[English] 日本語
 Yorodumi
Yorodumi- EMDB-13698: Substrate-engaged mycobacterial Proteasome-associated ATPase in c... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-13698 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Substrate-engaged mycobacterial Proteasome-associated ATPase in complex with open-gate 20S CP - composite map (state B) | |||||||||
 Map data Map data | Substrate-engaged mycobacterial proteasomal ATPase in complex with open gate 20S proteasome | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | AAA motor / ATPAse / mycobacterium / proteasome activator / 20S CP / CYTOSOLIC PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationprotein pupylation / retrograde protein transport, ER to cytosol / proteasome binding / proteasome endopeptidase complex / proteasome core complex, beta-subunit complex / polyubiquitin modification-dependent protein binding / proteasomal protein catabolic process / proteasome core complex, alpha-subunit complex / threonine-type endopeptidase activity / proteasome complex ...protein pupylation / retrograde protein transport, ER to cytosol / proteasome binding / proteasome endopeptidase complex / proteasome core complex, beta-subunit complex / polyubiquitin modification-dependent protein binding / proteasomal protein catabolic process / proteasome core complex, alpha-subunit complex / threonine-type endopeptidase activity / proteasome complex / modification-dependent protein catabolic process / protein tag activity / ATP hydrolysis activity / ATP binding / cytoplasm / cytosol Similarity search - Function | |||||||||
| Biological species |  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 4.0 Å | |||||||||
 Authors Authors | Jomaa A / Kavalchuk M | |||||||||
| Funding support |  Switzerland, 1 items Switzerland, 1 items
| |||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2022 Journal: Nat Commun / Year: 2022Title: Structural basis of prokaryotic ubiquitin-like protein engagement and translocation by the mycobacterial Mpa-proteasome complex. Authors: Mikhail Kavalchuk / Ahmad Jomaa / Andreas U Müller / Eilika Weber-Ban /  Abstract: Proteasomes are present in eukaryotes, archaea and Actinobacteria, including the human pathogen Mycobacterium tuberculosis, where proteasomal degradation supports persistence inside the host. In ...Proteasomes are present in eukaryotes, archaea and Actinobacteria, including the human pathogen Mycobacterium tuberculosis, where proteasomal degradation supports persistence inside the host. In mycobacteria and other members of Actinobacteria, prokaryotic ubiquitin-like protein (Pup) serves as a degradation tag post-translationally conjugated to target proteins for their recruitment to the mycobacterial proteasome ATPase (Mpa). Here, we use single-particle cryo-electron microscopy to determine the structure of Mpa in complex with the 20S core particle at an early stage of pupylated substrate recruitment, shedding light on the mechanism of substrate translocation. Two conformational states of Mpa show how substrate is translocated stepwise towards the degradation chamber of the proteasome core particle. We also demonstrate, in vitro and in vivo, the importance of a structural feature in Mpa that allows formation of alternating charge-complementary interactions with the proteasome resulting in radial, rail-guided movements during the ATPase conformational cycle. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_13698.map.gz emd_13698.map.gz | 10.8 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-13698-v30.xml emd-13698-v30.xml emd-13698.xml emd-13698.xml | 16.4 KB 16.4 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_13698.png emd_13698.png | 47.7 KB | ||
| Filedesc metadata |  emd-13698.cif.gz emd-13698.cif.gz | 6.9 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-13698 http://ftp.pdbj.org/pub/emdb/structures/EMD-13698 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-13698 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-13698 | HTTPS FTP |
-Validation report
| Summary document |  emd_13698_validation.pdf.gz emd_13698_validation.pdf.gz | 398 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_13698_full_validation.pdf.gz emd_13698_full_validation.pdf.gz | 397.6 KB | Display | |
| Data in XML |  emd_13698_validation.xml.gz emd_13698_validation.xml.gz | 6.8 KB | Display | |
| Data in CIF |  emd_13698_validation.cif.gz emd_13698_validation.cif.gz | 7.7 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-13698 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-13698 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-13698 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-13698 | HTTPS FTP |
-Related structure data
| Related structure data |  7pxdMC  7px9C  7pxaC  7pxbC  7pxcC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_13698.map.gz / Format: CCP4 / Size: 166.4 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_13698.map.gz / Format: CCP4 / Size: 166.4 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Substrate-engaged mycobacterial proteasomal ATPase in complex with open gate 20S proteasome | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.222 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
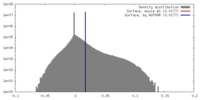
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : Mycobacterial Proteasome-associated ATPase in complex with substr...
| Entire | Name: Mycobacterial Proteasome-associated ATPase in complex with substrate and open-gate 20SCP |
|---|---|
| Components |
|
-Supramolecule #1: Mycobacterial Proteasome-associated ATPase in complex with substr...
