+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Human GluN1-2A with IgG 007-168 | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Channel / heterotetramer / receptor / antibody / MEMBRANE PROTEIN-IMMUNE SYSTEM complex | |||||||||
| Function / homology |  Function and homology information Function and homology informationglycine-gated cation channel activity / excitatory chemical synaptic transmission / Synaptic adhesion-like molecules / propylene metabolic process / response to glycine / Assembly and cell surface presentation of NMDA receptors / neurotransmitter receptor complex / Neurexins and neuroligins / regulation of monoatomic cation transmembrane transport / NMDA glutamate receptor activity ...glycine-gated cation channel activity / excitatory chemical synaptic transmission / Synaptic adhesion-like molecules / propylene metabolic process / response to glycine / Assembly and cell surface presentation of NMDA receptors / neurotransmitter receptor complex / Neurexins and neuroligins / regulation of monoatomic cation transmembrane transport / NMDA glutamate receptor activity / NMDA selective glutamate receptor complex / glutamate binding / ligand-gated sodium channel activity / calcium ion transmembrane import into cytosol / protein heterotetramerization / glycine binding / positive regulation of reactive oxygen species biosynthetic process / Negative regulation of NMDA receptor-mediated neuronal transmission / Unblocking of NMDA receptors, glutamate binding and activation / monoatomic cation transmembrane transport / Long-term potentiation / positive regulation of calcium ion transport into cytosol / regulation of neuronal synaptic plasticity / monoatomic cation transport / ligand-gated monoatomic ion channel activity / monoatomic cation channel activity / synaptic cleft / calcium ion homeostasis / positive regulation of synaptic transmission, glutamatergic / EPHB-mediated forward signaling / glutamate-gated calcium ion channel activity / excitatory synapse / ionotropic glutamate receptor signaling pathway / cytoplasmic vesicle membrane / Ras activation upon Ca2+ influx through NMDA receptor / positive regulation of excitatory postsynaptic potential / sodium ion transmembrane transport / synaptic membrane / regulation of membrane potential / excitatory postsynaptic potential / brain development / visual learning / regulation of synaptic plasticity / postsynaptic density membrane / calcium ion transmembrane transport / terminal bouton / synaptic vesicle / amyloid-beta binding / RAF/MAP kinase cascade / signaling receptor activity / dendritic spine / chemical synaptic transmission / response to ethanol / learning or memory / calmodulin binding / postsynaptic membrane / neuron projection / postsynaptic density / calcium ion binding / synapse / dendrite / endoplasmic reticulum membrane / protein-containing complex binding / cell surface / endoplasmic reticulum / positive regulation of transcription by RNA polymerase II / metal ion binding / plasma membrane / cytoplasm Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.99 Å | |||||||||
