[English] 日本語
 Yorodumi
Yorodumi- EMDB-11271: E2 core of the fungal Pyruvate dehydrogenase complex with asymmet... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-11271 | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | E2 core of the fungal Pyruvate dehydrogenase complex with asymmetric interior PX261 component | ||||||||||||||||||
 Map data Map data | Fungal PDC (N. crassa). Recombinant preparation-tE2/PX261. Enforced symmetry: I2. Periphery (PX only) is absent. Core is structured. Interior is oversymm. | ||||||||||||||||||
 Sample Sample |
| ||||||||||||||||||
| Biological species |  Neurospora crassa (fungus) Neurospora crassa (fungus) | ||||||||||||||||||
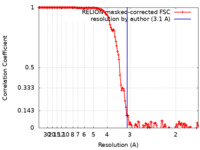
| Method | single particle reconstruction / cryo EM / Resolution: 3.1 Å | ||||||||||||||||||
 Authors Authors | Forsberg BO / Lindahl E / Aibara S / Howard RJ / Mortezaei N | ||||||||||||||||||
| Funding support |  Sweden, European Union, 5 items Sweden, European Union, 5 items
| ||||||||||||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2020 Journal: Nat Commun / Year: 2020Title: Arrangement and symmetry of the fungal E3BP-containing core of the pyruvate dehydrogenase complex. Authors: B O Forsberg / S Aibara / R J Howard / N Mortezaei / E Lindahl /   Abstract: The pyruvate dehydrogenase complex (PDC) is a multienzyme complex central to aerobic respiration, connecting glycolysis to mitochondrial oxidation of pyruvate. Similar to the E3-binding protein (E3BP) ...The pyruvate dehydrogenase complex (PDC) is a multienzyme complex central to aerobic respiration, connecting glycolysis to mitochondrial oxidation of pyruvate. Similar to the E3-binding protein (E3BP) of mammalian PDC, PX selectively recruits E3 to the fungal PDC, but its divergent sequence suggests a distinct structural mechanism. Here, we report reconstructions of PDC from the filamentous fungus Neurospora crassa by cryo-electron microscopy, where we find protein X (PX) interior to the PDC core as opposed to substituting E2 core subunits as in mammals. Steric occlusion limits PX binding, resulting in predominantly tetrahedral symmetry, explaining previous observations in Saccharomyces cerevisiae. The PX-binding site is conserved in (and specific to) fungi, and complements possible C-terminal binding motifs in PX that are absent in mammalian E3BP. Consideration of multiple symmetries thus reveals a differential structural basis for E3BP-like function in fungal PDC. | ||||||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_11271.map.gz emd_11271.map.gz | 44 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-11271-v30.xml emd-11271-v30.xml emd-11271.xml emd-11271.xml | 12.7 KB 12.7 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_11271_fsc.xml emd_11271_fsc.xml | 13.6 KB | Display |  FSC data file FSC data file |
| Images |  emd_11271.png emd_11271.png | 86.4 KB | ||
| Others |  emd_11271_additional.map.gz emd_11271_additional.map.gz emd_11271_additional_1.map.gz emd_11271_additional_1.map.gz | 202.4 MB 202.4 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-11271 http://ftp.pdbj.org/pub/emdb/structures/EMD-11271 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-11271 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-11271 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_11271.map.gz / Format: CCP4 / Size: 216 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_11271.map.gz / Format: CCP4 / Size: 216 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Fungal PDC (N. crassa). Recombinant preparation-tE2/PX261. Enforced symmetry: I2. Periphery (PX only) is absent. Core is structured. Interior is oversymm. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.86 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Additional map: Fungal PDC (N. crassa). Recombinant preparation-tE2/PX261. Enforced symmetry:...
| File | emd_11271_additional.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Fungal PDC (N. crassa). Recombinant preparation-tE2/PX261. Enforced symmetry: I2. Periphery (PX only) is absent. Core is structured. Interior is oversymm. Unmasked | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Additional map: Fungal PDC (N. crassa). Recombinant preparation-tE2/PX261. Enforced symmetry:...
| File | emd_11271_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Fungal PDC (N. crassa). Recombinant preparation-tE2/PX261. Enforced symmetry: I2. Periphery (PX only) is absent. Core is structured. Interior is oversymm. Unmasked | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : E2 core of the fungal Pyruvate dehydrogenase complex with asymmet...
| Entire | Name: E2 core of the fungal Pyruvate dehydrogenase complex with asymmetric inetrior PX261 component |
|---|---|
| Components |
|
-Supramolecule #1: E2 core of the fungal Pyruvate dehydrogenase complex with asymmet...
| Supramolecule | Name: E2 core of the fungal Pyruvate dehydrogenase complex with asymmetric inetrior PX261 component type: complex / ID: 1 / Parent: 0 |
|---|---|
| Source (natural) | Organism:  Neurospora crassa (fungus) Neurospora crassa (fungus) |
| Recombinant expression | Organism:  |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 3 mg/mL |
|---|---|
| Buffer | pH: 7.5 |
| Grid | Model: Quantifoil R1.2/1.3 / Material: COPPER / Mesh: 300 / Support film - Material: CARBON / Support film - topology: HOLEY / Pretreatment - Type: GLOW DISCHARGE |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Average electron dose: 31.4 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller
















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)