+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 |  | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| タイトル | XPA repositioning Core7 of TFIIH relative to XPC-DNA lesion (AP) | ||||||||||||
 マップデータ マップデータ | |||||||||||||
 試料 試料 |
| ||||||||||||
 キーワード キーワード | protein-DNA complex / DNA BINDING PROTEIN-DNA complex | ||||||||||||
| 機能・相同性 |  機能・相同性情報 機能・相同性情報nucleotide-excision repair factor 1 complex / nucleotide-excision repair involved in interstrand cross-link repair / XPC complex / nucleotide-excision repair, DNA damage recognition / 9+2 motile cilium / core TFIIH complex portion of holo TFIIH complex / MMXD complex / photoreceptor connecting cilium / heterotrimeric G-protein binding / Cytosolic iron-sulfur cluster assembly ...nucleotide-excision repair factor 1 complex / nucleotide-excision repair involved in interstrand cross-link repair / XPC complex / nucleotide-excision repair, DNA damage recognition / 9+2 motile cilium / core TFIIH complex portion of holo TFIIH complex / MMXD complex / photoreceptor connecting cilium / heterotrimeric G-protein binding / Cytosolic iron-sulfur cluster assembly / nucleotide-excision repair, DNA duplex unwinding / transcription export complex 2 / central nervous system myelin formation / response to auditory stimulus / positive regulation of mitotic recombination / hair follicle maturation / hair cell differentiation / nucleotide-excision repair factor 3 complex / nucleotide-excision repair, preincision complex assembly / nuclear pore nuclear basket / CAK-ERCC2 complex / UV protection / embryonic cleavage / G protein-coupled receptor internalization / UV-damage excision repair / DNA 3'-5' helicase / transcription factor TFIIH core complex / transcription factor TFIIH holo complex / transcription preinitiation complex / RNA Polymerase I Transcription Termination / nuclear thyroid hormone receptor binding / regulation of mitotic cell cycle phase transition / hematopoietic stem cell proliferation / intercellular bridge / regulation of cyclin-dependent protein serine/threonine kinase activity / spinal cord development / RNA Pol II CTD phosphorylation and interaction with CE during HIV infection / RNA Pol II CTD phosphorylation and interaction with CE / Formation of the Early Elongation Complex / Formation of the HIV-1 Early Elongation Complex / mRNA Capping / bone mineralization / HIV Transcription Initiation / RNA Polymerase II HIV Promoter Escape / Transcription of the HIV genome / RNA Polymerase II Promoter Escape / RNA Polymerase II Transcription Pre-Initiation And Promoter Opening / RNA Polymerase II Transcription Initiation / RNA Polymerase II Transcription Initiation And Promoter Clearance / erythrocyte maturation / 3'-5' DNA helicase activity / RNA Polymerase I Transcription Initiation / centriole replication / transcription by RNA polymerase I / DNA topological change / ATPase activator activity / transcription factor TFIID complex / intrinsic apoptotic signaling pathway by p53 class mediator / protein localization to nucleus / RNA polymerase II general transcription initiation factor activity / hematopoietic stem cell differentiation / transcription elongation by RNA polymerase I / Tat-mediated elongation of the HIV-1 transcript / mRNA transport / Formation of HIV-1 elongation complex containing HIV-1 Tat / embryonic organ development / transcription-coupled nucleotide-excision repair / Formation of HIV elongation complex in the absence of HIV Tat / SUMOylation of DNA damage response and repair proteins / RNA Polymerase II Transcription Elongation / Formation of RNA Pol II elongation complex / response to UV / DNA helicase activity / Loss of Nlp from mitotic