[English] 日本語
 Yorodumi
Yorodumi- EMDB-29972: CryoEM structure of beta-2-adrenergic receptor in complex with nu... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
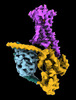
| Title | CryoEM structure of beta-2-adrenergic receptor in complex with nucleotide-free Gs heterotrimer (#20 of 20) | |||||||||
 Map data Map data | Sharpened map | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | GPCR / Adrenergic / Receptor / G protein / SIGNALING PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationbeta2-adrenergic receptor activity / positive regulation of mini excitatory postsynaptic potential / AMPA selective glutamate receptor signaling pathway / norepinephrine-epinephrine-mediated vasodilation involved in regulation of systemic arterial blood pressure / adenylate cyclase-inhibiting adrenergic receptor signaling pathway / positive regulation of autophagosome maturation / heat generation / norepinephrine binding / Adrenoceptors / negative regulation of smooth muscle contraction ...beta2-adrenergic receptor activity / positive regulation of mini excitatory postsynaptic potential / AMPA selective glutamate receptor signaling pathway / norepinephrine-epinephrine-mediated vasodilation involved in regulation of systemic arterial blood pressure / adenylate cyclase-inhibiting adrenergic receptor signaling pathway / positive regulation of autophagosome maturation / heat generation / norepinephrine binding / Adrenoceptors / negative regulation of smooth muscle contraction / positive regulation of cardiac muscle cell contraction / positive regulation of lipophagy / negative regulation of multicellular organism growth / negative regulation of G protein-coupled receptor signaling pathway / adrenergic receptor signaling pathway / diet induced thermogenesis / response to psychosocial stress / endosome to lysosome transport / adenylate cyclase-activating G protein-coupled bile acid receptor signaling pathway / adenylate cyclase-activating serotonin receptor signaling pathway / positive regulation of cAMP/PKA signal transduction / regulation of skeletal muscle contraction / smooth muscle contraction / negative regulation of cardiac muscle cell apoptotic process / adenylate cyclase binding / PKA activation in glucagon signalling / hair follicle placode formation / developmental growth / intracellular transport / potassium channel regulator activity / bone resorption / positive regulation of bone mineralization / D1 dopamine receptor binding / neuronal dense core vesicle / vascular endothelial cell response to laminar fluid shear stress / renal water homeostasis / intercellular bridge / activation of adenylate cyclase activity / Hedgehog 'off' state / regulation of sodium ion transport / cellular response to acidic pH / adenylate cyclase-activating adrenergic receptor signaling pathway / positive regulation of cardiac muscle cell apoptotic process / cellular response to glucagon stimulus / intracellular glucose homeostasis / receptor-mediated endocytosis / brown fat cell differentiation / response to cold / positive regulation of insulin secretion involved in cellular response to glucose stimulus / adenylate cyclase activator activity / trans-Golgi network membrane / clathrin-coated endocytic vesicle membrane / negative regulation of inflammatory response to antigenic stimulus / response to prostaglandin E / bone development / platelet aggregation / cognition / cellular response to amyloid-beta / G-protein beta/gamma-subunit complex binding / adenylate cyclase-modulating G protein-coupled receptor signaling pathway / Olfactory Signaling Pathway / Activation of the phototransduction cascade / positive regulation of insulin secretion / G protein-coupled acetylcholine receptor signaling pathway / G beta:gamma signalling through PLC beta / Presynaptic function of Kainate receptors / Thromboxane signalling through TP receptor / Activation of G protein gated Potassium