
Open data Basic information
Basic information Components
Components Keywords
Keywords Function and homology information
Function and homology information
 X-RAY DIFFRACTION /
X-RAY DIFFRACTION /  MOLECULAR REPLACEMENT / Resolution: 2.001 Å
MOLECULAR REPLACEMENT / Resolution: 2.001 Å  Authors
Authors Citation
Citation Journal: J.Biol.Chem. / Year: 2007
Journal: J.Biol.Chem. / Year: 2007 Structure visualization
Structure visualization Molmil
Molmil Jmol/JSmol
Jmol/JSmol Downloads & links
Downloads & links Download
Download 2p4r.cif.gz
2p4r.cif.gz PDBx/mmCIF format
PDBx/mmCIF format pdb2p4r.ent.gz
pdb2p4r.ent.gz PDB format
PDB format 2p4r.json.gz
2p4r.json.gz PDBx/mmJSON format
PDBx/mmJSON format Other downloads
Other downloads https://data.pdbj.org/pub/pdb/validation_reports/p4/2p4r
https://data.pdbj.org/pub/pdb/validation_reports/p4/2p4r ftp://data.pdbj.org/pub/pdb/validation_reports/p4/2p4r
ftp://data.pdbj.org/pub/pdb/validation_reports/p4/2p4r
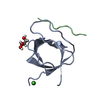
 Links
Links Assembly
Assembly
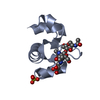
 Components
Components

 X-RAY DIFFRACTION / Number of used crystals: 1
X-RAY DIFFRACTION / Number of used crystals: 1  Sample preparation
Sample preparation ROTATING ANODE / Type: RIGAKU / Wavelength: 1.54 Å
ROTATING ANODE / Type: RIGAKU / Wavelength: 1.54 Å Processing
Processing MOLECULAR REPLACEMENT
MOLECULAR REPLACEMENT Movie
Movie Controller
Controller









 PDBj
PDBj












