[English] 日本語
 Yorodumi
Yorodumi- EMDB-9686: Cryo-EM structure of Echovirus 6 complexed with its uncoating rec... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-9686 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
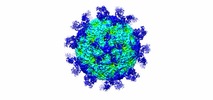
| Title | Cryo-EM structure of Echovirus 6 complexed with its uncoating receptor FcRn at PH 5.5 | |||||||||
 Map data Map data | Cryo-EM structure of Echovirus 6 complexed with its uncoating receptor FcRn at PH 5.5 | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | virus-receptor complex / VIRUS | |||||||||
| Biological species |  Echovirus E6 Echovirus E6 | |||||||||
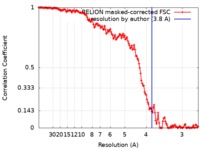
| Method | single particle reconstruction / cryo EM / Resolution: 3.8 Å | |||||||||
 Authors Authors | Gao GF / Liu S | |||||||||
| Funding support |  China, 1 items China, 1 items
| |||||||||
 Citation Citation |  Journal: Cell / Year: 2019 Journal: Cell / Year: 2019Title: Human Neonatal Fc Receptor Is the Cellular Uncoating Receptor for Enterovirus B. Authors: Xin Zhao / Guigen Zhang / Sheng Liu / Xiangpeng Chen / Ruchao Peng / Lianpan Dai / Xiao Qu / Shihua Li / Hao Song / Zhengrong Gao / Pengfei Yuan / Zhiheng Liu / Changyao Li / Zifang Shang / ...Authors: Xin Zhao / Guigen Zhang / Sheng Liu / Xiangpeng Chen / Ruchao Peng / Lianpan Dai / Xiao Qu / Shihua Li / Hao Song / Zhengrong Gao / Pengfei Yuan / Zhiheng Liu / Changyao Li / Zifang Shang / Yan Li / Meifan Zhang / Jianxun Qi / Han Wang / Ning Du / Yan Wu / Yuhai Bi / Shan Gao / Yi Shi / Jinghua Yan / Yong Zhang / Zhengde Xie / Wensheng Wei / George F Gao /  Abstract: Enterovirus B (EV-B), a major proportion of the genus Enterovirus in the family Picornaviridae, is the causative agent of severe human infectious diseases. Although cellular receptors for ...Enterovirus B (EV-B), a major proportion of the genus Enterovirus in the family Picornaviridae, is the causative agent of severe human infectious diseases. Although cellular receptors for coxsackievirus B in EV-B have been identified, receptors mediating virus entry, especially the uncoating process of echovirus and other EV-B remain obscure. Here, we found that human neonatal Fc receptor (FcRn) is the uncoating receptor for major EV-B. FcRn binds to the virus particles in the "canyon" through its FCGRT subunit. By obtaining multiple cryo-electron microscopy structures at different stages of virus entry at atomic or near-atomic resolution, we deciphered the underlying mechanisms of enterovirus attachment and uncoating. These structures revealed that different from the attachment receptor CD55, binding of FcRn to the virions induces efficient release of "pocket factor" under acidic conditions and initiates the conformational changes in viral particle, providing a structural basis for understanding the mechanisms of enterovirus entry. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_9686.map.gz emd_9686.map.gz | 36.6 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-9686-v30.xml emd-9686-v30.xml emd-9686.xml emd-9686.xml | 15.2 KB 15.2 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_9686_fsc.xml emd_9686_fsc.xml | 13.9 KB | Display |  FSC data file FSC data file |
| Images |  emd_9686.png emd_9686.png | 252.2 KB | ||
| Filedesc metadata |  emd-9686.cif.gz emd-9686.cif.gz | 5.7 KB | ||
| Others |  emd_9686_additional.map.gz emd_9686_additional.map.gz | 7.2 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-9686 http://ftp.pdbj.org/pub/emdb/structures/EMD-9686 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-9686 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-9686 | HTTPS FTP |
-Related structure data
| Related structure data |  6illMC  9684C  9685C  9687C  9688C  9689C  9690C  6iljC  6ilkC  6ilmC  6ilnC  6iloC  6ilpC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_9686.map.gz / Format: CCP4 / Size: 244.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_9686.map.gz / Format: CCP4 / Size: 244.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Cryo-EM structure of Echovirus 6 complexed with its uncoating receptor FcRn at PH 5.5 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.35 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
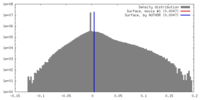
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Additional map: Cryo-EM structure of Echovirus 6 complexed with its...
| File | emd_9686_additional.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Cryo-EM structure of Echovirus 6 complexed with its uncoating receptor FcRn at PH 5.5(map_bin2) | ||||||||||||
| Projections & Slices |
| ||||||||||||
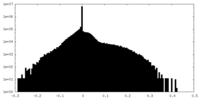
| Density Histograms |
- Sample components
Sample components
-Entire : Cryo-EM structure of Echovirus 6 complexed with its uncoating rec...
