+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 | データベース: EMDB / ID: EMD-9395 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| タイトル | Electron Cryo-Microscopy of Chikungunya in Complex with Mouse Mxra8 Receptor | |||||||||
 マップデータ マップデータ | Chikungunya in Complex with Mouse Mxra8 Receptor | |||||||||
 試料 試料 |
| |||||||||
 キーワード キーワード | Chikungunya / viral receptor / Mxra8 / Structural Genomics / Center for Structural Genomics of Infectious Diseases / CSGID / VIRUS | |||||||||
| 機能・相同性 |  機能・相同性情報 機能・相同性情報establishment of glial blood-brain barrier / Regulation of Insulin-like Growth Factor (IGF) transport and uptake by Insulin-like Growth Factor Binding Proteins (IGFBPs) / Post-translational protein phosphorylation / T=4 icosahedral viral capsid / ciliary membrane / bicellular tight junction / host cell cytoplasm / cell adhesion / serine-type endopeptidase activity / fusion of virus membrane with host endosome membrane ...establishment of glial blood-brain barrier / Regulation of Insulin-like Growth Factor (IGF) transport and uptake by Insulin-like Growth Factor Binding Proteins (IGFBPs) / Post-translational protein phosphorylation / T=4 icosahedral viral capsid / ciliary membrane / bicellular tight junction / host cell cytoplasm / cell adhesion / serine-type endopeptidase activity / fusion of virus membrane with host endosome membrane / symbiont entry into host cell / virion attachment to host cell / host cell plasma membrane / virion membrane / structural molecule activity / cell surface / proteolysis / nucleus / membrane / cytoplasm 類似検索 - 分子機能 | |||||||||
| 生物種 |   Chikungunya virus (チクングニヤウイルス) / Chikungunya virus (チクングニヤウイルス) /  | |||||||||
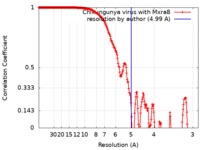
| 手法 | 単粒子再構成法 / クライオ電子顕微鏡法 / 解像度: 4.99 Å | |||||||||
 データ登録者 データ登録者 | Basore K / Kim AS | |||||||||
| 資金援助 |  米国, 1件 米国, 1件
| |||||||||
 引用 引用 |  ジャーナル: Cell / 年: 2019 ジャーナル: Cell / 年: 2019タイトル: Cryo-EM Structure of Chikungunya Virus in Complex with the Mxra8 Receptor. 著者: Katherine Basore / Arthur S Kim / Christopher A Nelson / Rong Zhang / Brittany K Smith / Carla Uranga / Lo Vang / Ming Cheng / Michael L Gross / Jonathan Smith / Michael S Diamond / Daved H Fremont /  要旨: Mxra8 is a receptor for multiple arthritogenic alphaviruses that cause debilitating acute and chronic musculoskeletal disease in humans. Herein, we present a 2.2 Å resolution X-ray crystal ...Mxra8 is a receptor for multiple arthritogenic alphaviruses that cause debilitating acute and chronic musculoskeletal disease in humans. Herein, we present a 2.2 Å resolution X-ray crystal structure of Mxra8 and 4 to 5 Å resolution cryo-electron microscopy reconstructions of Mxra8 bound to chikungunya (CHIKV) virus-like particles and infectious virus. The Mxra8 ectodomain contains two strand-swapped Ig-like domains oriented in a unique disulfide-linked head-to-head arrangement. Mxra8 binds by wedging into a cleft created by two adjacent CHIKV E2-E1 heterodimers