+ Open data
Open data
- Basic information
Basic information
| Entry | 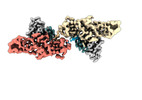 | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
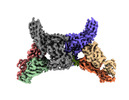
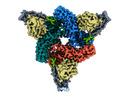
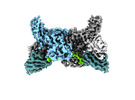
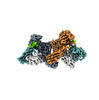
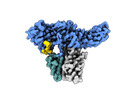
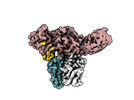
| Title | Structure of beta-arrestin1 in complex with C3aRpp | |||||||||||||||
 Map data Map data | ||||||||||||||||
 Sample Sample |
| |||||||||||||||
 Keywords Keywords | GPCR / Arrestin / SIGNALING PROTEIN / SIGNALING PROTEIN-IMMUNE SYSTEM complex | |||||||||||||||
| Function / homology |  Function and homology information Function and homology informationV2 vasopressin receptor binding / complement component C3a receptor activity / alpha-1A adrenergic receptor binding / follicle-stimulating hormone receptor binding / sensory perception of touch / G alpha (s) signalling events / alpha-1B adrenergic receptor binding / complement component C5a receptor activity / follicle-stimulating hormone signaling pathway / protein phosphorylated amino acid binding ...V2 vasopressin receptor binding / complement component C3a receptor activity / alpha-1A adrenergic receptor binding / follicle-stimulating hormone receptor binding / sensory perception of touch / G alpha (s) signalling events / alpha-1B adrenergic receptor binding / complement component C5a receptor activity / follicle-stimulating hormone signaling pathway / protein phosphorylated amino acid binding / angiotensin receptor binding / Lysosome Vesicle Biogenesis / AP-2 adaptor complex binding / Golgi Associated Vesicle Biogenesis / MAP2K and MAPK activation / Ub-specific processing proteases / positive regulation of smooth muscle cell apoptotic process / negative regulation of interleukin-8 production / Cargo recognition for clathrin-mediated endocytosis / Clathrin-mediated endocytosis / clathrin adaptor activity / complement receptor mediated signaling pathway / regulation of G protein-coupled receptor signaling pathway / arrestin family protein binding / G protein-coupled receptor internalization / positive regulation of neutrophil chemotaxis / response to morphine / blood circulation / Thrombin signalling through proteinase activated receptors (PARs) / mitogen-activated protein kinase kinase binding / azurophil granule membrane / positive regulation of Rho protein signal transduction / clathrin binding / positive regulation of macrophage chemotaxis / negative regulation of Notch signaling pathway / stress fiber assembly / pseudopodium / positive regulation of insulin secretion involved in cellular response to glucose stimulus / negative regulation of interleukin-6 production / cysteine-type endopeptidase inhibitor activity involved in apoptotic process / positive regulation of receptor internalization / phototransduction / positive regulation of vascular endothelial growth factor production / Purinergic signaling in leishmaniasis infection / specific granule membrane / clathrin-coated pit / negative regulation of protein ubiquitination / insulin-like growth factor receptor binding / visual perception / GTPase activator activity / Peptide ligand-binding receptors / negative regulation of protein phosphorylation / positive regulation of protein ubiquitination / Regulation of Complement cascade / G protein-coupled receptor binding / G protein-coupled receptor activity / nuclear estrogen receptor binding / phosphoprotein binding / calcium-mediated signaling / adenylate cyclase-modulating G protein-coupled receptor signaling pathway / negative regulation of ERK1 and ERK2 cascade / endocytosis / positive regulation of angiogenesis / chemotaxis / positive regulation of peptidyl-serine phosphorylation / protein transport / phospholipase C-activating G protein-coupled receptor signaling pathway / positive regulation of cytosolic calcium ion concentration / basolateral plasma membrane / G alpha (i) signalling events / cytoplasmic vesicle / ubiquitin-dependent protein catabolic process / postsynaptic membrane / proteasome-mediated ubiquitin-dependent protein catabolic process / regulation of apoptotic process / transcription coactivator activity / negative regulation of neuron apoptotic process / transmembrane transporter binding / positive regulation of ERK1 and ERK2 cascade / positive regulation of MAPK cascade / endosome / dendritic spine / postsynaptic density / protein ubiquitination / positive regulation of protein phosphorylation / response to xenobiotic stimulus / inflammatory response / G protein-coupled receptor signaling pathway / signaling receptor binding / positive regulation of cell population proliferation / ubiquitin protein ligase binding / glutamatergic synapse / regulation of transcription by RNA polymerase II / Neutrophil degranulation / chromatin / negative regulation of apoptotic process / regulation of DNA-templated transcription / enzyme binding / positive regulation of transcription by RNA polymerase II / nucleus Similarity search - Function | |||||||||||||||
| Biological species |    Homo sapiens (human) Homo sapiens (human) | |||||||||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.94 Å | |||||||||||||||
