+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-10101 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Apoferritin from mouse at 1.84 angstrom resolution | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | iron ion binding / ferroxidase / iron storage / cytoplasm / METAL BINDING PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationIron uptake and transport / Golgi Associated Vesicle Biogenesis / ferroxidase / autolysosome / negative regulation of ferroptosis / ferroxidase activity / negative regulation of fibroblast proliferation / endocytic vesicle lumen / Neutrophil degranulation / ferric iron binding ...Iron uptake and transport / Golgi Associated Vesicle Biogenesis / ferroxidase / autolysosome / negative regulation of ferroptosis / ferroxidase activity / negative regulation of fibroblast proliferation / endocytic vesicle lumen / Neutrophil degranulation / ferric iron binding / autophagosome / iron ion transport / ferrous iron binding / intracellular iron ion homeostasis / immune response / iron ion binding / negative regulation of cell population proliferation / mitochondrion / extracellular region / membrane / identical protein binding / cytoplasm / cytosol Similarity search - Function | |||||||||
| Biological species |  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 1.84 Å | |||||||||
 Authors Authors | Mills DJ / Pfeil-Gardiner O / Vonck J | |||||||||
 Citation Citation |  Journal: To Be Published Journal: To Be PublishedTitle: To be published later Authors: Vonck J / Mills DJ / Pfeil-Gardiner O | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_10101.map.gz emd_10101.map.gz | 96.6 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-10101-v30.xml emd-10101-v30.xml emd-10101.xml emd-10101.xml | 16.6 KB 16.6 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_10101_fsc.xml emd_10101_fsc.xml | 10.6 KB | Display |  FSC data file FSC data file |
| Images |  emd_10101.png emd_10101.png | 301.3 KB | ||
| Filedesc metadata |  emd-10101.cif.gz emd-10101.cif.gz | 5.7 KB | ||
| Others |  emd_10101_half_map_1.map.gz emd_10101_half_map_1.map.gz emd_10101_half_map_2.map.gz emd_10101_half_map_2.map.gz | 75.7 MB 75.7 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-10101 http://ftp.pdbj.org/pub/emdb/structures/EMD-10101 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-10101 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-10101 | HTTPS FTP |
-Related structure data
| Related structure data |  6s61MC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_10101.map.gz / Format: CCP4 / Size: 103 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_10101.map.gz / Format: CCP4 / Size: 103 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.656 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
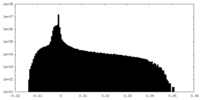
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Half map: mouse apoferritin, half map 1
| File | emd_10101_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | mouse apoferritin, half map 1 | ||||||||||||
| Projections & Slices |
| ||||||||||||
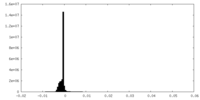
| Density Histograms |
-Half map: mouse apoferritin, half map 2
| File | emd_10101_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | mouse apoferritin, half map 2 | ||||||||||||
| Projections & Slices |
| ||||||||||||
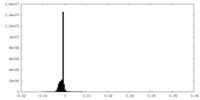
| Density Histograms |
- Sample components
Sample components
-Entire : Octahedral complex of 24 apoferritin chains
| Entire | Name: Octahedral complex of 24 apoferritin chains |
|---|---|
| Components |
|
-Supramolecule #1: Octahedral complex of 24 apoferritin chains
| Supramolecule | Name: Octahedral complex of 24 apoferritin chains / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 500 KDa |
-Macromolecule #1: Ferritin heavy chain
| Macromolecule | Name: Ferritin heavy chain / type: protein_or_peptide / ID: 1 / Number of copies: 24 / Enantiomer: LEVO / EC number: ferroxidase |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 21.097631 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MTTASPSQVR QNYHQDAEAA INRQINLELY ASYVYLSMSC YFDRDDVALK NFAKYFLHQS HEEREHAEKL MKLQNQRGGR IFLQDIKKP DRDDWESGLN AMECALHLEK SVNQSLLELH KLATDKNDPH LCDFIETYYL SEQVKSIKEL GDHVTNLRKM G APEAGMAE YLFDKHTLGH GDES UniProtKB: Ferritin heavy chain |
-Macromolecule #2: FE (III) ION
| Macromolecule | Name: FE (III) ION / type: ligand / ID: 2 / Number of copies: 6 / Formula: FE |
|---|---|
| Molecular weight | Theoretical: 55.845 Da |
-Macromolecule #3: ZINC ION
| Macromolecule | Name: ZINC ION / type: ligand / ID: 3 / Number of copies: 24 / Formula: ZN |
|---|---|
| Molecular weight | Theoretical: 65.409 Da |
-Macromolecule #4: water
| Macromolecule | Name: water / type: ligand / ID: 4 / Number of copies: 3854 / Formula: HOH |
|---|---|
| Molecular weight | Theoretical: 18.015 Da |
| Chemical component information |  ChemComp-HOH: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 1.5 mg/mL | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 7.5 / Component:
| |||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 283 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: FEI FALCON III (4k x 4k) / Detector mode: COUNTING / Number grids imaged: 1 / Number real images: 1788 / Average exposure time: 30.0 sec. / Average electron dose: 30.0 e/Å2 Details: Some images were discarding after motion correction (MotionCor2) and ctf estimation (CTFfind4), leaving 1745 for processing. |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Calibrated defocus max: 2.0 µm / Calibrated defocus min: 0.2 µm / Calibrated magnification: 182927 / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 1.5 µm / Nominal defocus min: 0.5 µm / Nominal magnification: 120000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller




































 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)