[English] 日本語
 Yorodumi
Yorodumi- EMDB-9239: Rabbit 80S ribosome with Z-site tRNA and IFRD2 (unrotated state) -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-9239 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Rabbit 80S ribosome with Z-site tRNA and IFRD2 (unrotated state) | |||||||||
 Map data Map data | Postprocessed map | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | translation / RIBOSOME | |||||||||
| Function / homology |  Function and homology information Function and homology information90S preribosome / ubiquitin ligase inhibitor activity / positive regulation of signal transduction by p53 class mediator / phagocytic cup / translation regulator activity / rough endoplasmic reticulum / ribosomal small subunit export from nucleus / gastrulation / MDM2/MDM4 family protein binding / cytosolic ribosome ...90S preribosome / ubiquitin ligase inhibitor activity / positive regulation of signal transduction by p53 class mediator / phagocytic cup / translation regulator activity / rough endoplasmic reticulum / ribosomal small subunit export from nucleus / gastrulation / MDM2/MDM4 family protein binding / cytosolic ribosome / ribosomal large subunit biogenesis / maturation of LSU-rRNA from tricistronic rRNA transcript (SSU-rRNA, 5.8S rRNA, LSU-rRNA) / maturation of SSU-rRNA / small-subunit processome / rRNA processing / positive regulation of canonical Wnt signaling pathway / rhythmic process / antimicrobial humoral immune response mediated by antimicrobial peptide / large ribosomal subunit / ribosomal small subunit assembly / ribosome binding / ribosomal small subunit biogenesis / 5S rRNA binding / small ribosomal subunit / ribosomal large subunit assembly / small ribosomal subunit rRNA binding / cytosolic small ribosomal subunit / large ribosomal subunit rRNA binding / killing of cells of another organism / cytosolic large ribosomal subunit / defense response to Gram-negative bacterium / perikaryon / cell differentiation / cytoplasmic translation / tRNA binding / postsynaptic density / rRNA binding / structural constituent of ribosome / ribosome / translation / ribonucleoprotein complex / mRNA binding / apoptotic process / centrosome / synapse / dendrite / nucleolus / perinuclear region of cytoplasm / endoplasmic reticulum / Golgi apparatus / RNA binding / zinc ion binding / nucleus / cytoplasm / cytosol Similarity search - Function | |||||||||
| Biological species |  | |||||||||
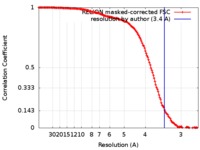
| Method | single particle reconstruction / cryo EM / Resolution: 3.4 Å | |||||||||
 Authors Authors | Brown A / Baird MR | |||||||||
 Citation Citation |  Journal: Elife / Year: 2018 Journal: Elife / Year: 2018Title: Structures of translationally inactive mammalian ribosomes. Authors: Alan Brown / Matthew R Baird / Matthew Cj Yip / Jason Murray / Sichen Shao /   Abstract: The cellular levels and activities of ribosomes directly regulate gene expression during numerous physiological processes. The mechanisms that globally repress translation are incompletely understood. ...The cellular levels and activities of ribosomes directly regulate gene expression during numerous physiological processes. The mechanisms that globally repress translation are incompletely understood. Here, we use electron cryomicroscopy to analyze inactive ribosomes isolated from mammalian reticulocytes, the penultimate stage of red blood cell differentiation. We identify two types of ribosomes that are translationally repressed by protein interactions. The first comprises ribosomes sequestered with elongation factor 2 (eEF2) by SERPINE mRNA binding protein 1 (SERBP1) occupying the ribosomal mRNA entrance channel. The second type are translationally repressed by a novel ribosome-binding protein, interferon-related developmental regulator 2 (IFRD2), which spans the P and E sites and inserts a C-terminal helix into the mRNA exit channel to preclude translation. IFRD2 binds ribosomes with a tRNA occupying a noncanonical binding site, the 'Z site', on the ribosome. These structures provide functional insights into how ribosomal interactions may suppress translation to regulate gene expression. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_9239.map.gz emd_9239.map.gz | 16.4 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-9239-v30.xml emd-9239-v30.xml emd-9239.xml emd-9239.xml | 102 KB 102 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_9239_fsc.xml emd_9239_fsc.xml | 14.1 KB | Display |  FSC data file FSC data file |
| Images |  emd_9239.png emd_9239.png | 174.1 KB | ||
| Filedesc metadata |  emd-9239.cif.gz emd-9239.cif.gz | 20.4 KB | ||
| Others |  emd_9239_additional.map.gz emd_9239_additional.map.gz emd_9239_half_map_1.map.gz emd_9239_half_map_1.map.gz emd_9239_half_map_2.map.gz emd_9239_half_map_2.map.gz | 214.8 MB 215.2 MB 214.9 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-9239 http://ftp.pdbj.org/pub/emdb/structures/EMD-9239 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-9239 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-9239 | HTTPS FTP |
-Related structure data
| Related structure data |  6mtcMC  9234C  9235C  9236C  9237C  9240C  9241C  9242C  6mtbC  6mtdC  6mteC C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_9239.map.gz / Format: CCP4 / Size: 244.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_9239.map.gz / Format: CCP4 / Size: 244.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Postprocessed map | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.34 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
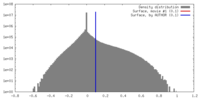
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Additional map: Pre-postprocessed map
| File | emd_9239_additional.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Pre-postprocessed map | ||||||||||||
| Projections & Slices |
| ||||||||||||
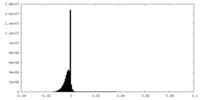
| Density Histograms |
-Half map: Half map 1
| File | emd_9239_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map 1 | ||||||||||||
| Projections & Slices |
| ||||||||||||
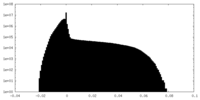
| Density Histograms |
-Half map: Half map 2
| File | emd_9239_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map 2 | ||||||||||||
| Projections & Slices |
| ||||||||||||
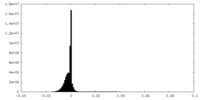
| Density Histograms |
- Sample components
Sample components
+Entire : Rabbit 80S ribosome with Z-site tRNA and IFRD2
+Supramolecule #1: Rabbit 80S ribosome with Z-site tRNA and IFRD2
+Macromolecule #1: Z-site tRNA
+Macromolecule #2: 28S rRNA
+Macromolecule #3: 5S rRNA
+Macromolecule #4: 5.8S rRNA
+Macromolecule #49: 18S rRNA
+Macromolecule #5: 60S ribosomal protein L8
+Macromolecule #6: 60S ribosomal protein L3
+Macromolecule #7: 60S ribosomal protein L4
+Macromolecule #8: 60S ribosomal protein L5
+Macromolecule #9: 60S ribosomal protein L6
+Macromolecule #10: 60S ribosomal protein L7
+Macromolecule #11: 60S ribosomal protein L7a
+Macromolecule #12: 60S ribosomal protein L9
+Macromolecule #13: 60S ribosomal protein L10
+Macromolecule #14: 60S ribosomal protein L11
+Macromolecule #15: 60S ribosomal protein L13
+Macromolecule #16: 60S ribosomal protein L14
+Macromolecule #17: 60S Ribosomal protein L15
+Macromolecule #18: 60S ribosomal protein L13a