in one trimeric spike and engaging a neighboring spike. Two binding modes are observed with the fully mature VLP, with one Mxra8 binding with unique contacts. Only the high-affinity binding mode was observed in the complex with infectious CHIKV, as viral maturation and E3 occupancy appear to influence receptor binding-site usage. Our studies provide insight into how Mxra8 binds CHIKV and creates a path for developing alphavirus entry inhibitors. | |||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| ムービー |
 ムービービューア ムービービューア |
|---|---|
| 構造ビューア | EMマップ:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| 添付画像 |
- ダウンロードとリンク
ダウンロードとリンク
-EMDBアーカイブ
| マップデータ |  emd_9395.map.gz emd_9395.map.gz | 764.4 MB |  EMDBマップデータ形式 EMDBマップデータ形式 | |
|---|---|---|---|---|
| ヘッダ (付随情報) |  emd-9395-v30.xml emd-9395-v30.xml emd-9395.xml emd-9395.xml | 23.3 KB 23.3 KB | 表示 表示 |  EMDBヘッダ EMDBヘッダ |
| FSC (解像度算出) |  emd_9395_fsc.xml emd_9395_fsc.xml | 20.7 KB | 表示 |  FSCデータファイル FSCデータファイル |
| 画像 |  emd_9395.png emd_9395.png | 161 KB | ||
| Filedesc metadata |  emd-9395.cif.gz emd-9395.cif.gz | 7.5 KB | ||
| アーカイブディレクトリ |  http://ftp.pdbj.org/pub/emdb/structures/EMD-9395 http://ftp.pdbj.org/pub/emdb/structures/EMD-9395 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-9395 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-9395 | HTTPS FTP |
-検証レポート
| 文書・要旨 |  emd_9395_validation.pdf.gz emd_9395_validation.pdf.gz | 754.4 KB | 表示 |  EMDB検証レポート EMDB検証レポート |
|---|---|---|---|---|
| 文書・詳細版 |  emd_9395_full_validation.pdf.gz emd_9395_full_validation.pdf.gz | 753.9 KB | 表示 | |
| XML形式データ |  emd_9395_validation.xml.gz emd_9395_validation.xml.gz | 18.3 KB | 表示 | |
| CIF形式データ |  emd_9395_validation.cif.gz emd_9395_validation.cif.gz | 25.6 KB | 表示 | |
| アーカイブディレクトリ |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-9395 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-9395 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-9395 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-9395 | HTTPS FTP |
-関連構造データ
- リンク
リンク
| EMDBのページ |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| 「今月の分子」の関連する項目 |
- マップ
マップ
| ファイル |  ダウンロード / ファイル: emd_9395.map.gz / 形式: CCP4 / 大きさ: 824 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) ダウンロード / ファイル: emd_9395.map.gz / 形式: CCP4 / 大きさ: 824 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | Chikungunya in Complex with Mouse Mxra8 Receptor | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 投影像・断面図 | 画像のコントロール
画像は Spider により作成 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ボクセルのサイズ | X=Y=Z: 1.403 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 密度 |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 対称性 | 空間群: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 詳細 | EMDB XML:
CCP4マップ ヘッダ情報:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-添付データ
- 試料の構成要素
試料の構成要素
-全体 : Chikungunya virus with Mxra8 receptor
| 全体 | 名称: Chikungunya virus with Mxra8 receptor |
