登録情報 データベース : EMDB / ID : EMD-30260タイトル Cryo-EM structure of mature Coxsackievirus A10 in complex with KRM1 at pH 5.5 ウイルス : タンパク質・ペプチド : Capsid protein VP1タンパク質・ペプチド : Capsid protein VP2タンパク質・ペプチド : Capsid protein VP3タンパク質・ペプチド : Capsid protein VP4タンパク質・ペプチド : KRM1リガンド : 2-acetamido-2-deoxy-beta-D-glucopyranose / / / / / / 機能・相同性 分子機能 ドメイン・相同性 構成要素
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / 生物種 / Homo sapiens (ヒト)手法 / / 解像度 : 3.0 Å Cui Y / Peng R / Gao GF / Qi J 資金援助 Organization Grant number 国 Chinese Academy of Sciences XDB29010202
ジャーナル : Proc Natl Acad Sci U S A / 年 : 2020タイトル : Molecular basis of Coxsackievirus A10 entry using the two-in-one attachment and uncoating receptor KRM1.著者 : Yingzi Cui / Ruchao Peng / Hao Song / Zhou Tong / Xiao Qu / Sheng Liu / Xin Zhao / Yan Chai / Peiyi Wang / George F Gao / Jianxun Qi / 要旨 : KREMEN1 (KRM1) has been identified as a functional receptor for Coxsackievirus A10 (CV-A10), a causative agent of hand-foot-and-mouth disease (HFMD), which poses a great threat to infants globally. ... KREMEN1 (KRM1) has been identified as a functional receptor for Coxsackievirus A10 (CV-A10), a causative agent of hand-foot-and-mouth disease (HFMD), which poses a great threat to infants globally. However, the underlying mechanisms for the viral entry process are not well understood. Here we determined the atomic structures of different forms of CV-A10 viral particles and its complex with KRM1 in both neutral and acidic conditions. These structures reveal that KRM1 selectively binds to the mature viral particle above the canyon of the viral protein 1 (VP1) subunit and contacts across two adjacent asymmetry units. The key residues for receptor binding are conserved among most KRM1-dependent enteroviruses, suggesting a uniform mechanism for receptor binding. Moreover, the binding of KRM1 induces the release of pocket factor, a process accelerated under acidic conditions. Further biochemical studies confirmed that receptor binding at acidic pH enabled CV-A10 virion uncoating in vitro. Taken together, these findings provide high-resolution snapshots of CV-A10 entry and identify KRM1 as a two-in-one receptor for enterovirus infection. 履歴 登録 2020年4月28日 - ヘッダ(付随情報) 公開 2020年7月22日 - マップ公開 2020年7月22日 - 更新 2025年6月18日 - 現状 2025年6月18日 処理サイト : PDBj / 状態 : 公開
すべて表示 表示を減らす
 データを開く
データを開く 基本情報
基本情報 マップデータ
マップデータ 試料
試料 キーワード
キーワード 機能・相同性情報
機能・相同性情報
 Coxsackievirus A10 (コクサッキーウイルス) /
Coxsackievirus A10 (コクサッキーウイルス) /  Homo sapiens (ヒト)
Homo sapiens (ヒト) データ登録者
データ登録者 中国, 1件
中国, 1件  引用
引用 ジャーナル: Proc Natl Acad Sci U S A / 年: 2020
ジャーナル: Proc Natl Acad Sci U S A / 年: 2020
 構造の表示
構造の表示 ムービービューア
ムービービューア SurfView
SurfView Molmil
Molmil Jmol/JSmol
Jmol/JSmol ダウンロードとリンク
ダウンロードとリンク emd_30260.map.gz
emd_30260.map.gz EMDBマップデータ形式
EMDBマップデータ形式 emd-30260-v30.xml
emd-30260-v30.xml emd-30260.xml
emd-30260.xml EMDBヘッダ
EMDBヘッダ emd_30260_fsc.xml
emd_30260_fsc.xml FSCデータファイル
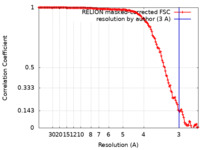
FSCデータファイル emd_30260.png
emd_30260.png emd-30260.cif.gz
emd-30260.cif.gz http://ftp.pdbj.org/pub/emdb/structures/EMD-30260
http://ftp.pdbj.org/pub/emdb/structures/EMD-30260 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-30260
ftp://ftp.pdbj.org/pub/emdb/structures/EMD-30260 リンク
リンク EMDB (EBI/PDBe) /
EMDB (EBI/PDBe) /  EMDataResource
EMDataResource マップ
マップ ダウンロード / ファイル: emd_30260.map.gz / 形式: CCP4 / 大きさ: 244.1 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES)
ダウンロード / ファイル: emd_30260.map.gz / 形式: CCP4 / 大きさ: 244.1 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) 試料の構成要素
試料の構成要素
 Coxsackievirus A10 (コクサッキーウイルス)
Coxsackievirus A10 (コクサッキーウイルス)
 Coxsackievirus A10 (コクサッキーウイルス)
Coxsackievirus A10 (コクサッキーウイルス)
 Coxsackievirus A10 (コクサッキーウイルス)
Coxsackievirus A10 (コクサッキーウイルス)
 Coxsackievirus A10 (コクサッキーウイルス)
Coxsackievirus A10 (コクサッキーウイルス)
 Coxsackievirus A10 (コクサッキーウイルス)
Coxsackievirus A10 (コクサッキーウイルス) Homo sapiens (ヒト)
Homo sapiens (ヒト) Homo sapiens (ヒト)
Homo sapiens (ヒト)
 解析
解析 試料調製
試料調製 電子顕微鏡法
電子顕微鏡法 FIELD EMISSION GUN
FIELD EMISSION GUN
 ムービー
ムービー コントローラー
コントローラー


































 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)