+Macromolecule #19: 60S ribosomal protein L17
+Macromolecule #20: 60S ribosomal protein L18
+Macromolecule #21: 60S ribosomal protein L19
+Macromolecule #22: 60S ribosomal protein L18a
+Macromolecule #23: 60S ribosomal protein L21
+Macromolecule #24: 60S ribosomal protein L22
+Macromolecule #25: 60S ribosomal protein L23
+Macromolecule #26: 60S ribosomal protein L24
+Macromolecule #27: 60S ribosomal protein L23a
+Macromolecule #28: 60S ribosomal protein L26
+Macromolecule #29: 60S ribosomal protein L27
+Macromolecule #30: 60S ribosomal protein L27a
+Macromolecule #31: 60S ribosomal protein L29
+Macromolecule #32: 60S ribosomal protein L30
+Macromolecule #33: 60S ribosomal protein L31
+Macromolecule #34: 60S ribosomal protein L32
+Macromolecule #35: 60S ribosomal protein L35a
+Macromolecule #36: 60S ribosomal protein L34
+Macromolecule #37: 60S ribosomal protein L35
+Macromolecule #38: 60S ribosomal protein L36
+Macromolecule #39: 60S ribosomal protein L37
+Macromolecule #40: 60S ribosomal protein L38
+Macromolecule #41: 60S ribosomal protein L39
+Macromolecule #42: 60S ribosomal protein L40
+Macromolecule #43: 60S ribosomal protein L41
+Macromolecule #44: 60S ribosomal protein L36a
+Macromolecule #45: 60S ribosomal protein L37a
+Macromolecule #46: 60S ribosomal protein L28
+Macromolecule #47: 60S ribosomal protein L10a
+Macromolecule #48: Interferon-related developmental regulator 2
+Macromolecule #50: 40S ribosomal protein SA
+Macromolecule #51: 40S ribosomal protein S3a
+Macromolecule #52: 40S ribosomal protein S2
+Macromolecule #53: 40S ribosomal protein S3
+Macromolecule #54: 40S ribosomal protein S4, X isoform
+Macromolecule #55: 40S ribosomal protein S5
+Macromolecule #56: 40S ribosomal protein S6
+Macromolecule #57: 40S ribosomal protein S7
+Macromolecule #58: 40S ribosomal protein S8
+Macromolecule #59: 40S ribosomal protein S9
+Macromolecule #60: 40S ribosomal protein S10
+Macromolecule #61: 40S ribosomal protein S11
+Macromolecule #62: 40S ribosomal protein S12
+Macromolecule #63: 40S ribosomal protein S13
+Macromolecule #64: 40S ribosomal protein S14
+Macromolecule #65: 40S ribosomal protein S15
+Macromolecule #66: 40S ribosomal protein S16
+Macromolecule #67: 40S ribosomal protein S17
+Macromolecule #68: 40S ribosomal protein S18
+Macromolecule #69: 40S ribosomal protein S19
+Macromolecule #70: 40S ribosomal protein S20
+Macromolecule #71: 40S ribosomal protein S21
+Macromolecule #72: 40S ribosomal protein S15a
+Macromolecule #73: 40S ribosomal protein S23
+Macromolecule #74: 40S ribosomal protein S24
+Macromolecule #75: 40S ribosomal protein S25
+Macromolecule #76: 40S ribosomal protein S26
+Macromolecule #77: 40S ribosomal protein S27
+Macromolecule #78: 40S ribosomal protein S28
+Macromolecule #79: 40S ribosomal protein S29
+Macromolecule #80: 40S ribosomal protein S30
+Macromolecule #81: 40S ribosomal protein S27a
+Macromolecule #82: Receptor of activated protein C kinase 1
+Macromolecule #83: MAGNESIUM ION
+Macromolecule #84: ZINC ION
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.4 |
|---|---|
| Grid | Model: Quantifoil R2/2 / Material: COPPER / Mesh: 400 / Support film - Material: CARBON / Support film - topology: CONTINUOUS / Support film - Film thickness: 5 |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 277 K |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: FEI FALCON II (4k x 4k) / Detector mode: INTEGRATING / Digitization - Frames/image: 1-17 / Average exposure time: 1.1 sec. / Average electron dose: 40.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller


















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)