+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 6kpg | ||||||
|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of CB1-G protein complex | ||||||
 Components Components |
| ||||||
 Keywords Keywords | MEMBRANE PROTEIN / GPCR / G protein / cryo-EM | ||||||
| Function / homology |  Function and homology information Function and homology informationcannabinoid signaling pathway / regulation of penile erection / retrograde trans-synaptic signaling by endocannabinoid / cannabinoid receptor activity / negative regulation of mast cell activation / trans-synaptic signaling by endocannabinoid, modulating synaptic transmission / negative regulation of fatty acid beta-oxidation / negative regulation of dopamine secretion / positive regulation of acute inflammatory response to antigenic stimulus / negative regulation of serotonin secretion ...cannabinoid signaling pathway / regulation of penile erection / retrograde trans-synaptic signaling by endocannabinoid / cannabinoid receptor activity / negative regulation of mast cell activation / trans-synaptic signaling by endocannabinoid, modulating synaptic transmission / negative regulation of fatty acid beta-oxidation / negative regulation of dopamine secretion / positive regulation of acute inflammatory response to antigenic stimulus / negative regulation of serotonin secretion / regulation of feeding behavior / regulation of presynaptic cytosolic calcium ion concentration / negative regulation of action potential / Class A/1 (Rhodopsin-like receptors) / positive regulation of blood pressure / positive regulation of fever generation / regulation of metabolic process / axonal fasciculation / regulation of synaptic transmission, GABAergic / regulation of insulin secretion / G protein-coupled receptor signaling pathway, coupled to cyclic nucleotide second messenger / GABA-ergic synapse / Adenylate cyclase inhibitory pathway / positive regulation of protein localization to cell cortex / maternal process involved in female pregnancy / regulation of cAMP-mediated signaling / D2 dopamine receptor binding / G protein-coupled serotonin receptor binding / response to nutrient / regulation of mitotic spindle organization / cellular response to forskolin / regulation of synaptic transmission, glutamatergic / negative regulation of blood pressure / adenylate cyclase-inhibiting G protein-coupled receptor signaling pathway / response to cocaine / Regulation of insulin secretion / G protein-coupled receptor binding / G protein-coupled receptor activity / response to nicotine / G-protein beta/gamma-subunit complex binding / Olfactory Signaling Pathway / Activation of the phototransduction cascade / adenylate cyclase-modulating G protein-coupled receptor signaling pathway / G beta:gamma signalling through PLC beta / Presynaptic function of Kainate receptors / Thromboxane signalling through TP receptor / adenylate cyclase-activating G protein-coupled receptor signaling pathway / G protein-coupled acetylcholine receptor signaling pathway / G-protein activation / Activation of G protein gated Potassium channels / Inhibition of voltage gated Ca2+ channels via Gbeta/gamma subunits / Prostacyclin signalling through prostacyclin receptor / Glucagon signaling in metabolic regulation / G beta:gamma signalling through CDC42 / memory / response to peptide hormone / ADP signalling through P2Y purinoceptor 12 / G beta:gamma signalling through BTK / Synthesis, secretion, and inactivation of Glucagon-like Peptide-1 (GLP-1) / Sensory perception of sweet, bitter, and umami (glutamate) taste / photoreceptor disc membrane / positive regulation of neuron projection development / Adrenaline,noradrenaline inhibits insulin secretion / Glucagon-type ligand receptors / Vasopressin regulates renal water homeostasis via Aquaporins / G alpha (z) signalling events / cellular response to catecholamine stimulus / Glucagon-like Peptide-1 (GLP1) regulates insulin secretion / ADORA2B mediated anti-inflammatory cytokines production / sensory perception of taste / ADP signalling through P2Y purinoceptor 1 / adenylate cyclase-activating dopamine receptor signaling pathway / G beta:gamma signalling through PI3Kgamma / cellular response to prostaglandin E stimulus / Cooperation of PDCL (PhLP1) and TRiC/CCT in G-protein beta folding / GPER1 signaling / GDP binding / G-protein beta-subunit binding / Inactivation, recovery and regulation of the phototransduction cascade / heterotrimeric G-protein complex / G alpha (12/13) signalling events / extracellular vesicle / glucose homeostasis / actin cytoskeleton / signaling receptor complex adaptor activity / Thrombin signalling through proteinase activated receptors (PARs) / GTPase binding / retina development in camera-type eye / Ca2+ pathway / presynaptic membrane / phospholipase C-activating G protein-coupled receptor signaling pathway / cell cortex / midbody / G alpha (i) signalling events / growth cone / fibroblast proliferation / G alpha (s) signalling events / spermatogenesis / G alpha (q) signalling events / Ras protein signal transduction Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 3 Å | ||||||
 Authors Authors | Hua, T. / Li, X.T. / Wu, L.J. / Makriyannis, A. / Wang, Y.X. / Shen, L. / Liu, Z.J. | ||||||