| Entire | Name: Cryo-EM structure of Echovirus 6 complexed with its uncoating receptor FcRn at PH 5.5 |
|---|---|
| Components |
|
-Supramolecule #1: Cryo-EM structure of Echovirus 6 complexed with its uncoating rec...
| Supramolecule | Name: Cryo-EM structure of Echovirus 6 complexed with its uncoating receptor FcRn at PH 5.5 type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  Echovirus E6 Echovirus E6 |
-Macromolecule #1: Capsid protein VP1
| Macromolecule | Name: Capsid protein VP1 / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Echovirus E6 Echovirus E6 |
| Molecular weight | Theoretical: 31.12574 KDa |
| Sequence | String: VVRVADTMPS GPSNSESIPA LTAAETGHTS QVVPSDTIQT RHVRNFHVRS ESSVENFLSR SACVYIVEYK TRDDTPDKMY DSWVINTRQ VAQLRRKLEF FTYVRFDVEV TFVITSVQDD STRQNTDTPA LTHQIMYVPP GGPIPQAVDD YNWQTSTNPS V FWTEGNAP ...String: VVRVADTMPS GPSNSESIPA LTAAETGHTS QVVPSDTIQT RHVRNFHVRS ESSVENFLSR SACVYIVEYK TRDDTPDKMY DSWVINTRQ VAQLRRKLEF FTYVRFDVEV TFVITSVQDD STRQNTDTPA LTHQIMYVPP GGPIPQAVDD YNWQTSTNPS V FWTEGNAP PRMSIPFMSV GNAYSNFYDG WSHFSQTGVY GFNTLNNMGK LYFRHVNDKT ISPITSKVRI YFKPKHVKAW VP RPPRLCE YTHKDNVDFE PKGVTTSRTQ LTISNS |
-Macromolecule #2: Capsid protein VP2
| Macromolecule | Name: Capsid protein VP2 / type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Echovirus E6 Echovirus E6 |
| Molecular weight | Theoretical: 28.06452 KDa |
| Sequence | String: SDRVRSITLG NSTITTQESA NVVVGYGVWP DYLSDEEATA EDQPTQPDVA TCRFYTLDSV SWMKESQGWW WKFPDALRDM GLFGQNMQY HYLGRSGYTI HVQCNASKFH QGCLLVVCVP EAEMGAANIN EKINREHLSN GEVANTFSGT KSSNTNDVQQ A VFNAGMGV ...String: SDRVRSITLG NSTITTQESA NVVVGYGVWP DYLSDEEATA EDQPTQPDVA TCRFYTLDSV SWMKESQGWW WKFPDALRDM GLFGQNMQY HYLGRSGYTI HVQCNASKFH QGCLLVVCVP EAEMGAANIN EKINREHLSN GEVANTFSGT KSSNTNDVQQ A VFNAGMGV AVGNLTIFPH QWINLRTNNC ATIVMPYINS VPMDNMFRHY NFTLMIIPFA KLDYAAGSST YIPITVTVAP MC AEYNGLR LAGHQ |
-Macromolecule #3: Capsid protein VP3
| Macromolecule | Name: Capsid protein VP3 / type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Echovirus E6 Echovirus E6 |
| Molecular weight | Theoretical: 26.378936 KDa |
| Sequence | String: GLPVMNTPGS NQFLTSDDYQ SPTAMPQFDV TPEMNIPGEV KNLMEIAEVD SVVPVNNVNE NVNSLEAYRI PVHSVTETGA QVFGFTLQP GADTVMERTL LGEILNYYAN WSGSIKLTFM YCGSAMATGK FLLAYSPPGA GVPKNRREAM LGTHIIWDIG L QSSCVLCV ...String: GLPVMNTPGS NQFLTSDDYQ SPTAMPQFDV TPEMNIPGEV KNLMEIAEVD SVVPVNNVNE NVNSLEAYRI PVHSVTETGA QVFGFTLQP GADTVMERTL LGEILNYYAN WSGSIKLTFM YCGSAMATGK FLLAYSPPGA GVPKNRREAM LGTHIIWDIG L QSSCVLCV PWISQTHYRF VSKDIYTDAG FITCWYQTSI VVPAEVQNQS VILCFVSACN DFSVRLLRDS PFVRQTAFYQ |
-Macromolecule #4: Capsid protein VP4
| Macromolecule | Name: Capsid protein VP4 / type: protein_or_peptide / ID: 4 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Echovirus E6 Echovirus E6 |
| Molecular weight | Theoretical: 7.325959 KDa |
| Sequence | String: GAQVSTQKTG AHETSLSASG NSTIHYTNIN YYKDAASNSA NRQDFTQDPG KFTEPVKDIM VKSLPALN |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 5.5 |
|---|---|
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 277 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 QUANTUM (4k x 4k) / Detector mode: SUPER-RESOLUTION / Average electron dose: 40.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Refinement | Space: REAL / Protocol: FLEXIBLE FIT |
|---|---|
| Output model |  PDB-6ill: |
 Movie
Movie Controller
Controller










 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)