 Authors Authors | Michalski K / Furukawa H | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: Nat Struct Mol Biol / Year: 2024 Journal: Nat Struct Mol Biol / Year: 2024Title: Structural and functional mechanisms of anti-NMDAR autoimmune encephalitis. Authors: Kevin Michalski / Taha Abdulla / Sam Kleeman / Lars Schmidl / Ricardo Gómez / Noriko Simorowski / Francesca Vallese / Harald Prüss / Manfred Heckmann / Christian Geis / Hiro Furukawa /   Abstract: Autoantibodies against neuronal membrane proteins can manifest in autoimmune encephalitis, inducing seizures, cognitive dysfunction and psychosis. Anti-N-methyl-D-aspartate receptor (NMDAR) ...Autoantibodies against neuronal membrane proteins can manifest in autoimmune encephalitis, inducing seizures, cognitive dysfunction and psychosis. Anti-N-methyl-D-aspartate receptor (NMDAR) encephalitis is the most dominant autoimmune encephalitis; however, insights into how autoantibodies recognize and alter receptor functions remain limited. Here we determined structures of human and rat NMDARs bound to three distinct patient-derived antibodies using single-particle electron cryo-microscopy. These antibodies bind different regions within the amino-terminal domain of the GluN1 subunit. Through electrophysiology, we show that all three autoantibodies acutely and directly reduced NMDAR channel functions in primary neurons. Antibodies show different stoichiometry of binding and antibody-receptor complex formation, which in one antibody, 003-102, also results in reduced synaptic localization of NMDARs. These studies demonstrate mechanisms of diverse epitope recognition and direct channel regulation of anti-NMDAR autoantibodies underlying autoimmune encephalitis. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_43538.map.gz emd_43538.map.gz | 307.1 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-43538-v30.xml emd-43538-v30.xml emd-43538.xml emd-43538.xml | 23.7 KB 23.7 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_43538.png emd_43538.png | 84.4 KB | ||
| Filedesc metadata |  emd-43538.cif.gz emd-43538.cif.gz | 7.3 KB | ||
| Others |  emd_43538_half_map_1.map.gz emd_43538_half_map_1.map.gz emd_43538_half_map_2.map.gz emd_43538_half_map_2.map.gz | 301.9 MB 301.9 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-43538 http://ftp.pdbj.org/pub/emdb/structures/EMD-43538 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-43538 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-43538 | HTTPS FTP |
-Related structure data
| Related structure data |  8vusMC  8vuhC  8vujC  8vulC  8vunC  8vuqC  8vurC  8vutC  8vuuC  8vuvC  8vuyC  8vvhC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_43538.map.gz / Format: CCP4 / Size: 325 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_43538.map.gz / Format: CCP4 / Size: 325 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.856 Å | ||||||||||||||||||||||||||||||||||||
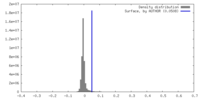
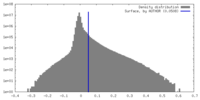
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #1
| File | emd_43538_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
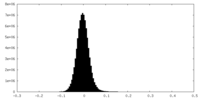
