[English] 日本語
 Yorodumi
Yorodumi- EMDB-3658: cryo-EM structure of the mTMEM16A ion channel at 6.6 A resolution. -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-3658 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | cryo-EM structure of the mTMEM16A ion channel at 6.6 A resolution. | |||||||||
 Map data Map data | None | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | TMEM16 family / ion channel / membrane protein / cryo-EM | |||||||||
| Function / homology |  Function and homology information Function and homology informationglial cell projection elongation / trachea development / iodide transmembrane transporter activity / mucus secretion / iodide transport / intracellularly calcium-gated chloride channel activity / cellular response to peptide / Stimuli-sensing channels / voltage-gated chloride channel activity / calcium-activated cation channel activity ...glial cell projection elongation / trachea development / iodide transmembrane transporter activity / mucus secretion / iodide transport / intracellularly calcium-gated chloride channel activity / cellular response to peptide / Stimuli-sensing channels / voltage-gated chloride channel activity / calcium-activated cation channel activity / protein localization to membrane / chloride transport / detection of temperature stimulus involved in sensory perception of pain / chloride channel activity / chloride channel complex / chloride transmembrane transport / regulation of membrane potential / cellular response to heat / presynaptic membrane / phospholipase C-activating G protein-coupled receptor signaling pathway / apical plasma membrane / signaling receptor binding / external side of plasma membrane / glutamatergic synapse / protein homodimerization activity / nucleoplasm / metal ion binding / identical protein binding / plasma membrane Similarity search - Function | |||||||||
| Biological species |  | |||||||||
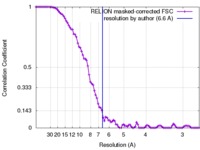
| Method | single particle reconstruction / cryo EM / Resolution: 6.6 Å | |||||||||
 Authors Authors | Paulino C / Neldner Y | |||||||||
| Funding support |  Switzerland, 2 items Switzerland, 2 items
| |||||||||
 Citation Citation |  Journal: Elife / Year: 2017 Journal: Elife / Year: 2017Title: Structural basis for anion conduction in the calcium-activated chloride channel TMEM16A. Authors: Cristina Paulino / Yvonne Neldner / Andy Km Lam / Valeria Kalienkova / Janine Denise Brunner / Stephan Schenck / Raimund Dutzler /  Abstract: The calcium-activated chloride channel TMEM16A is a member of a conserved protein family that comprises ion channels and lipid scramblases. Although the structure of the scramblase nhTMEM16 has ...The calcium-activated chloride channel TMEM16A is a member of a conserved protein family that comprises ion channels and lipid scramblases. Although the structure of the scramblase nhTMEM16 has defined the architecture of the family, it was unknown how a channel has adapted to cope with its distinct functional properties. Here we have addressed this question by the structure determination of mouse TMEM16A by cryo-electron microscopy and a complementary functional characterization. The protein shows a similar organization to nhTMEM16, except for changes at the site of catalysis. There, the conformation of transmembrane helices constituting a membrane-spanning furrow that provides a path for lipids in scramblases has changed to form an enclosed aqueous pore that is largely shielded from the membrane. Our study thus reveals the structural basis of anion conduction in a TMEM16 channel and it defines the foundation for the diverse functional behavior in the TMEM16 family. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_3658.map.gz emd_3658.map.gz | 57.3 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-3658-v30.xml emd-3658-v30.xml emd-3658.xml emd-3658.xml | 18.6 KB 18.6 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_3658_fsc.xml emd_3658_fsc.xml | 8.9 KB | Display |  FSC data file FSC data file |
| Images |  emd_3658.png emd_3658.png | 62.8 KB | ||
| Filedesc metadata |  emd-3658.cif.gz emd-3658.cif.gz | 7.2 KB | ||
| Others |  emd_3658_additional.map.gz emd_3658_additional.map.gz | 59.4 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-3658 http://ftp.pdbj.org/pub/emdb/structures/EMD-3658 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-3658 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-3658 | HTTPS FTP |
-Related structure data