 Authors Authors | Maharana J / Sarma P / Yadav MK / Chami M / Banerjee R / Shukla AK | |||||||||||||||
| Funding support |  India, 4 items India, 4 items
| |||||||||||||||
 Citation Citation |  Journal: Science / Year: 2024 Journal: Science / Year: 2024Title: Molecular insights into atypical modes of β-arrestin interaction with seven transmembrane receptors. Authors: Jagannath Maharana / Fumiya K Sano / Parishmita Sarma / Manish K Yadav / Longhan Duan / Tomasz M Stepniewski / Madhu Chaturvedi / Ashutosh Ranjan / Vinay Singh / Sayantan Saha / Gargi ...Authors: Jagannath Maharana / Fumiya K Sano / Parishmita Sarma / Manish K Yadav / Longhan Duan / Tomasz M Stepniewski / Madhu Chaturvedi / Ashutosh Ranjan / Vinay Singh / Sayantan Saha / Gargi Mahajan / Mohamed Chami / Wataru Shihoya / Jana Selent / Ka Young Chung / Ramanuj Banerjee / Osamu Nureki / Arun K Shukla /      Abstract: β-arrestins (βarrs) are multifunctional proteins involved in signaling and regulation of seven transmembrane receptors (7TMRs), and their interaction is driven primarily by agonist-induced receptor ...β-arrestins (βarrs) are multifunctional proteins involved in signaling and regulation of seven transmembrane receptors (7TMRs), and their interaction is driven primarily by agonist-induced receptor activation and phosphorylation. Here, we present seven cryo-electron microscopy structures of βarrs either in the basal state, activated by the muscarinic receptor subtype 2 (M2R) through its third intracellular loop, or activated by the βarr-biased decoy D6 receptor (D6R). Combined with biochemical, cellular, and biophysical experiments, these structural snapshots allow the visualization of atypical engagement of βarrs with 7TMRs and also reveal a structural transition in the carboxyl terminus of βarr2 from a β strand to an α helix upon activation by D6R. Our study provides previously unanticipated molecular insights into the structural and functional diversity encoded in 7TMR-βarr complexes with direct implications for exploring novel therapeutic avenues. | |||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
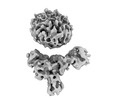
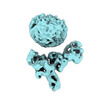
| Map data |  emd_36124.map.gz emd_36124.map.gz | 56.7 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-36124-v30.xml emd-36124-v30.xml emd-36124.xml emd-36124.xml | 22.6 KB 22.6 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_36124_fsc.xml emd_36124_fsc.xml | 8.4 KB | Display |  FSC data file FSC data file |
| Images |  emd_36124.png emd_36124.png | 64 KB | ||
| Filedesc metadata |  emd-36124.cif.gz emd-36124.cif.gz | 7 KB | ||
| Others |  emd_36124_half_map_1.map.gz emd_36124_half_map_1.map.gz emd_36124_half_map_2.map.gz emd_36124_half_map_2.map.gz | 59.3 MB 59.3 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-36124 http://ftp.pdbj.org/pub/emdb/structures/EMD-36124 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-36124 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-36124 | HTTPS FTP |
-Validation report
| Summary document |  emd_36124_validation.pdf.gz emd_36124_validation.pdf.gz | 720.3 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_36124_full_validation.pdf.gz emd_36124_full_validation.pdf.gz | 719.9 KB | Display | |
| Data in XML |  emd_36124_validation.xml.gz emd_36124_validation.xml.gz | 16.1 KB | Display | |
| Data in CIF |  emd_36124_validation.cif.gz emd_36124_validation.cif.gz | 21 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-36124 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-36124 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-36124 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-36124 | HTTPS FTP |
-Related structure data
| Related structure data |  8ja3MC  8go9C  8j8rC  8j8vC  8j8zC  8j97C  8j9kC  8jafC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_36124.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_36124.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Voxel size | X=Y=Z: 1.4268 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #2
| File | emd_36124_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #1
| File | emd_36124_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : beta-arrestin1 in complex with C3aRpp
| Entire | Name: beta-arrestin1 in complex with C3aRpp |
|---|---|
| Components |
|
-Supramolecule #1: beta-arrestin1 in complex with C3aRpp
| Supramolecule | Name: beta-arrestin1 in complex with C3aRpp / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|
-Supramolecule #2: beta-arrestin-1
| Supramolecule | Name: beta-arrestin-1 / type: complex / ID: 2 / Parent: 1 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:  |
-Supramolecule #3: Fab30 Heavy Chain
| Supramolecule | Name: Fab30 Heavy Chain / type: complex / ID: 3 / Parent: 1 / Macromolecule list: #3 |
|---|---|
| Source (natural) | Organism:  |
-Supramolecule #4: Fab30 Light Chain
| Supramolecule | Name: Fab30 Light Chain / type: complex / ID: 4 / Parent: 1 / Macromolecule list: #4 |
|---|---|
| Source (natural) | Organism:  |
-Supramolecule #5: Phosphopeptide derived from the C-terminal tail of C3aR receptor ...
