[English] 日本語
 Yorodumi
Yorodumi- EMDB-4721: Structure of a truncated adenylyl cyclase bound to MANT-GTP, fors... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-4721 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structure of a truncated adenylyl cyclase bound to MANT-GTP, forskolin and an activatedstimulatory Galphas protein | |||||||||
 Map data Map data | Truncated AC9 (AC9-1250) bound to MANT-GTP, forskolin and GalphaS | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Adenylyl cyclase / G protein / MANT-GTP / forskolin / MEMBRANE PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationAdenylate cyclase activating pathway / Adenylate cyclase inhibitory pathway / PKA activation / adenylate cyclase / Hedgehog 'off' state / sensory perception of chemical stimulus / mu-type opioid receptor binding / corticotropin-releasing hormone receptor 1 binding / cAMP biosynthetic process / adenylate cyclase activity ...Adenylate cyclase activating pathway / Adenylate cyclase inhibitory pathway / PKA activation / adenylate cyclase / Hedgehog 'off' state / sensory perception of chemical stimulus / mu-type opioid receptor binding / corticotropin-releasing hormone receptor 1 binding / cAMP biosynthetic process / adenylate cyclase activity / beta-2 adrenergic receptor binding / G alpha (z) signalling events / D1 dopamine receptor binding / adenylate cyclase-activating adrenergic receptor signaling pathway / insulin-like growth factor receptor binding / ionotropic glutamate receptor binding / adenylate cyclase activator activity / G-protein beta/gamma-subunit complex binding / adenylate cyclase-activating dopamine receptor signaling pathway / heterotrimeric G-protein complex / adenylate cyclase-activating G protein-coupled receptor signaling pathway / in utero embryonic development / Hydrolases; Acting on acid anhydrides; Acting on GTP to facilitate cellular and subcellular movement / intracellular signal transduction / ciliary basal body / GTPase activity / GTP binding / ATP binding / metal ion binding / plasma membrane / cytoplasm / cytosol Similarity search - Function | |||||||||
| Biological species |  | |||||||||
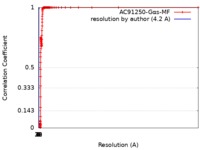
| Method | single particle reconstruction / cryo EM / Resolution: 4.2 Å | |||||||||
 Authors Authors | Qi C / Sorrentino S | |||||||||
| Funding support |  Switzerland, 1 items Switzerland, 1 items
| |||||||||
 Citation Citation |  Journal: Science / Year: 2019 Journal: Science / Year: 2019Title: The structure of a membrane adenylyl cyclase bound to an activated stimulatory G protein. Authors: Chao Qi / Simona Sorrentino / Ohad Medalia / Volodymyr M Korkhov /   Abstract: Membrane-integral adenylyl cyclases (ACs) are key enzymes in mammalian heterotrimeric GTP-binding protein (G protein)-dependent signal transduction, which is important in many cellular processes. ...Membrane-integral adenylyl cyclases (ACs) are key enzymes in mammalian heterotrimeric GTP-binding protein (G protein)-dependent signal transduction, which is important in many cellular processes. Signals received by the G protein-coupled receptors are conveyed to ACs through G proteins to modulate the levels of cellular cyclic adenosine monophosphate (cAMP). Here, we describe the cryo-electron microscopy structure of the bovine membrane AC9 bound to an activated G protein αs subunit at 3.4-angstrom resolution. The structure reveals the organization of the membrane domain and helical domain that spans between the membrane and catalytic domains of AC9. The carboxyl-terminal extension of the catalytic domain occludes both the catalytic and the allosteric sites of AC9, inducing a conformation distinct from the substrate- and activator-bound state, suggesting a regulatory role in cAMP production. #1:  Journal: To Be Published Journal: To Be PublishedTitle: The structure of a membrane adenylyl cyclase bound to an activated stimulatory G protein Authors: Qi C / Sorrentino S / Medalia O / Korkhov VM | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_4721.map.gz emd_4721.map.gz | 226.7 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-4721-v30.xml emd-4721-v30.xml emd-4721.xml emd-4721.xml | 18.1 KB 18.1 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_4721_fsc.xml emd_4721_fsc.xml | 6.8 KB | Display |  FSC data file FSC data file |
| Images |  emd_4721.png emd_4721.png | 67.4 KB | ||
| Filedesc metadata |  emd-4721.cif.gz emd-4721.cif.gz | 7.4 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-4721 http://ftp.pdbj.org/pub/emdb/structures/EMD-4721 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-4721 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-4721 | HTTPS FTP |
-Related structure data
| Related structure data |  6r4oMC  4719C  4722C  4723C  4724C  4725C  4726C  6r3qC  6r4pC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_4721.map.gz / Format: CCP4 / Size: 244.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_4721.map.gz / Format: CCP4 / Size: 244.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Truncated AC9 (AC9-1250) bound to MANT-GTP, forskolin and GalphaS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.8544 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
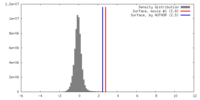
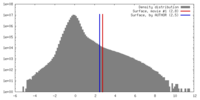
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
+Entire : Adenylyl cyclase AC9 (truncation, residues 1-1250) bound to MANT-...
+Supramolecule #1: Adenylyl cyclase AC9 (truncation, residues 1-1250) bound to MANT-...
+Supramolecule #2: Adenylyl cyclase AC9
+Supramolecule #3: GalphaS protein
+Macromolecule #1: Adenylate cyclase 9
+Macromolecule #2: Guanine nucleotide-binding protein G(s) subunit alpha isoforms short
+Macromolecule #3: 3'-O-(N-METHYLANTHRANILOYL)-GUANOSINE-5'-TRIPHOSPHATE
+Macromolecule #4: FORSKOLIN
+Macromolecule #5: MANGANESE (II) ION
+Macromolecule #6: 5'-GUANOSINE-DIPHOSPHATE-MONOTHIOPHOSPHATE
+Macromolecule #7: MAGNESIUM ION
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 8 |
|---|---|
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 278 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 QUANTUM (4k x 4k) / Detector mode: COUNTING / Number grids imaged: 2 / Number real images: 10758 / Average electron dose: 60.0 e/Å2 Details: Two datasets were merged using two different microscopes, using pixel size of 0.831 A/pix (4453 micrographs; 60 e-/A2) and 0.8544 A/pix (6305 micrographs; 45 e-/A2). Final dataset was scaled to 0.8544 A/pix. |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Refinement | Space: REAL / Protocol: AB INITIO MODEL / Overall B value: 110.92 / Target criteria: Cross-correlation coefficient |
|---|---|
| Output model |  PDB-6r4o: |
 Movie
Movie Controller
Controller

















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)



















 Homo sapiens (human)
Homo sapiens (human) Trichoplusia ni (cabbage looper)
Trichoplusia ni (cabbage looper)