channels / Inhibition of voltage gated Ca2+ channels via Gbeta/gamma subunits / G-protein activation / Glucagon signaling in metabolic regulation / G beta:gamma signalling through CDC42 / Prostacyclin signalling through prostacyclin receptor / sensory perception of smell / mitotic spindle / Synthesis, secretion, and inactivation of Glucagon-like Peptide-1 (GLP-1) / G beta:gamma signalling through BTK / photoreceptor disc membrane / ADP signalling through P2Y purinoceptor 12 / Sensory perception of sweet, bitter, and umami (glutamate) taste / Glucagon-type ligand receptors / Adrenaline,noradrenaline inhibits insulin secretion / Vasopressin regulates renal water homeostasis via Aquaporins / Glucagon-like Peptide-1 (GLP1) regulates insulin secretion / G alpha (z) signalling events / cellular response to catecholamine stimulus / ADP signalling through P2Y purinoceptor 1 / ADORA2B mediated anti-inflammatory cytokines production / G beta:gamma signalling through PI3Kgamma / Cargo recognition for clathrin-mediated endocytosis / adenylate cyclase-activating dopamine receptor signaling pathway / Cooperation of PDCL (PhLP1) and TRiC/CCT in G-protein beta folding / GPER1 signaling / cellular response to prostaglandin E stimulus / positive regulation of cold-induced thermogenesis / heterotrimeric G-protein complex / G alpha (12/13) signalling events / Inactivation, recovery and regulation of the phototransduction cascade / amyloid-beta binding / G-protein beta-subunit binding Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 4.2 Å | |||||||||
 Authors Authors | Papasergi-Scott MM / Skiniotis G | |||||||||
| Funding support |  United States, 2 items United States, 2 items
| |||||||||
 Citation Citation |  Journal: Nature / Year: 2024 Journal: Nature / Year: 2024Title: Time-resolved cryo-EM of G-protein activation by a GPCR. Authors: Makaía M Papasergi-Scott / Guillermo Pérez-Hernández / Hossein Batebi / Yang Gao / Gözde Eskici / Alpay B Seven / Ouliana Panova / Daniel Hilger / Marina Casiraghi / Feng He / Luis Maul ...Authors: Makaía M Papasergi-Scott / Guillermo Pérez-Hernández / Hossein Batebi / Yang Gao / Gözde Eskici / Alpay B Seven / Ouliana Panova / Daniel Hilger / Marina Casiraghi / Feng He / Luis Maul / Peter Gmeiner / Brian K Kobilka / Peter W Hildebrand / Georgios Skiniotis /    Abstract: G-protein-coupled receptors (GPCRs) activate heterotrimeric G proteins by stimulating guanine nucleotide exchange in the Gα subunit. To visualize this mechanism, we developed a time-resolved cryo-EM ...G-protein-coupled receptors (GPCRs) activate heterotrimeric G proteins by stimulating guanine nucleotide exchange in the Gα subunit. To visualize this mechanism, we developed a time-resolved cryo-EM approach that examines the progression of ensembles of pre-steady-state intermediates of a GPCR-G-protein complex. By monitoring the transitions of the stimulatory G protein in complex with the β-adrenergic receptor at short sequential time points after GTP addition, we identified the conformational trajectory underlying G-protein activation and functional dissociation from the receptor. Twenty structures generated from sequential overlapping particle subsets along this trajectory, compared to control structures, provide a high-resolution description of the order of main events driving G-protein activation in response to GTP binding. Structural changes propagate from the nucleotide-binding pocket and extend through the GTPase domain, enacting alterations to Gα switch regions and the α5 helix that weaken the G-protein-receptor interface. Molecular dynamics simulations with late structures in the cryo-EM trajectory support that enhanced ordering of GTP on closure of the α-helical domain against the nucleotide-bound Ras-homology domain correlates with α5 helix destabilization and eventual dissociation of the G protein from the GPCR. These findings also highlight the potential of time-resolved cryo-EM as a tool for mechanistic dissection of GPCR signalling events. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_29972.map.gz emd_29972.map.gz | 97.2 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-29972-v30.xml emd-29972-v30.xml emd-29972.xml emd-29972.xml | 22.4 KB 22.4 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_29972_fsc.xml emd_29972_fsc.xml | 9.9 KB | Display |  FSC data file FSC data file |
