| 登録情報 | データベース: PDB / ID: 1zpl
|
|---|
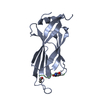
| タイトル | E. coli F17a-G lectin domain complex with GlcNAc(beta1-O)Me |
|---|
 要素 要素 | F17a-G |
|---|
 キーワード キーワード | CELL ADHESION / FIMBRIAE / LECTINS / BACTERIAL ADHESION / PROTEIN-SUGAR COMPLEX |
|---|
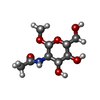
| 機能・相同性 |  機能・相同性情報 機能・相同性情報
adhesion of symbiont to host / cell adhesion involved in single-species biofilm formation / pilus / carbohydrate binding類似検索 - 分子機能 Bacterial adhesins - F17c-type / Fimbrial adhesin F17-AG, lectin domain / Fimbrial adhesin F17-AG, lectin domain / Fimbrial-type adhesion domain / Fimbrial protein / : / Fimbrial-type adhesion domain superfamily / Adhesion domain superfamily / Immunoglobulin-like / Sandwich / Mainly Beta類似検索 - ドメイン・相同性 methyl 2-acetamido-2-deoxy-beta-D-glucopyranoside / F17a-G fimbrial adhesin類似検索 - 構成要素 |
|---|
| 生物種 |   Escherichia coli (大腸菌) Escherichia coli (大腸菌) |
|---|
| 手法 |  X線回折 / X線回折 /  分子置換 / 解像度: 1.7 Å 分子置換 / 解像度: 1.7 Å |
|---|
 データ登録者 データ登録者 | Buts, L. / Wellens, A. / Van Molle, I. / Wyns, L. / Loris, R. / Lahmann, M. / Oscarson, S. / De Greve, H. / Bouckaert, J. |
|---|
 引用 引用 |  ジャーナル: Acta Crystallogr.,Sect.D / 年: 2005 ジャーナル: Acta Crystallogr.,Sect.D / 年: 2005
タイトル: Impact of natural variation in bacterial F17G adhesins on crystallization behaviour.
著者: Buts, L. / Wellens, A. / Van Molle, I. / Wyns, L. / Loris, R. / Lahmann, M. / Oscarson, S. / De Greve, H. / Bouckaert, J. |
|---|
| 履歴 | | 登録 | 2005年5月17日 | 登録サイト: RCSB / 処理サイト: RCSB |
|---|
| 改定 1.0 | 2006年5月23日 | Provider: repository / タイプ: Initial release |
|---|
| 改定 1.1 | 2008年4月30日 | Group: Version format compliance |
|---|
| 改定 1.2 | 2011年7月13日 | Group: Version format compliance |
|---|
| 改定 1.3 | 2011年10月5日 | Group: Derived calculations |
|---|
| 改定 1.4 | 2020年7月29日 | Group: Data collection / Derived calculations / Structure summary
カテゴリ: chem_comp / entity ...chem_comp / entity / pdbx_chem_comp_identifier / pdbx_entity_nonpoly / struct_site / struct_site_gen
Item: _chem_comp.mon_nstd_flag / _chem_comp.name ..._chem_comp.mon_nstd_flag / _chem_comp.name / _entity.pdbx_description / _pdbx_entity_nonpoly.name
解説: Carbohydrate remediation / Provider: repository / タイプ: Remediation |
|---|
| 改定 1.5 | 2023年8月23日 | Group: Data collection / Database references ...Data collection / Database references / Refinement description / Structure summary
カテゴリ: chem_comp / chem_comp_atom ...chem_comp / chem_comp_atom / chem_comp_bond / database_2 / pdbx_initial_refinement_model
Item: _chem_comp.pdbx_synonyms / _database_2.pdbx_DOI / _database_2.pdbx_database_accession |
|---|
| 改定 1.6 | 2024年10月30日 | Group: Structure summary
カテゴリ: pdbx_entry_details / pdbx_modification_feature |
|---|
|
|---|
 データを開く
データを開く 基本情報
基本情報 要素
要素 キーワード
キーワード 機能・相同性情報
機能・相同性情報
 X線回折 /
X線回折 /  分子置換 / 解像度: 1.7 Å
分子置換 / 解像度: 1.7 Å  データ登録者
データ登録者 引用
引用 ジャーナル: Acta Crystallogr.,Sect.D / 年: 2005
ジャーナル: Acta Crystallogr.,Sect.D / 年: 2005 構造の表示
構造の表示 Molmil
Molmil Jmol/JSmol
Jmol/JSmol ダウンロードとリンク
ダウンロードとリンク ダウンロード
ダウンロード 1zpl.cif.gz
1zpl.cif.gz PDBx/mmCIF形式
PDBx/mmCIF形式 pdb1zpl.ent.gz
pdb1zpl.ent.gz PDB形式
PDB形式 1zpl.json.gz
1zpl.json.gz PDBx/mmJSON形式
PDBx/mmJSON形式 その他のダウンロード
その他のダウンロード https://data.pdbj.org/pub/pdb/validation_reports/zp/1zpl
https://data.pdbj.org/pub/pdb/validation_reports/zp/1zpl ftp://data.pdbj.org/pub/pdb/validation_reports/zp/1zpl
ftp://data.pdbj.org/pub/pdb/validation_reports/zp/1zpl リンク
リンク 集合体
集合体


 要素
要素

 X線回折
X線回折 試料調製
試料調製 解析
解析 分子置換
分子置換 ムービー
ムービー コントローラー
コントローラー


















 PDBj
PDBj