| Density Histograms |
-Half map: #2
| File | emd_43538_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
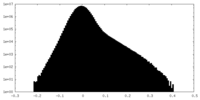
| Density Histograms |
- Sample components
Sample components
-Entire : Human GluN1-2A with IgG 007-168
| Entire | Name: Human GluN1-2A with IgG 007-168 |
|---|---|
| Components |
|
-Supramolecule #1: Human GluN1-2A with IgG 007-168
| Supramolecule | Name: Human GluN1-2A with IgG 007-168 / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: 007-168 Heavy
| Macromolecule | Name: 007-168 Heavy / type: protein_or_peptide / ID: 1 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 22.52225 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: QVQLVQSGAE AKKPGESLKI SCKASGYSFT TFWIGWVRQM PGSGLEWIGI IYPGDSDTRY SPSFQGHVTI SADRSTSTAY LQWSSLKAS DTAMYYCARS AVFDYWGQGT LVTVSSASTK GPSVFPLAPS SGTAALGCLV KDYFPEPVTV SWNSGALTSG V HTFPAVLQ ...String: QVQLVQSGAE AKKPGESLKI SCKASGYSFT TFWIGWVRQM PGSGLEWIGI IYPGDSDTRY SPSFQGHVTI SADRSTSTAY LQWSSLKAS DTAMYYCARS AVFDYWGQGT LVTVSSASTK GPSVFPLAPS SGTAALGCLV KDYFPEPVTV SWNSGALTSG V HTFPAVLQ SSGLYSLSSV VTVPSSSLGT QTYICNVNHK PSNTKVDKKV EPK |
-Macromolecule #2: 007-168 Light
| Macromolecule | Name: 007-168 Light / type: protein_or_peptide / ID: 2 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 23.455982 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: EIVMTQSPAT LSVSPGGRAT LSCRASQSVS SNLAWYQQKP GQAPRLLIYG ASTRATGIPV RFSGSGSGTE FTLTISSLQS EDFAVYYCQ QYNNWPTSWT FGQGTKVEIK RTVAAPSVFI FPPSDEQLKS GTASVVCLLN NFYPREAKVQ WKVDNALQSG N SQESVTEQ ...String: EIVMTQSPAT LSVSPGGRAT LSCRASQSVS SNLAWYQQKP GQAPRLLIYG ASTRATGIPV RFSGSGSGTE FTLTISSLQS EDFAVYYCQ QYNNWPTSWT FGQGTKVEIK RTVAAPSVFI FPPSDEQLKS GTASVVCLLN NFYPREAKVQ WKVDNALQSG N SQESVTEQ DSKDSTYSLS STLTLSKADY EKHKVYACEV THQGLSSPVT KSFNRGE |
-Macromolecule #3: Glutamate receptor ionotropic, NMDA 1
| Macromolecule | Name: Glutamate receptor ionotropic, NMDA 1 / type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 87.477797 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: DPKIVNIGAV LSTRKHEQMF REAVNQANKR HGSWKIQLNA TSVTHKPNAI QMALSVCEDL ISSQVYAILV SHPPTPNDHF TPTPVSYTA GFYRIPVLGL TTRMSIYSDK SIHLSFLRTV PPYSHQSSVW FEMMRVYSWN HIILLVSDDH EGRAAQKRLE T LLEERESK ...String: DPKIVNIGAV LSTRKHEQMF REAVNQANKR HGSWKIQLNA TSVTHKPNAI QMALSVCEDL ISSQVYAILV SHPPTPNDHF TPTPVSYTA GFYRIPVLGL TTRMSIYSDK SIHLSFLRTV PPYSHQSSVW FEMMRVYSWN HIILLVSDDH EGRAAQKRLE T LLEERESK AEKVLQFDPG TKNVTALLME AKELEARVII LSASEDDAAT VYRAAAMLNM TGSGYVWLVG EREISGNALR YA PDGILGL QLINGKNESA HISDAVGVVA QAVHELLEKE NITDPPRGCV GNTNIWKTGP LFKRVLMSSK YADGVTGRVE FNE DGDRKF ANYSIMNLQN RKLVQVGIYN GTHVIPNDRK IIWPGGETEK PRGYQMSTRL KIVTIHQEPF VYVKPTLSDG TCKE EFTVN GDPVKKVICT GPNDTSPGSP RHTVPQCCYG FCIDLLIKLA RTMNFTYEVH LVADGKFGTQ ERVNNSNKKE WNGMM GELL SGQADMIVAP LTINNERAQY IEFSKPFKYQ GLTILVKKEI PRSTLDSFMQ PFQSTLWLLV GLSVHVVAVM LYLLDR FSP FGRFKVNSEE EEEDALTLSS AMWFSWGVLL NSGIGEGAPR SFSARILGMV WAGFAMIIVA SYTANLAAFL VLDRPEE RI TGINDPRLRN PSDKFIYATV KQSSVDIYFR RQVELSTMYR HMEKHNYESA AEAIQAVRDN KLHAFIWDSA VLEFEASQ K CDLVTTGELF FRSGFGIGMR KDSPWKQNVS LSILKSHENG FMEDLDKTWV RYQEC UniProtKB: Glutamate receptor ionotropic, NMDA 1 |