| Images |  emd_29972.png emd_29972.png | 91.3 KB | ||
| Masks |  emd_29972_msk_1.map emd_29972_msk_1.map | 103 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-29972.cif.gz emd-29972.cif.gz | 6.9 KB | ||
| Others |  emd_29972_additional_1.map.gz emd_29972_additional_1.map.gz emd_29972_half_map_1.map.gz emd_29972_half_map_1.map.gz emd_29972_half_map_2.map.gz emd_29972_half_map_2.map.gz | 51.5 MB 95.7 MB 95.7 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-29972 http://ftp.pdbj.org/pub/emdb/structures/EMD-29972 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-29972 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-29972 | HTTPS FTP |
-Related structure data
| Related structure data |  8gejMC  8gdzC  8ge1C  8ge2C  8ge3C  8ge4C  8ge5C  8ge6C  8ge7C  8ge8C  8ge9C  8geaC  8gebC  8gecC  8gedC  8geeC  8gefC  8gegC  8gehC  8geiC  8gfvC  8gfwC  8gfxC  8gfyC  8gfzC  8gg0C  8gg1C  8gg2C  8gg3C  8gg4C  8gg5C  8gg6C  8gg7C  8gg8C  8gg9C  8ggaC  8ggbC  8ggcC  8ggeC  8ggfC  8ggiC  8ggjC  8ggkC  8gglC  8ggmC  8ggnC  8ggoC  8ggpC  8ggqC  8ggrC  8ggsC  8ggtC  8gguC  8ggvC  8ggwC  8ggxC  8ggyC  8ggzC  8gh0C  8gh1C  8unlC  8unmC  8unnC  8unoC  8unpC  8unqC  8unrC  8unsC  8untC  8unuC  8unvC  8unwC  8unxC  8unyC  8unzC  8uo0C  8uo1C  8uo2C  8uo3C  8uo4C M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_29972.map.gz / Format: CCP4 / Size: 103 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_29972.map.gz / Format: CCP4 / Size: 103 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Sharpened map | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.06 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Mask #1
| File |  emd_29972_msk_1.map emd_29972_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
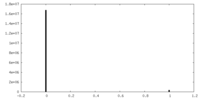
| Density Histograms |
-Additional map: Unsharpened map
| File | emd_29972_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Unsharpened map | ||||||||||||
| Projections & Slices |
| ||||||||||||
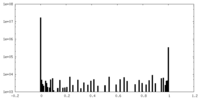
| Density Histograms |
-Half map: Half map
| File | emd_29972_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map | ||||||||||||
| Projections & Slices |
| ||||||||||||
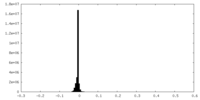
| Density Histograms |
-Half map: Half map
| File | emd_29972_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map | ||||||||||||
| Projections & Slices |
| ||||||||||||
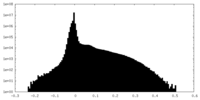
| Density Histograms |
- Sample components
Sample components
-Entire : Complex of beta-2 adrenergic receptor and Gs heterotrimer
| Entire | Name: Complex of beta-2 adrenergic receptor and Gs heterotrimer |
|---|---|
| Components |
|
-Supramolecule #1: Complex of beta-2 adrenergic receptor and Gs heterotrimer
| Supramolecule | Name: Complex of beta-2 adrenergic receptor and Gs heterotrimer type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#4 |
|---|
-Macromolecule #1: Guanine nucleotide-binding protein G(s) subunit alpha isoforms short
| Macromolecule | Name: Guanine nucleotide-binding protein G(s) subunit alpha isoforms short type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 44.32616 KDa |
