[English] 日本語
 Yorodumi
Yorodumi- EMDB-5268: Three-dimensional structure of Dengue virus serotype 1 complexed ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-5268 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Three-dimensional structure of Dengue virus serotype 1 complexed with HMAb 14c10 Fab | |||||||||
 Map data Map data | cryo-EM reconstruction of a complex of Dengue serotype 1 with 14C10 antibodies | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | virus / dengue / serotype 1 / mature virus / human antibody / neutralizing / serotype specific / Fab | |||||||||
| Function / homology |  Function and homology information Function and homology informationsymbiont-mediated suppression of host JAK-STAT cascade via inhibition of host TYK2 activity / host cell mitochondrion / symbiont-mediated suppression of host JAK-STAT cascade via inhibition of STAT2 activity / symbiont-mediated suppression of host cytoplasmic pattern recognition receptor signaling pathway via inhibition of MAVS activity / viral capsid / ribonucleoside triphosphate phosphatase activity / double-stranded RNA binding / channel activity / clathrin-dependent endocytosis of virus by host cell / monoatomic ion transmembrane transport ...symbiont-mediated suppression of host JAK-STAT cascade via inhibition of host TYK2 activity / host cell mitochondrion / symbiont-mediated suppression of host JAK-STAT cascade via inhibition of STAT2 activity / symbiont-mediated suppression of host cytoplasmic pattern recognition receptor signaling pathway via inhibition of MAVS activity / viral capsid / ribonucleoside triphosphate phosphatase activity / double-stranded RNA binding / channel activity / clathrin-dependent endocytosis of virus by host cell / monoatomic ion transmembrane transport / methyltransferase cap1 activity / mRNA 5'-cap (guanine-N7-)-methyltransferase activity / RNA helicase activity / protein dimerization activity / host cell endoplasmic reticulum membrane / symbiont-mediated suppression of host type I interferon-mediated signaling pathway / serine-type endopeptidase activity / symbiont-mediated activation of host autophagy / viral RNA genome replication / RNA-directed RNA polymerase activity / fusion of virus membrane with host endosome membrane / viral envelope / lipid binding / virion attachment to host cell / host cell nucleus / virion membrane / structural molecule activity / proteolysis / extracellular region / ATP binding / metal ion binding Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) / Homo sapiens (human) /  Dengue virus 1 Dengue virus 1 | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 7.0 Å | |||||||||
 Authors Authors | Teoh EP / Kukkaro P / Teo EW / Lim A / Tan TT / Shi PY / Yip A / Schul W / Leo S / Chan SH ...Teoh EP / Kukkaro P / Teo EW / Lim A / Tan TT / Shi PY / Yip A / Schul W / Leo S / Chan SH / Smith KGC / Ooi EE / Kemeny DM / Ng G / Ng ML / Alonso S / Fisher D / Hanson B / Lok SM / MacAry PA | |||||||||
 Citation Citation |  Journal: Sci Transl Med / Year: 2012 Journal: Sci Transl Med / Year: 2012Title: The structural basis for serotype-specific neutralization of dengue virus by a human antibody. Authors: Ee Ping Teoh / Petra Kukkaro / En Wei Teo / Angeline P C Lim / Tze Tong Tan / Andy Yip / Wouter Schul / Myint Aung / Victor A Kostyuchenko / Yee Sin Leo / Soh Ha Chan / Kenneth G C Smith / ...Authors: Ee Ping Teoh / Petra Kukkaro / En Wei Teo / Angeline P C Lim / Tze Tong Tan / Andy Yip / Wouter Schul / Myint Aung / Victor A Kostyuchenko / Yee Sin Leo / Soh Ha Chan / Kenneth G C Smith / Annie Hoi Yi Chan / Gang Zou / Eng Eong Ooi / D Michael Kemeny / Grace K Tan / Jowin K W Ng / Mah Lee Ng / Sylvie Alonso / Dale Fisher / Pei-Yong Shi / Brendon J Hanson / Shee-Mei Lok / Paul A MacAry /  Abstract: Dengue virus (DENV) is a mosquito-borne flavivirus that affects 2.5 billion people worldwide. There are four dengue serotypes (DENV1 to DENV4), and infection with one elicits lifelong immunity to ...Dengue virus (DENV) is a mosquito-borne flavivirus that affects 2.5 billion people worldwide. There are four dengue serotypes (DENV1 to DENV4), and infection with one elicits lifelong immunity to that serotype but offers only transient protection against the other serotypes. Identification of the protective determinants of the human antibody response to DENV is a vital requirement for the design and evaluation of future preventative therapies and treatments. Here, we describe the isolation of a neutralizing antibody from a DENV1-infected patient. The human antibody 14c10 (HM14c10) binds specifically to DENV1. HM14c10 neutralizes the virus principally by blocking virus attachment; at higher concentrations, a post-attachment step can also be inhibited. In vivo studies show that the HM14c10 antibody has antiviral activity at picomolar concentrations. A 7 Å resolution cryoelectron microscopy map of Fab fragments of HM14c10 in a complex with DENV1 shows targeting of a discontinuous epitope that spans the adjacent surface of envelope protein dimers. As found previously, a human antibody specific for the related West Nile virus binds to a similar quaternary structure, suggesting that this could be an immunodominant epitope. These findings provide a structural and molecular context for durable, serotype-specific immunity to DENV infection. