[English] 日本語
 Yorodumi
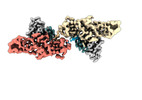
Yorodumi- PDB-8jaf: Structure of Muscarinic receptor (M2R) in complex with beta-arres... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 8jaf | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
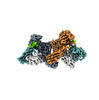
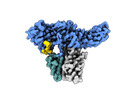
| Title | Structure of Muscarinic receptor (M2R) in complex with beta-arrestin1 (Local Refine, non-cross linked) | |||||||||||||||
 Components Components |
| |||||||||||||||
 Keywords Keywords |  SIGNALING PROTEIN/IMMUNE SYSTEM / SIGNALING PROTEIN/IMMUNE SYSTEM /  GPCR / GPCR /  Arrestin / Arrestin /  SIGNALING PROTEIN / SIGNALING PROTEIN /  SIGNALING PROTEIN-IMMUNE SYSTEM complex SIGNALING PROTEIN-IMMUNE SYSTEM complex | |||||||||||||||
| Function / homology |  Function and homology information Function and homology informationMAP2K and MAPK activation / Activation of SMO / Golgi Associated Vesicle Biogenesis / Lysosome Vesicle Biogenesis / AP-2 adaptor complex binding /  Muscarinic acetylcholine receptors / symmetric synapse / phospholipase C-activating G protein-coupled acetylcholine receptor signaling pathway / clathrin heavy chain binding / clathrin coat of coated pit ...MAP2K and MAPK activation / Activation of SMO / Golgi Associated Vesicle Biogenesis / Lysosome Vesicle Biogenesis / AP-2 adaptor complex binding / Muscarinic acetylcholine receptors / symmetric synapse / phospholipase C-activating G protein-coupled acetylcholine receptor signaling pathway / clathrin heavy chain binding / clathrin coat of coated pit ...MAP2K and MAPK activation / Activation of SMO / Golgi Associated Vesicle Biogenesis / Lysosome Vesicle Biogenesis / AP-2 adaptor complex binding /  Muscarinic acetylcholine receptors / symmetric synapse / phospholipase C-activating G protein-coupled acetylcholine receptor signaling pathway / clathrin heavy chain binding / clathrin coat of coated pit / Ub-specific processing proteases / G protein-coupled acetylcholine receptor activity / regulation of smooth muscle contraction / desensitization of G protein-coupled receptor signaling pathway / cholinergic synapse / Cargo recognition for clathrin-mediated endocytosis / Muscarinic acetylcholine receptors / symmetric synapse / phospholipase C-activating G protein-coupled acetylcholine receptor signaling pathway / clathrin heavy chain binding / clathrin coat of coated pit / Ub-specific processing proteases / G protein-coupled acetylcholine receptor activity / regulation of smooth muscle contraction / desensitization of G protein-coupled receptor signaling pathway / cholinergic synapse / Cargo recognition for clathrin-mediated endocytosis /  inositol hexakisphosphate binding / adenylate cyclase-inhibiting G protein-coupled acetylcholine receptor signaling pathway / inositol hexakisphosphate binding / adenylate cyclase-inhibiting G protein-coupled acetylcholine receptor signaling pathway /  Clathrin-mediated endocytosis / Clathrin-mediated endocytosis /  clathrin-dependent endocytosis / G protein-coupled serotonin receptor activity / arrestin family protein binding / G protein-coupled receptor internalization / clathrin-dependent endocytosis / G protein-coupled serotonin receptor activity / arrestin family protein binding / G protein-coupled receptor internalization /  acetylcholine receptor binding / Thrombin signalling through proteinase activated receptors (PARs) / G alpha (s) signalling events / acetylcholine receptor binding / Thrombin signalling through proteinase activated receptors (PARs) / G alpha (s) signalling events /  regulation of heart contraction / regulation of heart contraction /  clathrin binding / negative regulation of Notch signaling pathway / clathrin binding / negative regulation of Notch signaling pathway /  pseudopodium / G protein-coupled receptor signaling pathway, coupled to cyclic nucleotide second messenger / phosphatidylinositol-3,4,5-trisphosphate binding / pseudopodium / G protein-coupled receptor signaling pathway, coupled to cyclic nucleotide second messenger / phosphatidylinositol-3,4,5-trisphosphate binding /  small molecule binding / positive regulation of receptor internalization / asymmetric synapse / axon terminus / presynaptic modulation of chemical