centrosomes / Loss of proteins required for interphase microtubule organization from the centrosome / Recruitment of mitotic centrosome proteins and complexes / Recruitment of NuMA to mitotic centrosomes / RNA Polymerase II Pre-transcription Events / Anchoring of the basal body to the plasma membrane / centriole / hormone-mediated signaling pathway / AURKA Activation by TPX2 / extracellular matrix organization / maturation of SSU-rRNA from tricistronic rRNA transcript (SSU-rRNA, 5.8S rRNA, LSU-rRNA) / insulin-like growth factor receptor signaling pathway / ciliary basal body / post-embryonic development / regulation of cytokinesis / chromosome segregation / transcription elongation by RNA polymerase II / determination of adult lifespan / promoter-specific chromatin binding / nucleotide-excision repair / RNA Polymerase I Promoter Escape / TP53 Regulates Transcription of DNA Repair Genes / transcription initiation at RNA polymerase II promoter / DNA Damage Recognition in GG-NER / G-protein beta/gamma-subunit complex binding / NoRC negatively regulates rRNA expression / base-excision repair 類似検索 - 分子機能 | ||||||||||||
| 生物種 |  Homo sapiens (ヒト) / synthetic construct (人工物) Homo sapiens (ヒト) / synthetic construct (人工物) | ||||||||||||
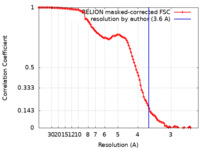
| 手法 | 単粒子再構成法 / クライオ電子顕微鏡法 / 解像度: 3.6 Å | ||||||||||||
 データ登録者 データ登録者 | Kim J / Yang W | ||||||||||||
| 資金援助 |  米国, 米国,  日本, 3件 日本, 3件
| ||||||||||||
 引用 引用 |  ジャーナル: Nature / 年: 2023 ジャーナル: Nature / 年: 2023タイトル: Lesion recognition by XPC, TFIIH and XPA in DNA excision repair. 著者: Jinseok Kim / Chia-Lung Li / Xuemin Chen / Yanxiang Cui / Filip M Golebiowski / Huaibin Wang / Fumio Hanaoka / Kaoru Sugasawa / Wei Yang /     要旨: Nucleotide excision repair removes DNA lesions caused by ultraviolet light, cisplatin-like compounds and bulky adducts. After initial recognition by XPC in global genome repair or a stalled RNA ...Nucleotide excision repair removes DNA lesions caused by ultraviolet light, cisplatin-like compounds and bulky adducts. After initial recognition by XPC in global genome repair or a stalled RNA polymerase in transcription-coupled repair, damaged DNA is transferred to the seven-subunit TFIIH core complex (Core7) for verification and dual incisions by the XPF and XPG nucleases. Structures capturing lesion recognition by the yeast XPC homologue Rad4 and TFIIH in transcription initiation or DNA repair have been separately reported. How two different lesion recognition pathways converge and how the XPB and XPD helicases of Core7 move the DNA lesion for verification are unclear. Here we report on structures revealing DNA lesion recognition by human XPC and DNA lesion hand-off from XPC to Core7 and XPA. XPA, which binds between XPB and XPD, kinks the DNA duplex and shifts XPC and the DNA lesion by nearly a helical turn relative to Core7. The DNA lesion is thus positioned outside of Core7, as would occur with RNA polymerase. XPB and XPD, which track the lesion-containing strand but translocate DNA in opposite directions, push and pull the lesion-containing strand into XPD for verification. | ||||||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| 添付画像 |
|---|
- ダウンロードとリンク
ダウンロードとリンク
-EMDBアーカイブ
| マップデータ |  emd_28001.map.gz emd_28001.map.gz | 95.1 MB |  EMDBマップデータ形式 EMDBマップデータ形式 | |
|---|---|---|---|---|
| ヘッダ (付随情報) |  emd-28001-v30.xml emd-28001-v30.xml emd-28001.xml emd-28001.xml | 36.7 KB 36.7 KB | 表示 表示 |  EMDBヘッダ EMDBヘッダ |
| FSC (解像度算出) |  emd_28001_fsc.xml emd_28001_fsc.xml | 10.7 KB | 表示 |  FSCデータファイル FSCデータファイル |
| 画像 |  emd_28001.png emd_28001.png | 80.7 KB | ||
| Filedesc metadata |  emd-28001.cif.gz emd-28001.cif.gz | 10.6 KB | ||