| Related structure data |  5nl2MC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_3658.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_3658.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | None | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.35 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Additional map: None
| File | emd_3658_additional.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | None | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : mTMEM16A
| Entire | Name: mTMEM16A |
|---|---|
| Components |
|
-Supramolecule #1: mTMEM16A
| Supramolecule | Name: mTMEM16A / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 110.916 KDa |
-Macromolecule #1: Anoctamin-1
| Macromolecule | Name: Anoctamin-1 / type: protein_or_peptide / ID: 1 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 111.058992 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MRVPEKYSTL PAEDRSVHIV NICAIEDLGY LPSEGTLLNS LSVDPDAECK YGLYFRDGKR KVDYILVYHH KRASGSRTLA RRGLQNDMV LGTRSVRQDQ PLPGKGSPVD AGSPEVPMDY HEDDKRFRRE EYEGNLLEAG LELENDEDTK IHGVGFVKIH A PWHVLCRE ...String: MRVPEKYSTL PAEDRSVHIV NICAIEDLGY LPSEGTLLNS LSVDPDAECK YGLYFRDGKR KVDYILVYHH KRASGSRTLA RRGLQNDMV LGTRSVRQDQ PLPGKGSPVD AGSPEVPMDY HEDDKRFRRE EYEGNLLEAG LELENDEDTK IHGVGFVKIH A PWHVLCRE AEFLKLKMPT KKVYHISETR GLLKTINSVL QKITDPIQPK VAEHRPQTTK RLSYPFSREK QHLFDLTDRD SF FDSKTRS TIVYEILKRT TCTKAKYSMG ITSLLANGVY SAAYPLHDGD YEGDNVEFND RKLLYEEWAS YGVFYKYQPI DLV RKYFGE KVGLYFAWLG AYTQMLIPAS IVGVIVFLYG CATVDENIPS MEMCDQRYNI TMCPLCDKTC SYWKMSSACA TARA SHLFD NPATVFFSVF MALWAATFME HWKRKQMRLN YRWDLTGFEE EEEAVKDHPR AEYEARVLEK SLRKESRNKE TDKVK LTWR DRFPAYFTNL VSIIFMIAVT FAIVLGVIIY RISTAAALAM NSSPSVRSNI RVTVTATAVI INLVVIILLD EVYGCI ARW LTKIEVPKTE KSFEERLTFK AFLLKFVNSY TPIFYVAFFK GRFVGRPGDY VYIFRSFRME ECAPGGCLME LCIQLSI IM LGKQLIQNNL FEIGIPKMKK FIRYLKLRRQ SPSDREEYVK RKQRYEVDFN LEPFAGLTPE YMEMIIQFGF VTLFVASF P LAPLFALLNN IIEIRLDAKK FVTELRRPVA IRAKDIGIWY NILRGVGKLA VIINAFVISF TSDFIPRLVY LYMYSQNGT MHGFVNHTLS SFNVSDFQNG TAPNDPLDLG YEVQICRYKD YREPPWSEHK YDISKDFWAV LAARLAFVIV FQNLVMFMSD FVDWVIPDI PKDISQQIHK EKVLMVELFM REEQGKQQLL DTWMEKEKPR DVPCNNHSPT THPEAGDGSP VPSYEYHGDA L UniProtKB: Anoctamin-1 |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 3.08 mg/mL |
|---|---|
| Buffer | pH: 7.5 / Component - Concentration: 20.0 mM / Component - Name: Hepes / Details: 20 mM Hepes 150 mM NaCl 0.5 mM CaCl2 <0.12% digitonin |
| Grid | Model: Quantifoil R1.2/1.3 / Material: GOLD / Mesh: 200 / Support film - Material: CARBON / Support film - topology: HOLEY / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 30 sec. |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 293 K / Instrument: FEI VITROBOT MARK IV / Details: 2.5 ul sample volume 1-5 sec blotting time. |
| Details | full-length (wild-type isoform ac) deglycosylated mTMEM16A |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Temperature | Min: 80.0 K / Max: 100.0 K |
| Specialist optics | Energy filter - Name: GIF / Energy filter - Lower energy threshold: -10 eV / Energy filter - Upper energy threshold: +10 eV |
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: SUPER-RESOLUTION / Digitization - Dimensions - Width: 7420 pixel / Digitization - Dimensions - Height: 7676 pixel / Digitization - Frames/image: 1-50 / Number grids imaged: 5 / Number real images: 5503 / Average exposure time: 15.0 sec. / Average electron dose: 80.0 e/Å2 Details: Data were collected in an automated fashion on a K2 Summit detector (Gatan) set to super-resolution mode with a pixel size of 0.675 A and a defocus range of -0.5 to -3.8 um using SerialEM. ...Details: Data were collected in an automated fashion on a K2 Summit detector (Gatan) set to super-resolution mode with a pixel size of 0.675 A and a defocus range of -0.5 to -3.8 um using SerialEM. Images were recorded for 15 sec with an initial sub-frame exposure time of 300 ms (50 frames total) with a dose of 1.5 e-/A^2/frame, and later with a sub-frame exposure time of 150 ms (100 frames total) with a dose of 0.8 e-/A^2/frame, resulting in a total accumulated dose on the specimen level of approximately 80 e-/A^2. |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 100.0 µm / Calibrated defocus max: 3.8000000000000003 µm / Calibrated defocus min: 0.5 µm / Calibrated magnification: 37037 / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 3.8000000000000003 µm / Nominal defocus min: 0.5 µm / Nominal magnification: 37037 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller










 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)