+検索条件
-Structure paper
| タイトル | Molecular basis for glycan recognition and reaction priming of eukaryotic oligosaccharyltransferase. |
|---|---|
| ジャーナル・号・ページ | Nat Commun, Vol. 13, Issue 1, Page 7296, Year 2022 |
| 掲載日 | 2022年11月26日 |
 著者 著者 | Ana S Ramírez / Mario de Capitani / Giorgio Pesciullesi / Julia Kowal / Joël S Bloch / Rossitza N Irobalieva / Jean-Louis Reymond / Markus Aebi / Kaspar P Locher /  |
| PubMed 要旨 | Oligosaccharyltransferase (OST) is the central enzyme of N-linked protein glycosylation. It catalyzes the transfer of a pre-assembled glycan, GlcNAcManGlc, from a dolichyl-pyrophosphate donor to ...Oligosaccharyltransferase (OST) is the central enzyme of N-linked protein glycosylation. It catalyzes the transfer of a pre-assembled glycan, GlcNAcManGlc, from a dolichyl-pyrophosphate donor to acceptor sites in secretory proteins in the lumen of the endoplasmic reticulum. Precise recognition of the fully assembled glycan by OST is essential for the subsequent quality control steps of glycoprotein biosynthesis. However, the molecular basis of the OST-donor glycan interaction is unknown. Here we present cryo-EM structures of S. cerevisiae OST in distinct functional states. Our findings reveal that the terminal glucoses (Glc) of a chemo-enzymatically generated donor glycan analog bind to a pocket formed by the non-catalytic subunits WBP1 and OST2. We further find that binding either donor or acceptor substrate leads to distinct primed states of OST, where subsequent binding of the other substrate triggers conformational changes required for catalysis. This alternate priming allows OST to efficiently process closely spaced N-glycosylation sites. |
 リンク リンク |  Nat Commun / Nat Commun /  PubMed:36435935 / PubMed:36435935 /  PubMed Central PubMed Central |
| 手法 | EM (単粒子) |
| 解像度 | 2.8 - 3.3 Å |
| 構造データ | EMDB-15419, PDB-8agb: EMDB-15420, PDB-8agc: EMDB-15421, PDB-8age: |
| 化合物 |  ChemComp-CPL:  ChemComp-PTY:  ChemComp-NAG:  ChemComp-MN: 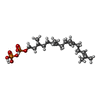 ChemComp-ELU: 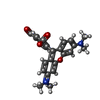 ChemComp-323: 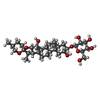 ChemComp-KZB:  ChemComp-HOH: |
| 由来 |
|
 キーワード キーワード | MEMBRANE PROTEIN / OST complex / oligosaccharyltransferase / N-linked glycosylation / yeast / TRANSFERASE / N-glycosylation OST complex |
 ムービー
ムービー コントローラー
コントローラー 構造ビューア
構造ビューア 万見文献について
万見文献について