| その他 |  emd_28001_half_map_1.map.gz emd_28001_half_map_1.map.gz emd_28001_half_map_2.map.gz emd_28001_half_map_2.map.gz | 80.9 MB 81 MB | ||
| アーカイブディレクトリ |  http://ftp.pdbj.org/pub/emdb/structures/EMD-28001 http://ftp.pdbj.org/pub/emdb/structures/EMD-28001 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-28001 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-28001 | HTTPS FTP |
-検証レポート
| 文書・要旨 |  emd_28001_validation.pdf.gz emd_28001_validation.pdf.gz | 715 KB | 表示 |  EMDB検証レポート EMDB検証レポート |
|---|---|---|---|---|
| 文書・詳細版 |  emd_28001_full_validation.pdf.gz emd_28001_full_validation.pdf.gz | 714.5 KB | 表示 | |
| XML形式データ |  emd_28001_validation.xml.gz emd_28001_validation.xml.gz | 17.8 KB | 表示 | |
| CIF形式データ |  emd_28001_validation.cif.gz emd_28001_validation.cif.gz | 23.5 KB | 表示 | |
| アーカイブディレクトリ |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-28001 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-28001 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-28001 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-28001 | HTTPS FTP |
-関連構造データ
| 関連構造データ |  8ebxMC  8ebsC  8ebtC  8ebuC  8ebvC  8ebwC  8ebyC C: 同じ文献を引用 ( M: このマップから作成された原子モデル |
|---|---|
| 類似構造データ | 類似検索 - 機能・相同性  F&H 検索 F&H 検索 |
- リンク
リンク
| EMDBのページ |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| 「今月の分子」の関連する項目 |
- マップ
マップ
| ファイル |  ダウンロード / ファイル: emd_28001.map.gz / 形式: CCP4 / 大きさ: 103 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) ダウンロード / ファイル: emd_28001.map.gz / 形式: CCP4 / 大きさ: 103 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ボクセルのサイズ | X=Y=Z: 1.245 Å | ||||||||||||||||||||
| 密度 |
| ||||||||||||||||||||
| 対称性 | 空間群: 1 | ||||||||||||||||||||
| 詳細 | EMDB XML:
|
-添付データ
-ハーフマップ: #1
| ファイル | emd_28001_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
-ハーフマップ: #2
| ファイル | emd_28001_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
- 試料の構成要素
試料の構成要素
+全体 : small DNA lesion recognition complex3
+超分子 #1: small DNA lesion recognition complex3
+分子 #1: TFIIH basal transcription factor complex helicase XPB subunit
+分子 #2: General transcription and DNA repair factor IIH helicase subunit XPD
+分子 #3: General transcription factor IIH subunit 1
+分子 #4: General transcription factor IIH subunit 4, p52
+分子 #5: General transcription factor IIH subunit 2
+分子 #6: General transcription factor IIH subunit 3
+分子 #7: General transcription factor IIH subunit 5
+分子 #8: Xeroderma pigmentosum, complementation group C, isoform CRA_a
+分子 #9: Centrin-2
+分子 #10: DNA repair protein complementing XP-A cells
+分子 #11: DNA (Ap)
+分子 #12: DNA
+分子 #13: IRON/SULFUR CLUSTER
+分子 #14: ZINC ION
+分子 #15: CALCIUM ION
-実験情報
-構造解析
| 手法 | クライオ電子顕微鏡法 |
|---|---|
 解析 解析 | 単粒子再構成法 |
| 試料の集合状態 | particle |
- 試料調製
試料調製
| 濃度 | 0.4 mg/mL | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 緩衝液 | pH: 7.9 構成要素:
| ||||||||||||||||||
| グリッド | モデル: Quantifoil R1.2/1.3 / 材質: COPPER / メッシュ: 300 / 支持フィルム - 材質: CARBON / 支持フィルム - トポロジー: HOLEY / 前処理 - タイプ: GLOW DISCHARGE / 前処理 - 時間: 30 sec. | ||||||||||||||||||
| 凍結 | 凍結剤: ETHANE / チャンバー内湿度: 100 % / チャンバー内温度: 277.15 K / 装置: FEI VITROBOT MARK IV |
- 電子顕微鏡法
電子顕微鏡法
| 顕微鏡 | FEI TITAN KRIOS |
|---|---|
| 撮影 | フィルム・検出器のモデル: GATAN K3 (6k x 4k) / 撮影したグリッド数: 1 / 実像数: 3883 / 平均露光時間: 2.5 sec. / 平均電子線量: 54.1 e/Å2 |
| 電子線 | 加速電圧: 300 kV / 電子線源:  FIELD EMISSION GUN FIELD EMISSION GUN |
| 電子光学系 | C2レンズ絞り径: 70.0 µm / 照射モード: FLOOD BEAM / 撮影モード: BRIGHT FIELD / Cs: 2.7 mm / 最大 デフォーカス(公称値): 3.0 µm / 最小 デフォーカス(公称値): 2.0 µm / 倍率(公称値): 105000 |
| 試料ステージ | 試料ホルダーモデル: FEI TITAN KRIOS AUTOGRID HOLDER ホルダー冷却材: NITROGEN |
| 実験機器 |  モデル: Titan Krios / 画像提供: FEI Company |
 ムービー
ムービー コントローラー
コントローラー






























 Z
Z Y
Y X
X