+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | DNA-PKcs monomer in dimer DNA-PKcs complex | |||||||||
 Map data Map data | DPKcs in dimer | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Dimer / Kinase / Complex / NHEJ / DNA BINDING PROTEIN | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
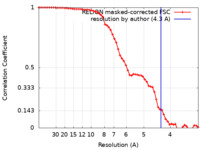
| Method | single particle reconstruction / cryo EM / Resolution: 4.3 Å | |||||||||
 Authors Authors | Chen S / He Y | |||||||||
| Funding support |  United States, 2 items United States, 2 items
| |||||||||
 Citation Citation |  Journal: Sci Adv / Year: 2023 Journal: Sci Adv / Year: 2023Title: Cryo-EM visualization of DNA-PKcs structural intermediates in NHEJ. Authors: Siyu Chen / Alex Vogt / Linda Lee / Tasmin Naila / Ryan McKeown / Alan E Tomkinson / Susan P Lees-Miller / Yuan He /   Abstract: DNA double-strand breaks (DSBs), one of the most cytotoxic forms of DNA damage, can be repaired by the tightly regulated nonhomologous end joining (NHEJ) machinery (Stinson and Loparo and Zhao ). ...DNA double-strand breaks (DSBs), one of the most cytotoxic forms of DNA damage, can be repaired by the tightly regulated nonhomologous end joining (NHEJ) machinery (Stinson and Loparo and Zhao ). Core NHEJ factors form an initial long-range (LR) synaptic complex that transitions into a DNA-PKcs (DNA-dependent protein kinase, catalytic subunit)-free, short-range state to align the DSB ends (Chen ). Using single-particle cryo-electron microscopy, we have visualized three additional key NHEJ complexes representing different transition states, with DNA-PKcs adopting distinct dimeric conformations within each of them. Upon DNA-PKcs autophosphorylation, the LR complex undergoes a substantial conformational change, with both Ku and DNA-PKcs rotating outward to promote DNA break exposure and DNA-PKcs dissociation. We also captured a dimeric state of catalytically inactive DNA-PKcs, which resembles structures of other PIKK (Phosphatidylinositol 3-kinase-related kinase) family kinases, revealing a model of the full regulatory cycle of DNA-PKcs during NHEJ. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_28734.map.gz emd_28734.map.gz | 16.1 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-28734-v30.xml emd-28734-v30.xml emd-28734.xml emd-28734.xml | 21.5 KB 21.5 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_28734_fsc.xml emd_28734_fsc.xml | 6 KB | Display |  FSC data file FSC data file |
| Images |  emd_28734.png emd_28734.png | 122.3 KB | ||