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_5268.map.gz emd_5268.map.gz | 421.8 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-5268-v30.xml emd-5268-v30.xml emd-5268.xml emd-5268.xml | 13 KB 13 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_5268_1.png emd_5268_1.png | 412 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-5268 http://ftp.pdbj.org/pub/emdb/structures/EMD-5268 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-5268 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-5268 | HTTPS FTP |
-Related structure data
| Related structure data |  3j05MC  4cauMC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_5268.map.gz / Format: CCP4 / Size: 465.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_5268.map.gz / Format: CCP4 / Size: 465.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | cryo-EM reconstruction of a complex of Dengue serotype 1 with 14C10 antibodies | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.9 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
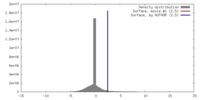
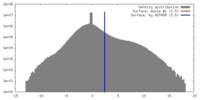
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : Dengue virus serotype 1 with Fab fragment of human monoclonal ant...
| Entire | Name: Dengue virus serotype 1 with Fab fragment of human monoclonal antibody 14c10 |
|---|---|
| Components |
|
-Supramolecule #1000: Dengue virus serotype 1 with Fab fragment of human monoclonal ant...
| Supramolecule | Name: Dengue virus serotype 1 with Fab fragment of human monoclonal antibody 14c10 type: sample / ID: 1000 Oligomeric state: 120 Fab molecules bind to one dengue virion Number unique components: 2 |
|---|
-Supramolecule #1: Dengue virus 1
| Supramolecule | Name: Dengue virus 1 / type: virus / ID: 1 / Name.synonym: Dengue 1 / Details: strain PVP 159 / NCBI-ID: 11053 / Sci species name: Dengue virus 1 / Database: NCBI / Virus type: VIRION / Virus isolate: STRAIN / Virus enveloped: Yes / Virus empty: No / Syn species name: Dengue 1 |
|---|---|
| Host (natural) | Organism:  |
| Virus shell | Shell ID: 1 / Diameter: 240 Å / T number (triangulation number): 3 |
-Macromolecule #1: antibody Fab fragment
| Macromolecule | Name: antibody Fab fragment / type: protein_or_peptide / ID: 1 / Name.synonym: Fab / Oligomeric state: monomer / Recombinant expression: Yes |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) / synonym: Human / Tissue: blood / Cell: CD22plus B cells / Location in cell: secreted Homo sapiens (human) / synonym: Human / Tissue: blood / Cell: CD22plus B cells / Location in cell: secreted |
| Molecular weight | Experimental: 50 MDa |
| Recombinant expression | Organism:  Homo sapiens (human) / Recombinant plasmid: pTT5 Homo sapiens (human) / Recombinant plasmid: pTT5 |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 8 / Details: 120 mM NaCl, 12 mM Tris-HCl, pH 8.0, 1mM EDTA |
|---|---|
| Grid | Details: lacey carbon copper grid |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Instrument: OTHER / Details: Vitrification instrument: Vitrobot / Method: 1 second blotting |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Temperature | Average: 100 K |
| Date | Sep 27, 2010 |
| Image recording | Category: CCD / Film or detector model: GENERIC GATAN / Digitization - Sampling interval: 15 µm / Number real images: 341 / Average electron dose: 16 e/Å2 / Details: 4k x 4k Gatan CCD / Bits/pixel: 16 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: SPOT SCAN / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 4.9 µm / Nominal defocus min: 0.76 µm / Nominal magnification: 47000 |
| Sample stage | Specimen holder model: OTHER |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Details | The particles were selected manually. |
|---|---|
| CTF correction | Details: Each particle |
| Final reconstruction | Algorithm: OTHER / Resolution.type: BY AUTHOR / Resolution: 7.0 Å / Resolution method: FSC 0.5 CUT-OFF / Software - Name: EMAN, MPSA / Number images used: 2129 |
-Atomic model buiding 1
| Initial model | PDB ID: |
|---|---|
| Software | Name:  Chimera Chimera |
| Details | Protocol: rigid body. The domains were fitted separately using Fit In Map feature in Chimera |
| Refinement | Space: REAL / Protocol: RIGID BODY FIT / Target criteria: correlation |
| Output model |  PDB-3j05:  PDB-4cau: |
 Movie
Movie Controller
Controller















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)