| Recombinant expression | Organism:  Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Sequence | String: MGCLGNSKTE DQRNEEKAQR EANKKIEKQL QKDKQVYRAT HRLLLLGAGE SGKSTIVKQM RILHVNGFNG DSEKATKVQD IKNNLKEAI ETIVAAMSNL VPPVELANPE NQFRVDYILS VMNVPDFDFP PEFYEHAKAL WEDEGVRACY ERSNEYQLID C AQYFLDKI ...String: MGCLGNSKTE DQRNEEKAQR EANKKIEKQL QKDKQVYRAT HRLLLLGAGE SGKSTIVKQM RILHVNGFNG DSEKATKVQD IKNNLKEAI ETIVAAMSNL VPPVELANPE NQFRVDYILS VMNVPDFDFP PEFYEHAKAL WEDEGVRACY ERSNEYQLID C AQYFLDKI DVIKQADYVP SDQDLLRCRV LTSGIFETKF QVDKVNFHMF DVGGQRDERR KWIQCFNDVT AIIFVVASSS YN MVIREDN QTNRLQEALN LFKSIWNNRW LRTISVILFL NKQDLLAEKV LAGKSKIEDY FPEFARYTTP EDATPEPGED PRV TRAKYF IRDEFLRIST ASGDGRHYCY PHFTCAVDTE NIRRVFNDCR DIIQRMHLRQ YELL UniProtKB: Guanine nucleotide-binding protein G(s) subunit alpha isoforms short |
-Macromolecule #2: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1
| Macromolecule | Name: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1 type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 37.573988 KDa |
| Recombinant expression | Organism:  Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Sequence | String: GSSGSELDQL RQEAEQLKNQ IRDARKACAD ATLSQITNNI DPVGRIQMRT RRTLRGHLAK IYAMHWGTDS RLLVSASQDG KLIIWDSYT TNKVHAIPLR SSWVMTCAYA PSGNYVACGG LDNICSIYNL KTREGNVRVS RELAGHTGYL SCCRFLDDNQ I VTSSGDTT ...String: GSSGSELDQL RQEAEQLKNQ IRDARKACAD ATLSQITNNI DPVGRIQMRT RRTLRGHLAK IYAMHWGTDS RLLVSASQDG KLIIWDSYT TNKVHAIPLR SSWVMTCAYA PSGNYVACGG LDNICSIYNL KTREGNVRVS RELAGHTGYL SCCRFLDDNQ I VTSSGDTT CALWDIETGQ QTTTFTGHTG DVMSLSLAPD TRLFVSGACD ASAKLWDVRE GMCRQTFTGH ESDINAICFF PN GNAFATG SDDATCRLFD LRADQELMTY SHDNIICGIT SVSFSKSGRL LLAGYDDFNC NVWDALKADR AGVLAGHDNR VSC LGVTDD GMAVATGSWD SFLKIWN UniProtKB: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1 |
-Macromolecule #3: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2
| Macromolecule | Name: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2 type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 7.861143 KDa |
| Recombinant expression | Organism:  Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Sequence | String: MASNNTASIA QARKLVEQLK MEANIDRIKV SKAAADLMAY CEAHAKEDPL LTPVPASENP FREKKFFCAI L UniProtKB: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2 |
-Macromolecule #4: Beta-2 adrenergic receptor
| Macromolecule | Name: Beta-2 adrenergic receptor / type: protein_or_peptide / ID: 4 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 51.767242 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MKTIIALSYI FCLVFADYKD DDDAMGQPGN GSAFLLAPNR SHAPDHDVEN LYFQGTQQRD EVWVVGMGIV MSLIVLAIVF GNVLVITAI AKFERLQTVT NYFITSLACA DLVMGLAVVP FGAAHILTKT WTFGNFWCEF WTSIDVLCVT ASIETLCVIA V DRYFAITS ...String: MKTIIALSYI FCLVFADYKD DDDAMGQPGN GSAFLLAPNR SHAPDHDVEN LYFQGTQQRD EVWVVGMGIV MSLIVLAIVF GNVLVITAI AKFERLQTVT NYFITSLACA DLVMGLAVVP FGAAHILTKT WTFGNFWCEF WTSIDVLCVT ASIETLCVIA V DRYFAITS PFKYQSLLTK NKARVIILMV WIVSGLTSFL PIQMHWYRAT HQEAINCYAE ETCCDFFTNQ AYAIASSIVS FY VPLVIMV FVYSRVFQEA KRQLQKIDKS EGRFHVQNLS QVEQDGRTGH GLRRSSKFCL KEHKALKTLG IIMGTFTLCW LPF FIVNIV HVIQDNLIRK EVYILLNWIG YVNSGFNPLI YCRSPDFRIA FQELLCLRRS SLKAYGNGYS SNGNTGEQSG LEVL FQGPY HVEQEKENKL LAEDLPGTED FVGHQGTVPS DNIDSQGRNA STNDSLLETS QVAPA UniProtKB: Beta-2 adrenergic receptor |
-Macromolecule #5: (5R,6R)-6-(methylamino)-5,6,7,8-tetrahydronaphthalene-1,2,5-triol
| Macromolecule | Name: (5R,6R)-6-(methylamino)-5,6,7,8-tetrahydronaphthalene-1,2,5-triol type: ligand / ID: 5 / Number of copies: 1 / Formula: G1I |
|---|---|
| Molecular weight | Theoretical: 209.242 Da |
| Chemical component information |  ChemComp-G1I: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Grid | Model: Quantifoil R1.2/1.3 / Support film - Material: CARBON / Support film - topology: HOLEY |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 277.15 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: COUNTING / Average electron dose: 52.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.5 µm / Nominal defocus min: 1.2 µm |
| Sample stage | Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller























































































































































































 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)