[English] 日本語
 Yorodumi
Yorodumi- EMDB-8576: Structure of the Plasmodium falciparum 80S ribosome bound to the ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-8576 | |||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structure of the Plasmodium falciparum 80S ribosome bound to the antimalarial drug mefloquine | |||||||||||||||||||||||||||
 Map data Map data | ||||||||||||||||||||||||||||
 Sample Sample |
| |||||||||||||||||||||||||||
 Keywords Keywords | Ribosome / protein synthesis / antimalarial | |||||||||||||||||||||||||||
| Function / homology |  Function and homology information Function and homology informationTranslesion synthesis by REV1 / Recognition of DNA damage by PCNA-containing replication complex / Translesion Synthesis by POLH / Translesion synthesis by POLK / Translesion synthesis by POLI / Josephin domain DUBs / Metalloprotease DUBs / DNA Damage Recognition in GG-NER / Formation of Incision Complex in GG-NER / Dual Incision in GG-NER ...Translesion synthesis by REV1 / Recognition of DNA damage by PCNA-containing replication complex / Translesion Synthesis by POLH / Translesion synthesis by POLK / Translesion synthesis by POLI / Josephin domain DUBs / Metalloprotease DUBs / DNA Damage Recognition in GG-NER / Formation of Incision Complex in GG-NER / Dual Incision in GG-NER / Formation of TC-NER Pre-Incision Complex / Dual incision in TC-NER / Gap-filling DNA repair synthesis and ligation in TC-NER / PTK6 Regulates RTKs and Their Effectors AKT1 and DOK1 / ER Quality Control Compartment (ERQC) / Iron uptake and transport / L13a-mediated translational silencing of Ceruloplasmin expression / SRP-dependent cotranslational protein targeting to membrane / Translation initiation complex formation / Formation of a pool of free 40S subunits / Formation of the ternary complex, and subsequently, the 43S complex / Ribosomal scanning and start codon recognition / GTP hydrolysis and joining of the 60S ribosomal subunit / Negative regulators of DDX58/IFIH1 signaling / Nonsense Mediated Decay (NMD) independent of the Exon Junction Complex (EJC) / Nonsense Mediated Decay (NMD) enhanced by the Exon Junction Complex (EJC) / Aggrephagy / Synthesis of active ubiquitin: roles of E1 and E2 enzymes / Orc1 removal from chromatin / CDK-mediated phosphorylation and removal of Cdc6 / FBXL7 down-regulates AURKA during mitotic entry and in early mitosis / KEAP1-NFE2L2 pathway / UCH proteinases / Ub-specific processing proteases / Neddylation / MAPK6/MAPK4 signaling / Antigen processing: Ubiquitination & Proteasome degradation / ABC-family proteins mediated transport / AUF1 (hnRNP D0) binds and destabilizes mRNA / protein-RNA complex assembly / maturation of LSU-rRNA / maturation of LSU-rRNA from tricistronic rRNA transcript (SSU-rRNA, 5.8S rRNA, LSU-rRNA) / ribosomal large subunit biogenesis / ribosomal large subunit assembly / modification-dependent protein catabolic process / protein tag activity / large ribosomal subunit / ribosome biogenesis / 5S rRNA binding / large ribosomal subunit rRNA binding / ubiquitin-dependent protein catabolic process / cytosolic small ribosomal subunit / cytosolic large ribosomal subunit / cytoplasmic translation / rRNA binding / negative regulation of translation / ribosome / protein ubiquitination / structural constituent of ribosome / translation / ribonucleoprotein complex / mRNA binding / ubiquitin protein ligase binding / RNA binding / nucleus / metal ion binding / cytoplasm Similarity search - Function | |||||||||||||||||||||||||||
| Biological species |  | |||||||||||||||||||||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.2 Å | |||||||||||||||||||||||||||
 Authors Authors | Wong W / Bai X-C | |||||||||||||||||||||||||||
| Funding support |  Australia, Australia,  United Kingdom, 8 items United Kingdom, 8 items
| |||||||||||||||||||||||||||