| Others |  emd_28734_half_map_1.map.gz emd_28734_half_map_1.map.gz emd_28734_half_map_2.map.gz emd_28734_half_map_2.map.gz | 13.8 MB 13.8 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-28734 http://ftp.pdbj.org/pub/emdb/structures/EMD-28734 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-28734 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-28734 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_28734.map.gz / Format: CCP4 / Size: 18.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_28734.map.gz / Format: CCP4 / Size: 18.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | DPKcs in dimer | ||||||||||||||||||||
| Voxel size | X=Y=Z: 1.6512 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: half1
| File | emd_28734_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | half1 | ||||||||||||
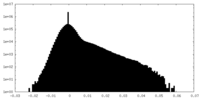
| Projections & Slices |
| ||||||||||||
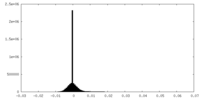
| Density Histograms |
-Half map: half2
| File | emd_28734_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | half2 | ||||||||||||
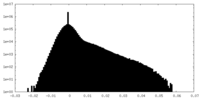
| Projections & Slices |
| ||||||||||||
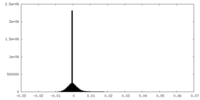
| Density Histograms |
- Sample components
Sample components
-Entire : dimer of DNA-PKcs
| Entire | Name: dimer of DNA-PKcs |
|---|---|
| Components |
|
-Supramolecule #1: dimer of DNA-PKcs
| Supramolecule | Name: dimer of DNA-PKcs / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 940 KDa |
-Macromolecule #1: PRKDC_HUMAN DNA-dependent protein kinase catalytic subunit
| Macromolecule | Name: PRKDC_HUMAN DNA-dependent protein kinase catalytic subunit type: protein_or_peptide / ID: 1 / Enantiomer: LEVO / EC number: non-specific serine/threonine protein kinase |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MAGSGAGVRC SLLRLQETLS AADRCGAALA GHQLIRGLGQ ECVLSSSPAV LALQTSLVFS RDFGLLVFV RKSLNSIEFR ECREEILKFL CIFLEKMGQK IAPYSVEIKN TCTSVYTKDR A AKCKIPAL DLLIKLLQTF RSSRLMDEFK IGELFSKFYG ELALKKKIPD ...String: MAGSGAGVRC SLLRLQETLS AADRCGAALA GHQLIRGLGQ ECVLSSSPAV LALQTSLVFS RDFGLLVFV RKSLNSIEFR ECREEILKFL CIFLEKMGQK IAPYSVEIKN TCTSVYTKDR A AKCKIPAL DLLIKLLQTF RSSRLMDEFK IGELFSKFYG ELALKKKIPD TVLEKVYELL GL LGEVHPS EMINNAENLF RAFLGELKTQ MTSAVREPKL PVLAGCLKGL SSLLCNFTKS MEE DPQTSR EIFNFVLKAI RPQIDLKRYA VPSAGLRLFA LHASQFSTCL LDNYVSLFEV LLKW CAHTN VELKKAALSA LESFLKQVSN MVAKNAEMHK NKLQYFMEQF YGIIRNVDSN NKELS IAIR GYGLFAGPCK VINAKDVDFM YVELIQRCKQ MFLTQTDTGD DRVYQMPSFL QSVASV LLY LDTVPEVYTP VLEHLVVMQI DSFPQYSPKM QLVCCRAIVK VFLALAAKGP VLRNCIS TV VHQGLIRICS KPVVLPKGPE SESEDHRASG EVRTGKWKVP TYKDYVDLFR HLLSSDQM M DSILADEAFF SVNSSSESLN HLLYDEFVKS VLKIVEKLDL TLEIQTVGEQ ENGDEAPGV WMIPTSDPAA NLHPAKPKDF