[English] 日本語
 Yorodumi
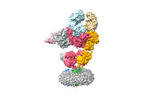
Yorodumi- PDB-7lep: The composite LBD-TMD structure combined from all hippocampal AMP... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 7lep | ||||||
|---|---|---|---|---|---|---|---|
| Title | The composite LBD-TMD structure combined from all hippocampal AMPAR subtypes at 3.25 Angstrom resolution | ||||||
 Components Components |
| ||||||
 Keywords Keywords | MEMBRANE PROTEIN / Native hippocampal ion channel | ||||||
| Function / homology |  Function and homology information Function and homology informationPhase 2 - plateau phase / Phase 0 - rapid depolarisation / negative regulation of receptor localization to synapse / negative regulation of anterograde synaptic vesicle transport / Cargo concentration in the ER / COPII-mediated vesicle transport / LGI-ADAM interactions / Trafficking of AMPA receptors / localization within membrane / neurotransmitter receptor transport, postsynaptic endosome to lysosome ...Phase 2 - plateau phase / Phase 0 - rapid depolarisation / negative regulation of receptor localization to synapse / negative regulation of anterograde synaptic vesicle transport / Cargo concentration in the ER / COPII-mediated vesicle transport / LGI-ADAM interactions / Trafficking of AMPA receptors / localization within membrane / neurotransmitter receptor transport, postsynaptic endosome to lysosome / L-type voltage-gated calcium channel complex / regulation of AMPA receptor activity / protein phosphatase 2B binding / neurotransmitter receptor internalization / postsynaptic neurotransmitter receptor diffusion trapping / transmission of nerve impulse / channel regulator activity / regulation of postsynaptic membrane neurotransmitter receptor levels / AMPA glutamate receptor complex / calcium channel regulator activity / voltage-gated calcium channel activity / positive regulation of synaptic transmission, glutamatergic / glutamate-gated receptor activity / vesicle-mediated transport / dendrite membrane / ionotropic glutamate receptor binding / regulation of membrane potential / dendritic shaft / synaptic transmission, glutamatergic / postsynaptic density membrane / Schaffer collateral - CA1 synapse / postsynaptic membrane / membrane => GO:0016020 / dendritic spine / postsynaptic density / glutamatergic synapse / synapse / dendrite / endoplasmic reticulum membrane Similarity search - Function | ||||||
| Biological species |  | ||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 3.25 Å | ||||||
 Authors Authors | Yu, J. / Rao, P. / Gouaux, E. | ||||||
| Funding support |  United States, 1items United States, 1items
| ||||||
 Citation Citation |  Journal: Nature / Year: 2021 Journal: Nature / Year: 2021Title: Hippocampal AMPA receptor assemblies and mechanism of allosteric inhibition. Authors: Jie Yu / Prashant Rao / Sarah Clark / Jaba Mitra / Taekjip Ha / Eric Gouaux /  Abstract: AMPA-selective glutamate receptors mediate the transduction of signals between the neuronal circuits of the hippocampus. The trafficking, localization, kinetics and pharmacology of AMPA receptors are ...AMPA-selective glutamate receptors mediate the transduction of signals between the neuronal circuits of the hippocampus. The trafficking, localization, kinetics and pharmacology of AMPA receptors are tuned by an ensemble of auxiliary protein subunits, which are integral membrane proteins that associate with the receptor to yield bona fide receptor signalling complexes. Thus far, extensive studies of recombinant AMPA receptor-auxiliary subunit complexes using engineered protein constructs have not been able to faithfully elucidate the molecular architecture of hippocampal AMPA receptor complexes. Here we obtain mouse hippocampal, calcium-impermeable AMPA receptor complexes using immunoaffinity purification and use single-molecule fluorescence and cryo-electron microscopy experiments to elucidate three major AMPA receptor-auxiliary subunit complexes. The GluA1-GluA2, GluA1-GluA2-GluA3 and GluA2-GluA3 receptors are the predominant assemblies, with the auxiliary subunits TARP-γ8 and CNIH2-SynDIG4 non-stochastically positioned at the B'/D' and A'/C' positions, respectively. We further demonstrate how the receptor-TARP-γ8 stoichiometry explains the mechanism of and submaximal inhibition by a clinically relevant, brain-region-specific allosteric inhibitor. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  7lep.cif.gz 7lep.cif.gz | 391.1 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb7lep.ent.gz pdb7lep.ent.gz | 310.8 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  7lep.json.gz 7lep.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Summary document |  7lep_validation.pdf.gz 7lep_validation.pdf.gz | 1.6 MB | Display |  wwPDB validaton report wwPDB validaton report |
|---|---|---|---|---|
| Full document |  7lep_full_validation.pdf.gz 7lep_full_validation.pdf.gz | 1.6 MB | Display | |
| Data in XML |  7lep_validation.xml.gz 7lep_validation.xml.gz | 70.6 KB | Display | |
| Data in CIF |  7lep_validation.cif.gz 7lep_validation.cif.gz | 101 KB | Display | |
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/le/7lep https://data.pdbj.org/pub/pdb/validation_reports/le/7lep ftp://data.pdbj.org/pub/pdb/validation_reports/le/7lep ftp://data.pdbj.org/pub/pdb/validation_reports/le/7lep | HTTPS FTP |
-Related structure data
| Related structure data |  23292MC  7lddC  7ldeC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
-Mix of AMPAR subunits (GluA1, GluA3, and ... , 2 types, 2 molecules AC
| #1: Protein | Mass: 43650.676 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
|---|---|
| #3: Protein | Mass: 43702.637 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
-Protein , 3 types, 6 molecules BDEFGH
| #2: Protein | Mass: 47252.312 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  #4: Protein | Mass: 18755.152 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  #5: Protein | Mass: 23242.705 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  |
|---|
-Non-polymers , 7 types, 42 molecules 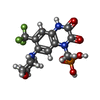



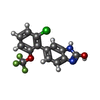
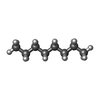







| #6: Chemical | ChemComp-ZK1 / {[ #7: Chemical | ChemComp-POV / ( #8: Chemical | ChemComp-C14 / | #9: Chemical | ChemComp-D10 / | #10: Chemical | #11: Chemical | #12: Chemical | |
|---|
-Details
| Has ligand of interest | Y |
|---|
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: Composite LBD-TMD complex from the combined hippocampal AMPAR subtypes Type: COMPLEX / Details: Bound with MPQX and JNJ55511118 / Entity ID: #1-#5 / Source: NATURAL |
|---|---|
| Molecular weight | Experimental value: NO |
| Source (natural) | Organism:  |
| Buffer solution | pH: 8 |
| Specimen | Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES |
| Vitrification | Instrument: FEI VITROBOT MARK IV / Cryogen name: ETHANE |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD |
| Image recording | Electron dose: 50 e/Å2 / Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) |
- Processing
Processing
| EM software | Name: cryoSPARC / Category: CTF correction |
|---|---|
| CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION |
| 3D reconstruction | Resolution: 3.25 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 116710 / Symmetry type: POINT |
 Movie
Movie Controller
Controller
















 PDBj
PDBj