| Supramolecule | Name: Phosphopeptide derived from the C-terminal tail of C3aR receptor (C3aRpp) type: complex / ID: 5 / Parent: 1 / Macromolecule list: #2 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: Beta-arrestin-1
| Macromolecule | Name: Beta-arrestin-1 / type: protein_or_peptide / ID: 1 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 40.075152 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MGDKGTRVFK KASPNGKLTV YLGKRDFVDH IDLVDPVDGV VLVDPEYLKE RRVYVTLTCA FRYGREDLDV LGLTFRKDLF VANVQSFPP APEDKKPLTR LQERLIKKLG EHAYPFTFEI PPNLPCSVTL QPGPEDTGKA CGVDYEVKAF CAENLEEKIH K RNSVRLVI ...String: MGDKGTRVFK KASPNGKLTV YLGKRDFVDH IDLVDPVDGV VLVDPEYLKE RRVYVTLTCA FRYGREDLDV LGLTFRKDLF VANVQSFPP APEDKKPLTR LQERLIKKLG EHAYPFTFEI PPNLPCSVTL QPGPEDTGKA CGVDYEVKAF CAENLEEKIH K RNSVRLVI RKVQYAPERP GPQPTAETTR QFLMSDKPLH LEASLDKEIY YHGEPISVNV HVTNNTNKTV KKIKISVRQY AD ICLFNTA QYKCPVAMEE ADDTVAPSST FCKVYTLTPF LANNREKRGL ALDGKLKHED TNLASSTLLR EGANREILGI IVS YKVKVK LVVSRGGLLG DLASSDVAVE LPFTLMHPKP K UniProtKB: Beta-arrestin-1 |
-Macromolecule #2: C3a anaphylatoxin chemotactic receptor
| Macromolecule | Name: C3a anaphylatoxin chemotactic receptor / type: protein_or_peptide / ID: 2 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 1.484165 KDa |
| Sequence | String: (SEP)EEL(TPO)R(SEP)(TPO)HC UniProtKB: C3a anaphylatoxin chemotactic receptor |
-Macromolecule #3: Fab30 heavy chain
| Macromolecule | Name: Fab30 heavy chain / type: protein_or_peptide / ID: 3 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 25.512354 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: EISEVQLVES GGGLVQPGGS LRLSCAASGF NVYSSSIHWV RQAPGKGLEW VASISSYYGY TYYADSVKGR FTISADTSKN TAYLQMNSL RAEDTAVYYC ARSRQFWYSG LDYWGQGTLV TVSSASTKGP SVFPLAPSSK STSGGTAALG CLVKDYFPEP V TVSWNSGA ...String: EISEVQLVES GGGLVQPGGS LRLSCAASGF NVYSSSIHWV RQAPGKGLEW VASISSYYGY TYYADSVKGR FTISADTSKN TAYLQMNSL RAEDTAVYYC ARSRQFWYSG LDYWGQGTLV TVSSASTKGP SVFPLAPSSK STSGGTAALG CLVKDYFPEP V TVSWNSGA LTSGVHTFPA VLQSSGLYSL SSVVTVPSSS LGTQTYICNV NHKPSNTKVD KKVEPKSCDK THHHHHHHH |
-Macromolecule #4: Fab30 light chain
| Macromolecule | Name: Fab30 light chain / type: protein_or_peptide / ID: 4 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 23.435064 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: SDIQMTQSPS SLSASVGDRV TITCRASQSV SSAVAWYQQK PGKAPKLLIY SASSLYSGVP SRFSGSRSGT DFTLTISSLQ PEDFATYYC QQYKYVPVTF GQGTKVEIKR TVAAPSVFIF PPSDSQLKSG TASVVCLLNN FYPREAKVQW KVDNALQSGN S QESVTEQD ...String: SDIQMTQSPS SLSASVGDRV TITCRASQSV SSAVAWYQQK PGKAPKLLIY SASSLYSGVP SRFSGSRSGT DFTLTISSLQ PEDFATYYC QQYKYVPVTF GQGTKVEIKR TVAAPSVFIF PPSDSQLKSG TASVVCLLNN FYPREAKVQW KVDNALQSGN S QESVTEQD SKDSTYSLSS TLTLSKADYE KHKVYACEVT HQGLSSPVTK SFNRGEC |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.4 Component:
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | TFS GLACIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Detector mode: COUNTING / Number real images: 13438 / Average electron dose: 49.4 e/Å2 |
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 2.5 µm / Nominal defocus min: 0.5 µm / Nominal magnification: 46000 |
| Sample stage | Cooling holder cryogen: NITROGEN |
 Movie
Movie Controller
Controller




























 Z
Z Y
Y X
X



