|---|---|
| 要素 |
|
-超分子 #1: Chikungunya virus with Mxra8 receptor
| 超分子 | 名称: Chikungunya virus with Mxra8 receptor / タイプ: complex / ID: 1 / 親要素: 0 / 含まれる分子: #1-#5 |
|---|
-超分子 #2: Chikungunya virus
| 超分子 | 名称: Chikungunya virus / タイプ: complex / ID: 2 / 親要素: 1 / 含まれる分子: #1-#4 |
|---|---|
| 由来(天然) | 生物種:   Chikungunya virus (チクングニヤウイルス) Chikungunya virus (チクングニヤウイルス)株: 181/25 |
-超分子 #3: mouse Mxra8 receptor ectodomain
| 超分子 | 名称: mouse Mxra8 receptor ectodomain / タイプ: complex / ID: 3 / 親要素: 1 / 含まれる分子: #5 |
|---|---|
| 由来(天然) | 生物種:  |
-分子 #1: E1 glycoprotein
| 分子 | 名称: E1 glycoprotein / タイプ: protein_or_peptide / ID: 1 / コピー数: 4 / 光学異性体: LEVO / EC番号: togavirin |
|---|---|
| 由来(天然) | 生物種:   Chikungunya virus (チクングニヤウイルス) Chikungunya virus (チクングニヤウイルス)株: 181/25 |
| 分子量 | 理論値: 47.497906 KDa |
| 組換発現 | 生物種:  Chlorocebus aethiops (ミドリザル) Chlorocebus aethiops (ミドリザル) |
| 配列 | 文字列: YEHVTVIPNT VGVPYKTLVN RPGYSPMVLE MELLSVTLEP TLSLDYITCE YKTVIPSPYV KCCGTAECKD KSLPDYSCKV FTGVYPFMW GGAYCFCDTE NTQLSEAHVE KSESCKTEFA SAYRAHTASA SAKLRVLYQG NNVTVSAYAN GDHAVTVKDA K FIVGPMSS ...文字列: YEHVTVIPNT VGVPYKTLVN RPGYSPMVLE MELLSVTLEP TLSLDYITCE YKTVIPSPYV KCCGTAECKD KSLPDYSCKV FTGVYPFMW GGAYCFCDTE NTQLSEAHVE KSESCKTEFA SAYRAHTASA SAKLRVLYQG NNVTVSAYAN GDHAVTVKDA K FIVGPMSS AWTPFDNKIV VYKGDVYNMD YPPFGAGRPG QFGDIQSRTP ESEDVYANTQ LVLQRPSAGT VHVPYSQAPS GF KYWLKER GASLQHTAPF GCQIATNPVR AMNCAVGNMP ISIDIPDAAF TRVVDAPSLT DMSCEVPACT HSSDFGGVAI IKY AASKKG KCAVHSMTNA VTIREAEIEV EGNSQLQISF STALASAEFR VQVCSTQVHC AAECHPPKDH IVNYPASHTT LGVQ DISVT AMSWVQKITG GVGLVVAVAA LILIVVLCVS FSRH UniProtKB: Structural polyprotein |
-分子 #2: E2 glycoprotein
| 分子 | 名称: E2 glycoprotein / タイプ: protein_or_peptide / ID: 2 / コピー数: 4 / 光学異性体: LEVO / EC番号: togavirin |
|---|---|
| 由来(天然) | 生物種:   Chikungunya virus (チクングニヤウイルス) Chikungunya virus (チクングニヤウイルス)株: 181/25 |
| 分子量 | 理論値: 47.077719 KDa |
| 組換発現 | 生物種:  Chlorocebus aethiops (ミドリザル) Chlorocebus aethiops (ミドリザル) |
| 配列 | 文字列: NFNVYKAIRP YLAHCPDCGE GHSCHSPVAL ERIRNEATDG TLKIQVSLQI GIKTDDSHDW TKLRYMDNHM PADAERARLF VRTSAPCTI TGTMGHFILA RCPKGETLTV GFTDGRKISH SCTHPFHHDP PVIGREKFHS RPQHGRELPC STYAQSTAAT A EEIEVHMP ...文字列: NFNVYKAIRP YLAHCPDCGE GHSCHSPVAL ERIRNEATDG TLKIQVSLQI GIKTDDSHDW TKLRYMDNHM PADAERARLF VRTSAPCTI TGTMGHFILA RCPKGETLTV GFTDGRKISH SCTHPFHHDP PVIGREKFHS RPQHGRELPC STYAQSTAAT A EEIEVHMP PDTPDRTLMS QQSGNVKITV NSQTVRYKCN CGDSNEGLTT TDKVINNCKV DQCHAAVTNH KKWQYNSPLV PR NAELGDR KGKVHIPFPL ANVTCRVPKA RNPTVTYGKN QVIMLLYPDH PTLLSYRNMG EEPNYQEEWV THKKEIRLTV PTE GLEVTW GNNEPYKYWP QLSTNGTAHG HPHEIILYYY ELYPTMTVVV VSVASFVLLS MVGVAVGMCM CARRRCITPY ELTP GATVP FLLSLICCIR TAKA UniProtKB: Structural polyprotein |
-分子 #3: Capsid protein
| 分子 | 名称: Capsid protein / タイプ: protein_or_peptide / ID: 3 / コピー数: 4 / 光学異性体: LEVO / EC番号: togavirin |
|---|---|
| 由来(天然) | 生物種:   Chikungunya virus (チクングニヤウイルス) Chikungunya virus (チクングニヤウイルス)株: 181/25 |
| 分子量 | 理論値: 16.428607 KDa |
| 組換発現 | 生物種:  Chlorocebus aethiops (ミドリザル) Chlorocebus aethiops (ミドリザル) |