 Citation Citation |  Journal: Nat Microbiol / Year: 2017 Journal: Nat Microbiol / Year: 2017Title: Mefloquine targets the Plasmodium falciparum 80S ribosome to inhibit protein synthesis. Authors: Wilson Wong / Xiao-Chen Bai / Brad E Sleebs / Tony Triglia / Alan Brown / Jennifer K Thompson / Katherine E Jackson / Eric Hanssen / Danushka S Marapana / Israel S Fernandez / Stuart A Ralph ...Authors: Wilson Wong / Xiao-Chen Bai / Brad E Sleebs / Tony Triglia / Alan Brown / Jennifer K Thompson / Katherine E Jackson / Eric Hanssen / Danushka S Marapana / Israel S Fernandez / Stuart A Ralph / Alan F Cowman / Sjors H W Scheres / Jake Baum /   Abstract: Malaria control is heavily dependent on chemotherapeutic agents for disease prevention and drug treatment. Defining the mechanism of action for licensed drugs, for which no target is characterized, ...Malaria control is heavily dependent on chemotherapeutic agents for disease prevention and drug treatment. Defining the mechanism of action for licensed drugs, for which no target is characterized, is critical to the development of their second-generation derivatives to improve drug potency towards inhibition of their molecular targets. Mefloquine is a widely used antimalarial without a known mode of action. Here, we demonstrate that mefloquine is a protein synthesis inhibitor. We solved a 3.2 Å cryo-electron microscopy structure of the Plasmodium falciparum 80S ribosome with the (+)-mefloquine enantiomer bound to the ribosome GTPase-associated centre. Mutagenesis of mefloquine-binding residues generates parasites with increased resistance, confirming the parasite-killing mechanism. Furthermore, structure-guided derivatives with an altered piperidine group, predicted to improve binding, show enhanced parasiticidal effect. These data reveal one possible mode of action for mefloquine and demonstrate the vast potential of cryo-electron microscopy to guide the development of mefloquine derivatives to inhibit parasite protein synthesis. | |||||||||||||||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_8576.map.gz emd_8576.map.gz | 319.3 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-8576-v30.xml emd-8576-v30.xml emd-8576.xml emd-8576.xml | 67.6 KB 67.6 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_8576.png emd_8576.png | 86.6 KB | ||
| Filedesc metadata |  emd-8576.cif.gz emd-8576.cif.gz | 14 KB | ||
| Others |  emd_8576_half_map_1.map.gz emd_8576_half_map_1.map.gz emd_8576_half_map_2.map.gz emd_8576_half_map_2.map.gz | 140.5 MB 140.6 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-8576 http://ftp.pdbj.org/pub/emdb/structures/EMD-8576 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-8576 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-8576 | HTTPS FTP |
-Validation report
| Summary document |  emd_8576_validation.pdf.gz emd_8576_validation.pdf.gz | 387.9 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_8576_full_validation.pdf.gz emd_8576_full_validation.pdf.gz | 387 KB | Display | |
| Data in XML |  emd_8576_validation.xml.gz emd_8576_validation.xml.gz | 7.5 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-8576 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-8576 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-8576 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-8576 | HTTPS FTP |
-Related structure data
| Related structure data |  5umdMC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_8576.map.gz / Format: CCP4 / Size: 347.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_8576.map.gz / Format: CCP4 / Size: 347.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Voxel size | X=Y=Z: 1.03 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Half map: #1
| File | emd_8576_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #2
| File | emd_8576_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