synaptic transmission / small molecule binding / positive regulation of receptor internalization / asymmetric synapse / axon terminus / presynaptic modulation of chemical synaptic transmission /  visual perception / G protein-coupled receptor binding / clathrin-coated endocytic vesicle membrane / response to virus / adenylate cyclase-modulating G protein-coupled receptor signaling pathway / G protein-coupled acetylcholine receptor signaling pathway / visual perception / G protein-coupled receptor binding / clathrin-coated endocytic vesicle membrane / response to virus / adenylate cyclase-modulating G protein-coupled receptor signaling pathway / G protein-coupled acetylcholine receptor signaling pathway /  receptor internalization / receptor internalization /  protein transport / Cargo recognition for clathrin-mediated endocytosis / protein transport / Cargo recognition for clathrin-mediated endocytosis /  presynaptic membrane / presynaptic membrane /  Clathrin-mediated endocytosis / Clathrin-mediated endocytosis /  nervous system development / G alpha (i) signalling events / ubiquitin-dependent protein catabolic process / chemical synaptic transmission / cytoplasmic vesicle / nervous system development / G alpha (i) signalling events / ubiquitin-dependent protein catabolic process / chemical synaptic transmission / cytoplasmic vesicle /  postsynaptic membrane / positive regulation of ERK1 and ERK2 cascade / molecular adaptor activity / positive regulation of protein phosphorylation / G protein-coupled receptor signaling pathway / postsynaptic membrane / positive regulation of ERK1 and ERK2 cascade / molecular adaptor activity / positive regulation of protein phosphorylation / G protein-coupled receptor signaling pathway /  dendrite / neuronal cell body / glutamatergic synapse / dendrite / neuronal cell body / glutamatergic synapse /  synapse / synapse /  signal transduction / signal transduction /  membrane / membrane /  nucleus / nucleus /  plasma membrane / plasma membrane /  cytosol / cytosol /  cytoplasm cytoplasmSimilarity search - Function | |||||||||||||||
| Biological species |   Bos taurus (cattle) Bos taurus (cattle)  Mus musculus (house mouse) Mus musculus (house mouse)  Homo sapiens (human) Homo sapiens (human) | |||||||||||||||
| Method |  ELECTRON MICROSCOPY / ELECTRON MICROSCOPY /  single particle reconstruction / single particle reconstruction /  cryo EM / Resolution: 3.1 Å cryo EM / Resolution: 3.1 Å | |||||||||||||||
 Authors Authors | Maharana, J. / Sano, F.K. / Shihoya, W. / Banerjee, R. / Nureki, O. / Shukla, A.K. | |||||||||||||||
| Funding support |  India, 4items India, 4items
| |||||||||||||||
 Citation Citation |  Journal: Science / Year: 2024 Journal: Science / Year: 2024Title: Molecular insights into atypical modes of β-arrestin interaction with seven transmembrane receptors. Authors: Jagannath Maharana / Fumiya K Sano / Parishmita Sarma / Manish K Yadav / Longhan Duan / Tomasz M Stepniewski / Madhu Chaturvedi / Ashutosh Ranjan / Vinay Singh / Sayantan Saha / Gargi ...Authors: Jagannath Maharana / Fumiya K Sano / Parishmita Sarma / Manish K Yadav / Longhan Duan / Tomasz M Stepniewski / Madhu Chaturvedi / Ashutosh Ranjan / Vinay Singh / Sayantan Saha / Gargi Mahajan / Mohamed Chami / Wataru Shihoya / Jana Selent / Ka Young Chung / Ramanuj Banerjee / Osamu Nureki / Arun K Shukla /      Abstract: β-arrestins (βarrs) are multifunctional proteins involved in signaling and regulation of seven transmembrane receptors (7TMRs), and their interaction is driven primarily by agonist-induced receptor ...β-arrestins (βarrs) are multifunctional proteins involved in signaling and regulation of seven transmembrane receptors (7TMRs), and their interaction is driven primarily by agonist-induced receptor activation and phosphorylation. Here, we present seven cryo-electron microscopy structures of βarrs either in the basal state, activated by the muscarinic receptor subtype 2 (M2R) through its third intracellular loop, or activated by the βarr-biased decoy D6 receptor (D6R). Combined with biochemical, cellular, and biophysical experiments, these structural snapshots allow the visualization of atypical engagement of βarrs with 7TMRs and also reveal a structural transition in the carboxyl terminus of βarr2 from a β strand to an α helix upon activation by D6R. Our study provides previously unanticipated molecular insights into the structural and functional diversity encoded in 7TMR-βarr complexes with direct implications for exploring novel therapeutic avenues. | |||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  8jaf.cif.gz 8jaf.cif.gz | 104.4 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb8jaf.ent.gz pdb8jaf.ent.gz | 74.1 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  8jaf.json.gz 8jaf.