[English] 日本語
 Yorodumi
Yorodumi- EMDB-17399: Homomeric GluA2 flip R/G-edited Q/R-edited F231A mutant in tandem... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
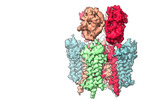
| Title | Homomeric GluA2 flip R/G-edited Q/R-edited F231A mutant in tandem with TARP gamma-2, desensitized conformation 3 | ||||||||||||
 Map data Map data | |||||||||||||
 Sample Sample |
| ||||||||||||
 Keywords Keywords | AMPA-type glutamate neurotransmitter receptor / auxiliary subunit complex / agonist / desensitized / MEMBRANE PROTEIN | ||||||||||||
| Function / homology |  Function and homology information Function and homology informationPresynaptic depolarization and calcium channel opening / eye blink reflex / positive regulation of protein localization to basolateral plasma membrane / cerebellar mossy fiber / LGI-ADAM interactions / Trafficking of AMPA receptors / postsynaptic neurotransmitter receptor diffusion trapping / regulation of AMPA receptor activity / channel regulator activity / membrane hyperpolarization ...Presynaptic depolarization and calcium channel opening / eye blink reflex / positive regulation of protein localization to basolateral plasma membrane / cerebellar mossy fiber / LGI-ADAM interactions / Trafficking of AMPA receptors / postsynaptic neurotransmitter receptor diffusion trapping / regulation of AMPA receptor activity / channel regulator activity / membrane hyperpolarization / nervous system process / protein targeting to membrane / voltage-gated calcium channel complex / spine synapse / dendritic spine neck / dendritic spine cytoplasm / dendritic spine head / cellular response to amine stimulus / Activation of AMPA receptors / neurotransmitter receptor localization to postsynaptic specialization membrane / ligand-gated monoatomic cation channel activity / perisynaptic space / neuromuscular junction development / Trafficking of GluR2-containing AMPA receptors / response to lithium ion / AMPA glutamate receptor activity / AMPA glutamate receptor clustering / transmission of nerve impulse / kainate selective glutamate receptor activity / immunoglobulin binding / AMPA glutamate receptor complex / regulation of receptor recycling / extracellularly glutamate-gated ion channel activity / cellular response to glycine / ionotropic glutamate receptor complex / asymmetric synapse / Unblocking of NMDA receptors, glutamate binding and activation / glutamate receptor binding / membrane depolarization / conditioned place preference / positive regulation of synaptic transmission / regulation of postsynaptic membrane neurotransmitter receptor levels / voltage-gated calcium channel activity / response to fungicide / regulation of synaptic transmission, glutamatergic / extracellular ligand-gated monoatomic ion channel activity / cytoskeletal protein binding / glutamate-gated receptor activity / cellular response to brain-derived neurotrophic factor stimulus / regulation of long-term synaptic depression / positive regulation of synaptic transmission, glutamatergic / somatodendritic compartment / glutamate-gated calcium ion channel activity / presynaptic active zone membrane / excitatory synapse / ionotropic glutamate receptor signaling pathway / ionotropic glutamate receptor binding / dendrite membrane / dendrite cytoplasm / ligand-gated monoatomic ion channel activity involved in regulation of presynaptic membrane potential / positive regulation of excitatory postsynaptic potential / hippocampal mossy fiber to CA3 synapse / dendritic shaft / SNARE binding / synaptic membrane / PDZ domain binding / calcium channel regulator activity / establishment of protein localization / protein tetramerization / transmitter-gated monoatomic ion channel activity involved in regulation of postsynaptic membrane potential / synaptic transmission, glutamatergic / regulation of membrane potential / cerebral cortex development / response to calcium ion / receptor internalization / postsynaptic density