SAFINLVEFC REILPEKQAE FFEPWVYSFS YELILQSTRL PLISGFYKL LSITVRNAKK IKYFEGVSPK SLKHSPEDPE KYSCFALFVK FGKEVAVKMK Q YKDELLAS CLTFLLSLPH NIIELDVRAY VPALQMAFKL GLSYTPLAEV GLNALEEWSI YI DRHVMQP YYKDILPCLD GYLKTSALSD ETKNNWEVSA LSRAAQKGFN KVVLKHLKKT KNL SSNEAI SLEEIRIRVV QMLGSLGGQI NKNLLTVTSS DEMMKSYVAW DREKRLSFAV PFRE MKPVI FLDVFLPRVT ELALTASDRQ TKVAACELLH SMVMFMLGKA TQMPEGGQGA PPMYQ LYKR TFPVLLRLAC DVDQVTRQLY EPLVMQLIHW FTNNKKFESQ DTVALLEAIL DGIVDP VDS TLRDFCGRCI REFLKWSIKQ ITPQQQEKSP VNTKSLFKRL YSLALHPNAF KRLGASL AF NNIYREFREE ESLVEQFVFE ALVIYMESLA LAHADEKSLG TIQQCCDAID HLCRIIEK K HVSLNKAKKR RLPRGFPPSA SLCLLDLVKW LLAHCGRPQT ECRHKSIELF YKFVPLLPG NRSPNLWLKD VLKEEGVSFL INTFEGGGCG QPSGILAQPT LLYLRGPFSL QATLCWLDLL LAALECYNT FIGERTVGAL QVLGTEAQSS LLKAVAFFLE SIAMHDIIAA EKCFGTGAAG N RTSPQEGE RYNYSKCTVV VRIMEFTTTL LNTSPEGWKL LKKDLCNTHL MRVLVQTLCE PA SIGFNIG DVQVMAHLPD VCVNLMKALK MSPYKDILET HLREKITAQS IEELCAVNLY GPD AQVDRS RLAAVVSACK QLHRAGLLHN ILPSQSTDLH HSVGTELLSL VYKGIAPGDE RQCL PSLDL SCKQLASGLL ELAFAFGGLC ERLVSLLLNP AVLSTASLGS SQGSVIHFSH GEYFY SLFS ETINTELLKN LDLAVLELMQ SSVDNTKMVS AVLNGMLDQS FRERANQKHQ GLKLAT TIL QHWKKCDSWW AKDSPLETKM AVLALLAKIL QIDSSVSFNT SHGSFPEVFT TYISLLA DT KLDLHLKGQA VTLLPFFTSL TGGSLEELRR VLEQLIVAHF PMQSREFPPG TPRFNNYV D CMKKFLDALE LSQSPMLLEL MTEVLCREQQ HVMEELFQSS FRRIARRGSC VTQVGLLES VYEMFRKDDP RLSFTRQSFV DRSLLTLLWH CSLDALREFF STIVVDAIDV LKSRFTKLNE STFDTQITK KMGYYKILDV MYSRLPKDDV HAKESKINQV FHGSCITEGN ELTKTLIKLC Y DAFTENMA GENQLLERRR LYHCAAYNCA ISVICCVFNE LKFYQGFLFS EKPEKNLLIF EN LIDLKRR YNFPVEVEVP MERKKKYIEI RKEAREAANG DSDGPSYMSS LSYLADSTLS EEM SQFDFS TGVQSYSYSS QDPRPATGRF RRREQRDPTV HDDVLELEMD ELNRHECMAP LTAL VKHMH RSLGPPQGEE DSVPRDLPSW MKFLHGKLGN PIVPLNIRLF LAKLVINTEE VFRPY AKHW LSPLLQLAAS ENNGGEGIHY MVVEIVATIL SWTGLATPTG VPKDEVLANR LLNFLM KHV FHPKRAVFRH NLEIIKTLVE CWKDCLSIPY RLIFEKFSGK DPNSKDNSVG IQLLGIV MA NDLPPYDPQC GIQSSEYFQA LVNNMSFVRY KEVYAAAAEV LGLILRYVME RKNILEES L CELVAKQLKQ HQNTMEDKFI VCLNKVTKSF PPLADRFMNA VFFLLPKFHG VLKTLCLEV VLCRVEGMTE LYFQLKSKDF VQVMRHRDDE RQKVCLDIIY KMMPKLKPVE LRELLNPVVE FVSHPSTTC REQMYNILMW IHDNYRDPES ETDNDSQEIF KLAKDVLIQG LIDENPGLQL I IRNFWSHE TRLPSNTLDR LLALNSLYSP KIEVHFLSLA TNFLLEMTSM SPDYPNPMFE HP LSECEFQ EYTIDSDWRF RSTVLTPMFV ETQASQGTLQ TRTQEGSLSA RWPVAGQIRA TQQ QHDFTL TQTADGRSSF DWLTGSSTDP LVDHTSPSSD SLLFAHKRSE RLQRAPLKSV GPDF GKKRL GLPGDEVDNK VKGAAGRTDL LRLRRRFMRD QEKLSLMYAR KGVAEQKREK EIKSE LKMK QDAQVVLYRS YRHGDLPDIQ IKHSSLITPL QAVAQRDPII AKQLFSSLFS GILKEM DKF KTLSEKNNIT QKLLQDFNRF LNTTFSFFPP FVSCIQDISC QHAALLSLDP AAVSAGC LA SLQQPVGIRL LEEALLRLLP AELPAKRVRG KARLPPDVLR WVELAKLYRS IGEYDVLR G IFTSEIGTKQ ITQSALLAEA