119L
 
 | |
118L
 
 | |
120L
 
 | |
122L
 
 | |
123L
 
 | |
125L
 
 | |
126L
 
 | |
146L
 
 | |
128L
 
 | |
160L
 
 | |
173L
 
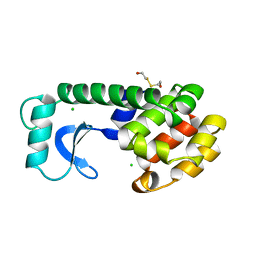
 | | PROTEIN FLEXIBILITY AND ADAPTABILITY SEEN IN 25 CRYSTAL FORMS OF T4 LYSOZYME | | 分子名称: | BETA-MERCAPTOETHANOL, T4 LYSOZYME | | 著者 | Xiong, X.-P, Zhang, X.-J, Sun, D, Matthews, B.W. | | 登録日 | 1995-03-24 | | 公開日 | 1995-07-10 | | 最終更新日 | 2024-02-07 | | 実験手法 | X-RAY DIFFRACTION (1.7 Å) | | 主引用文献 | Protein flexibility and adaptability seen in 25 crystal forms of T4 lysozyme.
J.Mol.Biol., 250, 1995
|
|
127L
 
 | |
184L
 
 | |
137L
 
 | |
140L
 
 | |
141L
 
 | |
143L
 
 | |
142L
 
 | |
145L
 
 | |
144L
 
 | |
162L
 
 | |
183L
 
 | |
197L
 
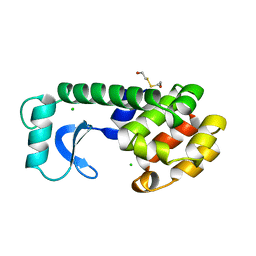
 | | THERMODYNAMIC AND STRUCTURAL COMPENSATION IN "SIZE-SWITCH" CORE-REPACKING VARIANTS OF T4 LYSOZYME | | 分子名称: | BETA-MERCAPTOETHANOL, CHLORIDE ION, LYSOZYME | | 著者 | Baldwin, E, Xu, J, Hajiseyedjavadi, O, Matthews, B.W. | | 登録日 | 1995-11-06 | | 公開日 | 1996-03-08 | | 最終更新日 | 2024-02-07 | | 実験手法 | X-RAY DIFFRACTION (2.1 Å) | | 主引用文献 | Thermodynamic and structural compensation in "size-switch" core repacking variants of bacteriophage T4 lysozyme.
J.Mol.Biol., 259, 1996
|
|
1QSQ
 
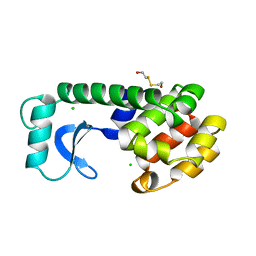
 | | CAVITY CREATING MUTATION | | 分子名称: | 2-HYDROXYETHYL DISULFIDE, CHLORIDE ION, LYSOZYME | | 著者 | Gassner, N.C, Baase, W.A, Lindstrom, J, Matthews, B.W. | | 登録日 | 1999-06-22 | | 公開日 | 1999-06-29 | | 最終更新日 | 2024-02-14 | | 実験手法 | X-RAY DIFFRACTION (1.9 Å) | | 主引用文献 | Methionine and alanine substitutions show that the formation of wild-type-like structure in the carboxy-terminal domain of T4 lysozyme is a rate-limiting step in folding.
Biochemistry, 38, 1999
|
|
1P56
 
 | |