membrane / modulation of chemical synaptic transmission / Schaffer collateral - CA1 synapse / terminal bouton / synaptic vesicle / long-term synaptic potentiation / amyloid-beta binding / synaptic vesicle membrane / growth cone / presynapse / signaling receptor activity / scaffold protein binding / presynaptic membrane / dendritic spine / chemical synaptic transmission / perikaryon / postsynaptic membrane / neuron projection / postsynaptic density / external side of plasma membrane / axon / neuronal cell body / synapse / dendrite / protein kinase binding Similarity search - Function | ||||||||||||
| Biological species |  | ||||||||||||
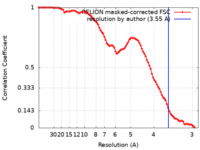
| Method | single particle reconstruction / cryo EM / Resolution: 3.55 Å | ||||||||||||
 Authors Authors | Krieger JM / Zhang D / Yamashita K / Greger IH | ||||||||||||
| Funding support |  United Kingdom, European Union, 3 items United Kingdom, European Union, 3 items
| ||||||||||||
 Citation Citation |  Journal: Nature / Year: 2023 Journal: Nature / Year: 2023Title: Structural mobility tunes signalling of the GluA1 AMPA glutamate receptor. Authors: Danyang Zhang / Josip Ivica / James M Krieger / Hinze Ho / Keitaro Yamashita / Imogen Stockwell / Rozbeh Baradaran / Ondrej Cais / Ingo H Greger /   Abstract: AMPA glutamate receptors (AMPARs), the primary mediators of excitatory neurotransmission in the brain, are either GluA2 subunit-containing and thus Ca-impermeable, or GluA2-lacking and Ca-permeable. ...AMPA glutamate receptors (AMPARs), the primary mediators of excitatory neurotransmission in the brain, are either GluA2 subunit-containing and thus Ca-impermeable, or GluA2-lacking and Ca-permeable. Despite their prominent expression throughout interneurons and glia, their role in long-term potentiation and their involvement in a range of neuropathologies, structural information for GluA2-lacking receptors is currently absent. Here we determine and characterize cryo-electron microscopy structures of the GluA1 homotetramer, fully occupied with TARPγ3 auxiliary subunits (GluA1/γ3). The gating core of both resting and open-state GluA1/γ3 closely resembles GluA2-containing receptors. However, the sequence-diverse N-terminal domains (NTDs) give rise to a highly mobile assembly, enabling domain swapping and subunit re-alignments in the ligand-binding domain tier that are pronounced in desensitized states. These transitions underlie the unique kinetic properties of GluA1. A GluA2 mutant (F231A) increasing NTD dynamics phenocopies this behaviour, and exhibits reduced synaptic responses, reflecting the anchoring function of the AMPAR NTD at the synapse. Together, this work underscores how the subunit-diverse NTDs determine subunit arrangement, gating properties and ultimately synaptic signalling efficiency among AMPAR subtypes. | ||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_17399.map.gz emd_17399.map.gz | 32.5 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-17399-v30.xml emd-17399-v30.xml emd-17399.xml emd-17399.xml | 16.2 KB 16.2 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_17399_fsc.xml emd_17399_fsc.xml | 9.1 KB | Display |  FSC data file FSC data file |
| Images |  emd_17399.png emd_17399.png | 70.6 KB | ||
| Filedesc metadata |  emd-17399.cif.gz emd-17399.cif.gz | 6.2 KB | ||
| Others |  emd_17399_half_map_1.map.gz emd_17399_half_map_1.map.gz emd_17399_half_map_2.map.gz emd_17399_half_map_2.map.gz | 59.4 MB 59.4 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-17399 http://ftp.pdbj.org/pub/emdb/structures/EMD-17399 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-17399 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-17399 | HTTPS FTP |
-Related structure data
| Related structure data |  8p3yMC  8c1pC  8c1qC  8c1rC  8c1sC  8c2hC  8c2iC  8p3qC  8p3sC  8p3tC  8p3uC  8p3vC  8p3wC  8p3xC  8p3zC  8pivC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_17399.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_17399.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.4455 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #2