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/ja/8jaf https://data.pdbj.org/pub/pdb/validation_reports/ja/8jaf ftp://data.pdbj.org/pub/pdb/validation_reports/ja/8jaf ftp://data.pdbj.org/pub/pdb/validation_reports/ja/8jaf | HTTPS FTP |
|---|
-Related structure data
| Related structure data | 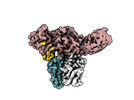 36126MC  8go9C  8j8rC  8j8vC  8j8zC  8j97C  8j9kC  8ja3C M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
| #1: Protein |  Arrestin / Arrestin beta-1 / Arrestin-2 Arrestin / Arrestin beta-1 / Arrestin-2Mass: 40247.270 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Bos taurus (cattle) / Gene: ARRB1 / Production host: Bos taurus (cattle) / Gene: ARRB1 / Production host:   Spodoptera frugiperda (fall armyworm) / References: UniProt: P17870 Spodoptera frugiperda (fall armyworm) / References: UniProt: P17870 |
|---|---|
| #2: Antibody | Mass: 12861.245 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Mus musculus (house mouse) / Production host: Mus musculus (house mouse) / Production host:   Escherichia coli (E. coli) Escherichia coli (E. coli) |
| #3: Antibody | Mass: 11500.820 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Mus musculus (house mouse) / Production host: Mus musculus (house mouse) / Production host:   Escherichia coli (E. coli) Escherichia coli (E. coli) |
| #4: Protein/peptide |  Mass: 983.637 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Homo sapiens (human) / Gene: CHRM2 / Production host: Homo sapiens (human) / Gene: CHRM2 / Production host:   Spodoptera frugiperda (fall armyworm) / References: UniProt: P08172 Spodoptera frugiperda (fall armyworm) / References: UniProt: P08172 |
| Has ligand of interest | Y |
-Experimental details
-Experiment
| Experiment | Method:  ELECTRON MICROSCOPY ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method:  single particle reconstruction single particle reconstruction |
- Sample preparation
Sample preparation
| Component |
| ||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Molecular weight | Experimental value: NO | ||||||||||||||||||||||||||||||
| Source (natural) |
| ||||||||||||||||||||||||||||||
| Source (recombinant) |
| ||||||||||||||||||||||||||||||
| Buffer solution | pH: 7.4 | ||||||||||||||||||||||||||||||
| Specimen | Embedding applied: NO / Shadowing applied: NO / Staining applied : NO / Vitrification applied : NO / Vitrification applied : YES : YES | ||||||||||||||||||||||||||||||
Vitrification | Cryogen name: ETHANE |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source : :  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD Bright-field microscopy / Nominal defocus max: 1600 nm / Nominal defocus min: 800 nm / Cs Bright-field microscopy / Nominal defocus max: 1600 nm / Nominal defocus min: 800 nm / Cs : 2.7 mm : 2.7 mm |
| Specimen holder | Cryogen: NITROGEN / Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER |
| Image recording | Electron dose: 50 e/Å2 / Detector mode: COUNTING / Film or detector model: GATAN K3 (6k x 4k) / Num. of real images: 32158 |
| Image scans | Movie frames/image: 40 |
- Processing
Processing
| EM software |
| ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
CTF correction | Type: NONE | ||||||||||||||||||||||||
| Particle selection | Num. of particles selected: 17218446 | ||||||||||||||||||||||||
3D reconstruction | Resolution: 3.1 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 159811 / Symmetry type: POINT | ||||||||||||||||||||||||
| Atomic model building | Protocol: FLEXIBLE FIT / Space: REAL | ||||||||||||||||||||||||
| Atomic model building | PDB-ID: 8GO8 Accession code: 8GO8 / Source name: PDB / Type: experimental model |
 Movie
Movie Controller
Controller



 PDBj
PDBj