-Macromolecule #4: Glutamate receptor ionotropic, NMDA 2A
| Macromolecule | Name: Glutamate receptor ionotropic, NMDA 2A / type: protein_or_peptide / ID: 4 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 86.50793 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: LNIAVMLGHS HDVTERELRT LWGPEQAAGL PLDVNVVALL MNRTDPKSLI THVCDLMSGA RIHGLVFGDD TDQEAVAQML DFISSHTFV PILGIHGGAS MIMADKDPTS TFFQFGASIQ QQATVMLKIM QDYDWHVFSL VTTIFPGYRE FISFVKTTVD N SFVGWDMQ ...String: LNIAVMLGHS HDVTERELRT LWGPEQAAGL PLDVNVVALL MNRTDPKSLI THVCDLMSGA RIHGLVFGDD TDQEAVAQML DFISSHTFV PILGIHGGAS MIMADKDPTS TFFQFGASIQ QQATVMLKIM QDYDWHVFSL VTTIFPGYRE FISFVKTTVD N SFVGWDMQ NVITLDTSFE DAKTQVQLKK IHSSVILLYC SKDEAVLILS EARSLGLTGY DFFWIVPSLV SGNTELIPKE FP SGLISVS YDDWDYSLEA RVRDGIGILT TAASSMLEKF SYIPEAKASC YGQMERPEVP MHTLHPFMVN VTWDGKDLSF TEE GYQVHP RLVVIVLNKD REWEKVGKWE NHTLSLRHAV WPRYKSFSDC EPDDNHLSIV TLEEAPFVIV EDIDPLTETC VRNT VPCRK FVKINNSTNE GMNVKKCCKG FCIDILKKLS RTVKFTYDLY LVTNGKHGKK VNNVWNGMIG EVVYQRAVMA VGSLT INEE RSEVVDFSVP FVETGISVMV SRSNGTVSPS AFLEPFSASV WVMMFVMLLI VSAIAVFVFE YFSPVGYNRC LADGRE PGG PSFTIGKAIW LLWGLVFNNS VPVQNPKGTT SKIMVSVWAF FAVIFLASYT ANLAAFMIQE EFVDQVTGLS DKKFQRP HD YSPPFRFGTV PNGSTERNIR NNYPYMHQYM TKFNQKGVED ALVSLKTGKL DAFIYDAAVL NYKAGRDEGC KLVTIGSG Y IFATTGYGIA LQKGSPWKRQ IDLALLQFVG DGEMEELETL WLTGICHNEK UniProtKB: Glutamate receptor ionotropic, NMDA 2A |
-Macromolecule #5: Glutamate receptor ionotropic, NMDA 1
| Macromolecule | Name: Glutamate receptor ionotropic, NMDA 1 / type: protein_or_peptide / ID: 5 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 87.534922 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: DPKIVNIGAV LSTRKHEQMF REAVNQANKR HASWKIQLNA TSVTHKPNAI QMALSVCEDL ISSQVYAILV SHPPTPNDHF TPTPVSYTA GFYRIPVLGL TTRMSIYSDK SIHLSFLRTV PPYSHQSSVW FEMMRVYSWN HIILLVSDDH EGRAAQKRLE T LLEERESK ...String: DPKIVNIGAV LSTRKHEQMF REAVNQANKR HASWKIQLNA TSVTHKPNAI QMALSVCEDL ISSQVYAILV SHPPTPNDHF TPTPVSYTA GFYRIPVLGL TTRMSIYSDK SIHLSFLRTV PPYSHQSSVW FEMMRVYSWN HIILLVSDDH EGRAAQKRLE T LLEERESK AEKVLQFDPG TKNVTALLME AKELEARVII LSASEDDAAT VYRAAAMLNM TGSGYVWLVG EREISGNALR YA PDGILGL QLINGKNESA HISDAVGVVA QAVHELLEKE NITDPPRGCV GNTNIWKTGP LFKRVLMSSK YADGVTGRVE FNE DGDRKF ANYSIMNLQR RKLVQVGIYN GTHVIPNDRK IIWPGGETEK PRGYQMSTRL KIVTIHQEPF VYVKPTLSDG TCKE EFTVN GDPVKKVICT GPNDTSPGSP RHTVPQCCYG FCIDLLIKLA RTMNFTYEVH LVADGKFGTQ ERVNNSNKKE WNGMM GELL SGQADMIVAP LTINNERAQY IEFSKPFKYQ GLTILVKKEI PRSTLDSFMQ PFQSTLWLLV GLSVHVVAVM LYLLDR FSP FGRFKVNSEE EEEDALTLSS AMWFSWGVLL NSGIGEGAPR SFSARILGMV WAGFAMIIVA SYTANLAAFL VLDRPEE RI TGINDPRLRN PSDKFIYATV KQSSVDIYFR RQVELSTMYR HMEKHNYESA AEAIQAVRDN KLHAFIWDSA VLEFEASQ K CDLVTTGELF FRSGFGIGMR KDSPWKQNVS LSILKSHENG FMEDLDKTWV RYQEC UniProtKB: Glutamate receptor ionotropic, NMDA 1 |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 60.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: OTHER / Imaging mode: OTHER / Cs: 2.7 mm / Nominal defocus max: 2.2 µm / Nominal defocus min: 0.8 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller

























 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)