| File | emd_17399_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
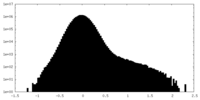
| Projections & Slices |
| ||||||||||||
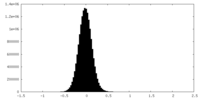
| Density Histograms |
-Half map: #1
| File | emd_17399_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
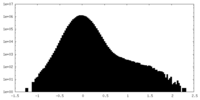
| Projections & Slices |
| ||||||||||||
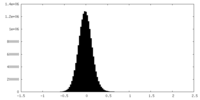
| Density Histograms |
- Sample components
Sample components
-Entire : GluA2igR_F231A-gamma2 tandem
| Entire | Name: GluA2igR_F231A-gamma2 tandem |
|---|---|
| Components |
|
-Supramolecule #1: GluA2igR_F231A-gamma2 tandem
| Supramolecule | Name: GluA2igR_F231A-gamma2 tandem / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all Details: GluA2 C-terminus fused to gamma-2 N-terminus with a linker |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 540 KDa |
-Macromolecule #1: Glutamate receptor 2
| Macromolecule | Name: Glutamate receptor 2 / type: protein_or_peptide / ID: 1 / Number of copies: 4 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 98.655672 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MQKIMHISVL LSPVLWGLIF GVSSNSIQIG GLFPRGADQE YSAFRVGMVQ FSTSEFRLTP HIDNLEVANS FAVTNAFCSQ FSRGVYAIF GFYDKKSVNT ITSFCGTLHV SFIGFPTDGT HPFVIQMRPD LKGALLSLIE YYQWDKFAYL YDSDRGLSTL Q AVLDSAAE ...String: MQKIMHISVL LSPVLWGLIF GVSSNSIQIG GLFPRGADQE YSAFRVGMVQ FSTSEFRLTP HIDNLEVANS FAVTNAFCSQ FSRGVYAIF GFYDKKSVNT ITSFCGTLHV SFIGFPTDGT HPFVIQMRPD LKGALLSLIE YYQWDKFAYL YDSDRGLSTL Q AVLDSAAE KKWQVTAINV GNINNDKKDE TYRSLFQDLE LKKERRVILD CERDKVNDIV DQVITIGKHV KGYHYIIANL GF TDGDLLK IQFGGANVSG FQIVDYDDSL VSKFIERWST LEEKEYPGAH TATIKYTSAL TYDAVQVMTE AFRNLRKQRI EIS RRGNAG DCLANPAVPW GQGVEIERAL KQVQVEGLSG NIKFDQNGKR INYTINIMEL KTNGPRKIGY WSEVDKMVVT LTEL PSGND TSGLENKTVV VTTILESPYV MMKKNHEMLE GNERYEGYCV DLAAEIAKHC GFKYKLTIVG DGKYGARDAD TKIWN GMVG ELVYGKADIA IAPLTITLVR EEVIDFSKPF MSLGISIMIK KPQKSKPGVF SFLDPLAYEI WMCIVFAYIG VSVVLF LVS RFSPYEWHTE EFEDGRETQS SESTNEFGIF NSLWFSLGAF MRQGCDISPR SLSGRIVGGV WWFFTLIIIS SYTANLA AF LTVERMVSPI ESAEDLSKQT EIAYGTLDSG STKEFFRRSK IAVFDKMWTY MRSAEPSVFV RTTAEGVARV RKSKGKYA Y LLESTMNEYI EQRKPCDTMK VGGNLDSKGY GIATPKGSSL GTPVNLAVLK LSEQGVLDKL KNKWWYDKGE CGAKDSGSK EKTSALSLSN VAGVFYILVG GLGLAMLVAL IEFCYKSRAE AKRMKVAKNP QNINPSSSQN SQNFATYKEG YNVYGIESVK I UniProtKB: Glutamate receptor 2 |
-Macromolecule #2: Voltage-dependent calcium channel gamma-2 subunit
| Macromolecule | Name: Voltage-dependent calcium channel gamma-2 subunit / type: protein_or_peptide / ID: 2 / Number of copies: 4 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 35.938746 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MGLFDRGVQM LLTTVGAFAA FSLMTIAVGT DYWLYSRGVC KTKSVSENET SKKNEEVMTH SGLWRTCCLE GNFKGLCKQI DHFPEDADY EADTAEYFLR AVRASSIFPI LSVILLFMGG LCIAASEFYK TRHNIILSAG IFFVSAGLSN IIGIIVYISA N AGDPSKSD ...String: MGLFDRGVQM LLTTVGAFAA FSLMTIAVGT DYWLYSRGVC KTKSVSENET SKKNEEVMTH SGLWRTCCLE GNFKGLCKQI DHFPEDADY EADTAEYFLR AVRASSIFPI LSVILLFMGG LCIAASEFYK TRHNIILSAG IFFVSAGLSN IIGIIVYISA N AGDPSKSD SKKNSYSYGW SFYFGALSFI IAEMVGVLAV HMFIDRHKQL RATARATDYL QASAITRIPS YRYRYQRRSR SS SRSTEPS HSRDASPVGV KGFNTLPSTE ISMYTLSRDP LKAATTPTAT YNSDRDNSFL QVHNCIQKDS KDSLHANTAN RRT TPV UniProtKB: Voltage-dependent calcium channel gamma-2 subunit |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 8 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | TFS KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Average electron dose: 50.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.4 µm / Nominal defocus min: 1.4000000000000001 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Refinement | Space: REAL / Protocol: RIGID BODY FIT |
|---|---|
| Output model |  PDB-8p3y: |
 Movie
Movie Controller
Controller























 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)