+Entire : Plasmodium falciparum 80S ribosome bound to mefloquine
+Supramolecule #1: Plasmodium falciparum 80S ribosome bound to mefloquine
+Macromolecule #1: 28S ribosomal RNA
+Macromolecule #2: 5S ribosomal RNA
+Macromolecule #3: 5.8S ribosomal RNA
+Macromolecule #4: 60S ribosomal protein L2
+Macromolecule #5: 60S ribosomal protein L3
+Macromolecule #6: 60S ribosomal protein L4
+Macromolecule #7: 60S ribosomal protein L11a, putative
+Macromolecule #8: 60S ribosomal protein L6, putative
+Macromolecule #9: 60S ribosomal protein L6-2, putative
+Macromolecule #10: 60S ribosomal protein L7-3, putative
+Macromolecule #11: 60S ribosomal protein L13, putative
+Macromolecule #12: 60S ribosomal protein L13
+Macromolecule #13: 60S ribosomal protein L23, putative
+Macromolecule #14: 60S ribosomal protein L14, putative
+Macromolecule #15: 60S ribosomal protein L27a, putative
+Macromolecule #16: Ribosomal protein L15
+Macromolecule #17: 60S ribosomal protein L10, putative
+Macromolecule #18: 60S ribosomal protein L5, putative
+Macromolecule #19: 60S ribosomal protein L18-2, putative
+Macromolecule #20: 60S ribosomal protein L19
+Macromolecule #21: 60S ribosomal protein L18a
+Macromolecule #22: 60S ribosomal protein L21
+Macromolecule #23: 60S ribosomal protein L17, putative
+Macromolecule #24: 60S ribosomal protein L22, putative
+Macromolecule #25: 60S ribosomal protein L23
+Macromolecule #26: 60S ribosomal protein L26, putative
+Macromolecule #27: 60S ribosomal protein L24, putative
+Macromolecule #28: 60S ribosomal protein L27
+Macromolecule #29: 60S ribosomal protein L28
+Macromolecule #30: 60S ribosomal protein L35, putative
+Macromolecule #31: 60S ribosomal protein L29, putative
+Macromolecule #32: 60S ribosomal protein L7, putative
+Macromolecule #33: 60S ribosomal protein L30e, putative
+Macromolecule #34: 60S ribosomal protein L31
+Macromolecule #35: 60S ribosomal protein L32
+Macromolecule #36: 60S ribosomal protein L35Ae, putative
+Macromolecule #37: 60S ribosomal protein L34
+Macromolecule #38: 60S ribosomal protein L36
+Macromolecule #39: Ribosomal protein L37
+Macromolecule #40: 60S ribosomal protein L38
+Macromolecule #41: 60S ribosomal protein L39
+Macromolecule #42: Ubiquitin-60S ribosomal protein L40
+Macromolecule #43: 60S ribosomal protein L41
+Macromolecule #44: 60S ribosomal protein L37a
+Macromolecule #45: 60S ribosomal protein L44
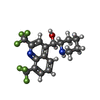
+Macromolecule #46: Mefloquine
+Macromolecule #47: MAGNESIUM ION
+Macromolecule #48: ZINC ION
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.4 Details: 20 mM Hepes pH 7.4, 40 mM KAc, 10 mM NH4Ac, 10 mM Mg(Ac)2 and 5 mM 2-mecaptoethanol |
|---|---|
| Grid | Model: Quantifoil R2/2 / Material: COPPER / Support film - Material: CARBON / Support film - topology: CONTINUOUS / Support film - Film thickness: 0.02 / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 30 sec. / Details: Blot 2.5s before plunging |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 90 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI POLARA 300 |
|---|---|
| Temperature | Min: 80.0 K / Max: 90.0 K |
| Image recording | Film or detector model: FEI FALCON II (4k x 4k) / Number grids imaged: 1 / Average electron dose: 20.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Calibrated defocus max: 3.8000000000000003 µm / Calibrated defocus min: 0.8 µm / Calibrated magnification: 104748 / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.0 mm / Nominal magnification: 78000 |
| Sample stage | Specimen holder model: GATAN LIQUID NITROGEN / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Tecnai Polara / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Refinement | Space: RECIPROCAL / Protocol: RIGID BODY FIT / Overall B value: 129.3 |
|---|---|
| Output model |  PDB-5umd: |
 Movie
Movie Controller
Controller




















 Z
Z Y
Y X
X