| 配列 | 文字列: NDCIFEVKHE GKVTGYACLV GDKVMKPAHV KGTIDNADLA KLAFKRSSKY DLECAQIPVH MKSDASKFTH EKPEGYYNWH HGAVQYSGG RFTIPTGAGK PGDSGRPIFD NKGRVVAIVL GGANEGARTA LSVVTWNKDI VTKITPEGAE EW UniProtKB: Structural polyprotein |
-分子 #4: E3 glycoprotein
| 分子 | 名称: E3 glycoprotein / タイプ: protein_or_peptide / ID: 4 / コピー数: 4 / 光学異性体: LEVO / EC番号: togavirin |
|---|---|
| 由来(天然) | 生物種:   Chikungunya virus (チクングニヤウイルス) Chikungunya virus (チクングニヤウイルス)株: 181/25 |
| 分子量 | 理論値: 6.95507 KDa |
| 組換発現 | 生物種:  Chlorocebus aethiops (ミドリザル) Chlorocebus aethiops (ミドリザル) |
| 配列 | 文字列: PVMCLLANTT FPCSQPPCTP CCYEKEPEET LRMLEDNVMR PGYYQLLQAS LTCSPHRQRR UniProtKB: Structural polyprotein |
-分子 #5: Matrix remodeling-associated protein 8
| 分子 | 名称: Matrix remodeling-associated protein 8 / タイプ: protein_or_peptide / ID: 5 / コピー数: 1 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:  |
| 分子量 | 理論値: 30.022596 KDa |
| 組換発現 | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 配列 | 文字列: SSSLVSESVV SLAAGTQAVL RCQSPRMVWT QDRLHDRQRV VHWDLSGGPG SQRRRLVDMY SAGEQRVYEP RDRDRLLLSP SAFHDGNFS LLIRAVDRGD EGVYTCNLHH HYCHLDESLA VRLEVTEDPL LSRAYWDGEK EVLVVAHGAP ALMTCINRAH V WTDRHLEE ...文字列: SSSLVSESVV SLAAGTQAVL RCQSPRMVWT QDRLHDRQRV VHWDLSGGPG SQRRRLVDMY SAGEQRVYEP RDRDRLLLSP SAFHDGNFS LLIRAVDRGD EGVYTCNLHH HYCHLDESLA VRLEVTEDPL LSRAYWDGEK EVLVVAHGAP ALMTCINRAH V WTDRHLEE AQQVVHWDRQ LPGVSHDRAD RLLDLYASGE RRAYGPPFLR DRVSVNTNAF ARGDFSLRID ELERADEGIY SC HLHHHYC GLHERRVFHL QVTE UniProtKB: Matrix remodeling-associated protein 8 |
-分子 #6: 2-acetamido-2-deoxy-beta-D-glucopyranose
| 分子 | 名称: 2-acetamido-2-deoxy-beta-D-glucopyranose / タイプ: ligand / ID: 6 / コピー数: 5 / 式: NAG |
|---|---|
| 分子量 | 理論値: 221.208 Da |
| Chemical component information |  ChemComp-NAG: |
-実験情報
-構造解析
| 手法 | クライオ電子顕微鏡法 |
|---|---|
 解析 解析 | 単粒子再構成法 |
| 試料の集合状態 | particle |
- 試料調製
試料調製
| 緩衝液 | pH: 7.2 構成要素:
詳細: Mxra8 at pH 7.4 | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| グリッド | モデル: Quantifoil R2/2 / 材質: COPPER / メッシュ: 300 / 支持フィルム - 材質: CARBON / 支持フィルム - トポロジー: HOLEY ARRAY / 支持フィルム - Film thickness: 120 / 前処理 - タイプ: PLASMA CLEANING / 前処理 - 時間: 60 sec. / 前処理 - 雰囲気: OTHER / 詳細: 0.458 mbar.l/s O2 and 0.11 mbar.l/s H2 | ||||||||||||||
| 凍結 | 凍結剤: ETHANE / チャンバー内湿度: 100 % / チャンバー内温度: 277.00 K / 装置: FEI VITROBOT MARK IV | ||||||||||||||
| 詳細 | Chikungunya virus grown in Vero cells + 15 mg/mL Mxra8 expressed in HEK293 cells |
- 電子顕微鏡法
電子顕微鏡法
| 顕微鏡 | FEI TITAN KRIOS |
|---|---|
| 撮影 | フィルム・検出器のモデル: GATAN K2 SUMMIT (4k x 4k) 平均露光時間: 0.3 sec. / 平均電子線量: 50.0 e/Å2 |
| 電子線 | 加速電圧: 300 kV / 電子線源: OTHER |
| 電子光学系 | 照射モード: FLOOD BEAM / 撮影モード: BRIGHT FIELD |
| 実験機器 |  モデル: Titan Krios / 画像提供: FEI Company |
+ 画像解析
画像解析
-原子モデル構築 1
| 初期モデル |
| ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 精密化 | 空間: REAL / プロトコル: FLEXIBLE FIT | ||||||||||||||||
| 得られたモデル |  PDB-6nk7: |
 ムービー
ムービー コントローラー
コントローラー



 UCSF Chimera
UCSF Chimera

















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)