+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1s1n | ||||||
|---|---|---|---|---|---|---|---|
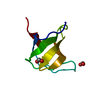
| Title | SH3 domain of human nephrocystin | ||||||
 Components Components | Nephrocystin 1 | ||||||
 Keywords Keywords | CELL ADHESION / BETA BARREL | ||||||
| Function / homology |  Function and homology information Function and homology informationprotein localization involved in establishment of planar polarity / spermatid differentiation / visual behavior / photoreceptor connecting cilium / positive regulation of bicellular tight junction assembly / cell projection organization / motile cilium / bicellular tight junction / axoneme / Anchoring of the basal body to the plasma membrane ...protein localization involved in establishment of planar polarity / spermatid differentiation / visual behavior / photoreceptor connecting cilium / positive regulation of bicellular tight junction assembly / cell projection organization / motile cilium / bicellular tight junction / axoneme / Anchoring of the basal body to the plasma membrane / adherens junction / cell-cell adhesion / cell-cell junction / retina development in camera-type eye / actin cytoskeleton organization / cilium / structural molecule activity / signal transduction / membrane / cytoplasm / cytosol Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method | SOLUTION NMR / distance geometry follwed by restrained simulated annealing | ||||||
 Authors Authors | Le Maire, A. / Weber, T. / Saunier, S. / Antignac, C. / Ducruix, A. / Dardel, F. | ||||||
 Citation Citation |  Journal: Proteins / Year: 2005 Journal: Proteins / Year: 2005Title: Solution NMR structure of the SH3 domain of human nephrocystin and analysis of a mutation-causing juvenile nephronophthisis. Authors: le Maire, A. / Weber, T. / Saunier, S. / Broutin, I. / Antignac, C. / Ducruix, A. / Dardel, F. #1:  Journal: HUM.MOL.GENET. / Year: 1997 Journal: HUM.MOL.GENET. / Year: 1997Title: A novel gene that encodes a protein with a putative src homology 3 domain is a candidate gene for familial juvenile nephronophthisis Authors: SAUNIER, S. / CALADO, J. / HEILIG, R. / SILBERMANN, F. / BENESSY, F. / MORIN, G. / KONRAD, M. / BROYER, M. / GUBLER, M.C. / WEISSENBACH, J. #2:  Journal: NAT.GENET. / Year: 1997 Journal: NAT.GENET. / Year: 1997Title: A novel gene encoding an SH3 domain protein is mutated in nephronophthisis type 1 Authors: HILDEBRANDT, F. / OTTO, E. / RENSIG, C. / NOTHWANG, H.G. / VOLLMER, M. / ADOLPHS, J. / HANUSH, H. / BRANDIS, M. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1s1n.cif.gz 1s1n.cif.gz | 321.5 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1s1n.ent.gz pdb1s1n.ent.gz | 267.1 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1s1n.json.gz 1s1n.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/s1/1s1n https://data.pdbj.org/pub/pdb/validation_reports/s1/1s1n ftp://data.pdbj.org/pub/pdb/validation_reports/s1/1s1n ftp://data.pdbj.org/pub/pdb/validation_reports/s1/1s1n | HTTPS FTP |
|---|
-Related structure data
| Similar structure data |
|---|
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| |||||||||
| NMR ensembles |
|
- Components
Components
| #1: Protein | Mass: 7623.472 Da / Num. of mol.: 1 / Fragment: SH3 DOMAIN Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: NPHP1, NPH1 / Plasmid: PGEX-4T-2 / Species (production host): Escherichia coli / Production host: Homo sapiens (human) / Gene: NPHP1, NPH1 / Plasmid: PGEX-4T-2 / Species (production host): Escherichia coli / Production host:  |
|---|
-Experimental details
-Experiment
| Experiment | Method: SOLUTION NMR | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NMR experiment |
| ||||||||||||||||||||||||
| NMR details | Text: The structure was determined using triple-resonance NMR spectroscopy |
- Sample preparation
Sample preparation
| Details | Contents: 1.5 mM SH3 domain U-15N,13C; 20mM phosphate buffer K, pH6.5 Solvent system: 90% H2O/10% D2O |
|---|---|
| Sample conditions | Ionic strength: 20 mM K-phosphate / pH: 6.5 / Pressure: ambient / Temperature: 293 K |
-NMR measurement
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M |
|---|---|
| Radiation wavelength | Relative weight: 1 |
| NMR spectrometer | Type: Bruker AVANCE / Manufacturer: Bruker / Model: AVANCE / Field strength: 600 MHz |
- Processing
Processing
| NMR software |
| ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method: distance geometry follwed by restrained simulated annealing Software ordinal: 1 Details: The structures are based on a total of 623 restraints. 540 are NOE-derived restraints : 110 intraresidue, 121 sequential, 37 medium-range and 272 long-range. 30 distance restraints from ...Details: The structures are based on a total of 623 restraints. 540 are NOE-derived restraints : 110 intraresidue, 121 sequential, 37 medium-range and 272 long-range. 30 distance restraints from hydrogen bonds. 53 dihedral angle restraints. | ||||||||||||
| NMR representative | Selection criteria: closest to the average,lowest energy | ||||||||||||
| NMR ensemble | Conformer selection criteria: structures with the least restraint violations,structures with the lowest energy Conformers calculated total number: 20 / Conformers submitted total number: 17 |
 Movie
Movie Controller
Controller












 PDBj
PDBj X-PLOR
X-PLOR