 Citation Citation |  Journal: Cell / Year: 2020 Journal: Cell / Year: 2020Title: Activation and Signaling Mechanism Revealed by Cannabinoid Receptor-G Complex Structures. Authors: Tian Hua / Xiaoting Li / Lijie Wu / Christos Iliopoulos-Tsoutsouvas / Yuxia Wang / Meng Wu / Ling Shen / Christina A Brust / Spyros P Nikas / Feng Song / Xiyong Song / Shuguang Yuan / ...Authors: Tian Hua / Xiaoting Li / Lijie Wu / Christos Iliopoulos-Tsoutsouvas / Yuxia Wang / Meng Wu / Ling Shen / Christina A Brust / Spyros P Nikas / Feng Song / Xiyong Song / Shuguang Yuan / Qianqian Sun / Yiran Wu / Shan Jiang / Travis W Grim / Othman Benchama / Edward L Stahl / Nikolai Zvonok / Suwen Zhao / Laura M Bohn / Alexandros Makriyannis / Zhi-Jie Liu /   Abstract: Human endocannabinoid systems modulate multiple physiological processes mainly through the activation of cannabinoid receptors CB1 and CB2. Their high sequence similarity, low agonist selectivity, ...Human endocannabinoid systems modulate multiple physiological processes mainly through the activation of cannabinoid receptors CB1 and CB2. Their high sequence similarity, low agonist selectivity, and lack of activation and G protein-coupling knowledge have hindered the development of therapeutic applications. Importantly, missing structural information has significantly held back the development of promising CB2-selective agonist drugs for treating inflammatory and neuropathic pain without the psychoactivity of CB1. Here, we report the cryoelectron microscopy structures of synthetic cannabinoid-bound CB2 and CB1 in complex with G, as well as agonist-bound CB2 crystal structure. Of important scientific and therapeutic benefit, our results reveal a diverse activation and signaling mechanism, the structural basis of CB2-selective agonists design, and the unexpected interaction of cholesterol with CB1, suggestive of its endogenous allosteric modulating role. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  6kpg.cif.gz 6kpg.cif.gz | 214.2 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb6kpg.ent.gz pdb6kpg.ent.gz | 168.3 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  6kpg.json.gz 6kpg.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Summary document |  6kpg_validation.pdf.gz 6kpg_validation.pdf.gz | 923.4 KB | Display |  wwPDB validaton report wwPDB validaton report |
|---|---|---|---|---|
| Full document |  6kpg_full_validation.pdf.gz 6kpg_full_validation.pdf.gz | 936.6 KB | Display | |
| Data in XML |  6kpg_validation.xml.gz 6kpg_validation.xml.gz | 36 KB | Display | |
| Data in CIF |  6kpg_validation.cif.gz 6kpg_validation.cif.gz | 55.4 KB | Display | |
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/kp/6kpg https://data.pdbj.org/pub/pdb/validation_reports/kp/6kpg ftp://data.pdbj.org/pub/pdb/validation_reports/kp/6kpg ftp://data.pdbj.org/pub/pdb/validation_reports/kp/6kpg | HTTPS FTP |
-Related structure data
| Related structure data |  0745MC  0744C  6kpcC  6kpfC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
-Guanine nucleotide-binding protein ... , 3 types, 3 molecules ABC
| #1: Protein | Mass: 40283.836 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: GNAI1 / Production host: Homo sapiens (human) / Gene: GNAI1 / Production host:  |
|---|---|
| #2: Protein | Mass: 38045.629 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: GNB1 / Production host: Homo sapiens (human) / Gene: GNB1 / Production host:  |
| #3: Protein | Mass: 7861.143 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: GNG2 / Production host: Homo sapiens (human) / Gene: GNG2 / Production host:  |
-Protein / Antibody / Non-polymers , 3 types, 3 molecules RS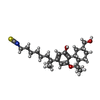

| #4: Protein | Mass: 52843.691 Da / Num. of mol.: 1 / Mutation: E273K, T283V, R340E Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: CNR1, CNR Homo sapiens (human) / Gene: CNR1, CNRProduction host: Mammalian expression vector HA-MCS-pcDNA3.1 (others) References: UniProt: P21554 |
|---|---|
| #5: Antibody | Mass: 27742.861 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Production host: Homo sapiens (human) / Production host:  |
| #6: Chemical | ChemComp-8D0 / ( |
-Details
| Has ligand of interest | Y |
|---|
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: CB1-G protein complex / Type: COMPLEX / Entity ID: #1-#5 / Source: RECOMBINANT |
|---|---|
| Molecular weight | Value: 140 kDa/nm / Experimental value: YES |
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Source (recombinant) | Organism:  |
| Buffer solution | pH: 7.5 |
| Specimen | Conc.: 1 mg/ml / Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES |
| Specimen support | Grid material: GOLD / Grid mesh size: 300 divisions/in. / Grid type: Quantifoil R1.2/1.3 |
| Vitrification | Instrument: FEI VITROBOT MARK IV / Cryogen name: ETHANE / Humidity: 100 % |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD / Nominal magnification: 130000 X / Nominal defocus max: -2000 nm / Nominal defocus min: -1000 nm / Cs: 2.7 mm / C2 aperture diameter: 50 µm / Alignment procedure: COMA FREE |
| Specimen holder | Cryogen: NITROGEN / Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Temperature (max): 70 K / Temperature (min): 70 K |
| Image recording | Electron dose: 1.333 e/Å2 / Detector mode: SUPER-RESOLUTION / Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Num. of real images: 5577 |
| EM imaging optics | Energyfilter name: GIF Quantum LS / Energyfilter slit width: 20 eV |
| Image scans | Width: 7420 / Height: 7676 / Movie frames/image: 45 / Used frames/image: 1-45 |
- Processing
Processing
| Software | Name: PHENIX / Version: 1.14_3260: / Classification: refinement | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| EM software |
| ||||||||||||||||||||||||||||||||||||
| CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION | ||||||||||||||||||||||||||||||||||||
| Particle selection | Num. of particles selected: 2186340 | ||||||||||||||||||||||||||||||||||||
| Symmetry | Point symmetry: C1 (asymmetric) | ||||||||||||||||||||||||||||||||||||
| 3D reconstruction | Resolution: 3 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 505644 / Algorithm: FOURIER SPACE / Symmetry type: POINT | ||||||||||||||||||||||||||||||||||||
| Atomic model building | B value: 72.4 / Protocol: RIGID BODY FIT / Space: REAL | ||||||||||||||||||||||||||||||||||||
| Atomic model building | PDB-ID: 6N4B | ||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller













 PDBj
PDBj