RSDYSEAAKQ YDEALNKQDW VDGEPTEAEK DFWELASLD CYNHLAEWKS LEYCSTASID SENPPDLNKI WSEPFYQETY LPYMIRSKLK LLLQGEADQS LLTFIDKAM HGELQKAILE LHYSQELSLL YLLQDDVDRA KYYIQNGIQS FMQNYSSIDV L LHQSRLTK LQSVQALTEI QEFISFISKQ GNLSSQVPLK RLLNTWTNRY PDAKMDPMNI WD DIITNRC FFLSKIEEKL TPLPEDNSMN VDQDGDPSDR MEVQEQEEDI SSLIRSCKFS MKM KMIDSA RKQNNFSLAM KLLKELHKES KTRDDWLVSW VQSYCRLSHC RSRSQGCSEQ VLTV LKTVS LLDENNVSSY LSKNILAFRD QNILLGTTYR IIANALSSEP ACLAEIEEDK ARRIL ELSG SSSEDSEKVI AGLYQRAFQH LSEAVQAAEE EAQPPSWSCG PAAGVIDAYM TLADFC DQQ LRKEEENASV IDSAELQAYP ALVVEKMLKA LKLNSNEARL KFPRLLQIIE RYPEETL SL MTKEISSVPC WQFISWISHM VALLDKDQAV AVQHSVEEIT DNYPQAIVYP FIISSESY S FKDTSTGHKN KEFVARIKSK LDQGGVIQDF INALDQLSNP ELLFKDWSND VRAELAKTP VNKKNIEKMY ERMYAALGDP KAPGLGAFRR KFIQTFGKEF DKHFGKGGSK LLRMKLSDFN DITNMLLLK MNKDSKPPGN LKECSPWMSD FKVEFLRNEL EIPGQYDGRG KPLPEYHVRI A GFDERVTV MASLRRPKRI IIRGHDEREH PFLVKGGEDL RQDQRVEQLF QVMNGILAQD SA CSQRALQ LRTYSVVPMT SRLGLIEWLE NTVTLKDLLL NTMSQEEKAA YLSDPRAPPC EYK DWLTKM SGKHDVGAYM LMYKGANRTE TVTSFRKRES KVPADLLKRA FVRMSTSPEA FLAL RSHFA SSHALICISH WILGIGDRHL NNFMVAMETG GVIGIDFGHA FGSATQFLPV PELMP FRLT RQFINLMLPM KETGLMYSIM VHALRAFRSD PGLLTNTMDV FVKEPSFDWK NFEQKM LKK GGSWIQEINV AEKNWYPRQK ICYAKRKLAG ANPAVITCDE LLLGHEKAPA FRDYVAV AR GSKDHNIRAQ EPESGLSEET QVKCLMDQAT DPNILGRTWE GWEPWM |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 1.3 mg/mL |
|---|---|
| Buffer | pH: 7.4 |
| Grid | Model: Quantifoil R2/1 / Material: COPPER / Mesh: 200 / Support film - Material: GRAPHENE OXIDE / Support film - topology: CONTINUOUS / Support film - Film thickness: 200 / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 10 sec. / Pretreatment - Atmosphere: AIR |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 302 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | TFS KRIOS |
|---|---|
| Temperature | Min: 70.0 K / Max: 70.0 K |
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Digitization - Dimensions - Width: 5760 pixel / Digitization - Dimensions - Height: 4092 pixel / Number grids imaged: 1 / Number real images: 13132 / Average exposure time: 4.0 sec. / Average electron dose: 65.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 100.0 µm / Calibrated defocus max: 4.0 µm / Calibrated defocus min: 2.0 µm / Calibrated magnification: 105000 / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.0 mm / Nominal defocus max: 4.0 µm / Nominal defocus min: 2.0 µm / Nominal magnification: 105000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Refinement | Space: REAL / Protocol: FLEXIBLE FIT / Target criteria: CC |
|---|
 Movie
Movie Controller
Controller














 Z
Z Y
Y X
X