| Supramolecule | Name: Mycobacterial Proteasome-associated ATPase in complex with substrate and open-gate 20SCP type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#4 |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 1.1 MDa |
-Macromolecule #1: Proteasome subunit alpha
| Macromolecule | Name: Proteasome subunit alpha / type: protein_or_peptide / ID: 1 Details: The first 7 residues were removed (open gate proteasome) Number of copies: 14 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 26.911039 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MSFPYFISPE QAMRERSELA RKGIARAKSV VALAYAGGVL FVAENPSRSL QKISELYDRV GFAAAGKFNE FDNLRRGGIQ FADTRGYAY DRRDVTGRQL ANVYAQTLGT IFTEQAKPYE VELCVAEVAH YGETKRPELY RITYDGSIAD EPHFVVMGGT T EPIANALK ...String: MSFPYFISPE QAMRERSELA RKGIARAKSV VALAYAGGVL FVAENPSRSL QKISELYDRV GFAAAGKFNE FDNLRRGGIQ FADTRGYAY DRRDVTGRQL ANVYAQTLGT IFTEQAKPYE VELCVAEVAH YGETKRPELY RITYDGSIAD EPHFVVMGGT T EPIANALK ESYAENASLT DALRIAVAAL RAGSADTSGG DQPTLGVASL EVAVLDANRP RRAFRRITGS ALQALLVDQE SP QSDGESS G UniProtKB: Proteasome subunit alpha |
-Macromolecule #2: AAA ATPase forming ring-shaped complexes
| Macromolecule | Name: AAA ATPase forming ring-shaped complexes / type: protein_or_peptide / ID: 2 Details: C-terminal extension of Mpa containing GQYL motif which interacts with the proteasome Number of copies: 7 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 67.48793 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MGESERSEAF GIPRDSPLSS GDAAELEQLR REAAVLREQL ENAVGSHAPT RSARDIHQLE ARIDSLAARN SKLMETLKEA RQQLLALRE EVDRLGQPPS GYGVLLATHD DDTVDVFTSG RKMRLTCSPN IDAASLKKGQ TVRLNEALTV VEAGTFEAVG E ISTLREIL ...String: MGESERSEAF GIPRDSPLSS GDAAELEQLR REAAVLREQL ENAVGSHAPT RSARDIHQLE ARIDSLAARN SKLMETLKEA RQQLLALRE EVDRLGQPPS GYGVLLATHD DDTVDVFTSG RKMRLTCSPN IDAASLKKGQ TVRLNEALTV VEAGTFEAVG E ISTLREIL ADGHRALVVG HADEERVVWL ADPLIAEDLP DGLPEALNDD TRPRKLRPGD SLLVDTKAGY AFERIPKAEV ED LVLEEVP DVSYADIGGL SRQIEQIRDA VELPFLHKEL YREYSLRPPK GVLLYGPPGC GKTLIAKAVA NSLAKKMAEV RGD DAHEAK SYFLNIKGPE LLNKFVGETE RHIRLIFQRA REKASEGTPV IVFFDEMDSI FRTRGTGVSS DVETTVVPQL LSEI DGVEG LENVIVIGAS NREDMIDPAI LRPGRLDVKI KIERPDAEAA QDIYSKYLTE FLPVHADDLA EFDGDRSACI KAMIE KVVD RMYAEIDDNR FLEVTYANGD KEVMYFKDFN SGAMIQNVVD RAKKNAIKSV LETGQPGLRI QHLLDSIVDE FAENED LPN TTNPDDWARI SGKKGERIVY IRTLVTGKSS SASRAIDTES NLGQYL UniProtKB: AAA ATPase forming ring-shaped complexes |
-Macromolecule #3: Prokaryotic ubiquitin-like protein Pup
| Macromolecule | Name: Prokaryotic ubiquitin-like protein Pup / type: protein_or_peptide / ID: 3 / Details: GS residues are left from the TEV cleavage site / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 7.095416 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: GSMAQEQTKR GGGGGDDDDI AGSTAAGQER REKLTEETDD LLDEIDDVLE ENAEDFVRAY VQKGGQ UniProtKB: Prokaryotic ubiquitin-like protein Pup |
-Macromolecule #4: Proteasome subunit beta
| Macromolecule | Name: Proteasome subunit beta / type: protein_or_peptide / ID: 4 Details: Propetide (the first N-terminal 57 residues) is removed Number of copies: 14 / Enantiomer: LEVO / EC number: proteasome endopeptidase complex |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 30.332006 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MTWPLPDRLS INSLSGTPAV DLSSFTDFLR RQAPELLPAS ISGGAPLAGG DAQLPHGTTI VALKYPGGVV MAGDRRSTQG NMISGRDVR KVYITDDYTA TGIAGTAAVA VEFARLYAVE LEHYEKLEGV PLTFAGKINR LAIMVRGNLA AAMQGLLALP L LAGYDIHA ...String: MTWPLPDRLS INSLSGTPAV DLSSFTDFLR RQAPELLPAS ISGGAPLAGG DAQLPHGTTI VALKYPGGVV MAGDRRSTQG NMISGRDVR KVYITDDYTA TGIAGTAAVA VEFARLYAVE LEHYEKLEGV PLTFAGKINR LAIMVRGNLA AAMQGLLALP L LAGYDIHA SDPQSAGRIV SFDAAGGWNI EEEGYQAVGS GSLFAKSSMK KLYSQVTDGD SGLRVAVEAL YDAADDDSAT GG PDLVRGI FPTAVIIDAD GAVDVPESRI AELARAIIES RSGADTFGSD GGEK UniProtKB: Proteasome subunit beta |
-Macromolecule #5: ADENOSINE-5'-TRIPHOSPHATE
| Macromolecule | Name: ADENOSINE-5'-TRIPHOSPHATE / type: ligand / ID: 5 / Number of copies: 6 / Formula: ATP |
|---|---|
| Molecular weight | Theoretical: 507.181 Da |
| Chemical component information |  ChemComp-ATP: |
-Macromolecule #6: MAGNESIUM ION
| Macromolecule | Name: MAGNESIUM ION / type: ligand / ID: 6 / Number of copies: 6 / Formula: MG |
|---|---|
| Molecular weight | Theoretical: 24.305 Da |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Vitrification | Cryogen name: ETHANE-PROPANE / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 50.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.5 µm / Nominal defocus min: 1.5 µm / Nominal magnification: 105000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Initial model |
| ||||||
|---|---|---|---|---|---|---|---|
| Refinement | Space: REAL / Protocol: RIGID BODY FIT | ||||||
| Output model |  PDB-7pxd: |
 Movie
Movie Controller
Controller

















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)