[English] 日本語
 Yorodumi
Yorodumi- PDB-9ovt: Heteromeric GluA1/A2 in the inactive state, composite map of LBD-TMD -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 9ovt | ||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Heteromeric GluA1/A2 in the inactive state, composite map of LBD-TMD | ||||||||||||||||||||||||||||||
 Components Components |
| ||||||||||||||||||||||||||||||
 Keywords Keywords | MEMBRANE PROTEIN / GluA1A2 heterotetramer ZK iGluR | ||||||||||||||||||||||||||||||
| Function / homology |  Function and homology information Function and homology informationCargo concentration in the ER / axonal spine / positive regulation of locomotion involved in locomotory behavior / COPII-mediated vesicle transport / positive regulation of membrane potential / cellular response to ammonium ion / response to sucrose / myosin V binding / neuron spine / Trafficking of AMPA receptors ...Cargo concentration in the ER / axonal spine / positive regulation of locomotion involved in locomotory behavior / COPII-mediated vesicle transport / positive regulation of membrane potential / cellular response to ammonium ion / response to sucrose / myosin V binding / neuron spine / Trafficking of AMPA receptors / proximal dendrite / response to arsenic-containing substance / regulation of monoatomic ion transmembrane transport / cellular response to L-glutamate / ligand-gated calcium channel activity / cellular response to dsRNA / dendritic spine membrane / beta-2 adrenergic receptor binding / Synaptic adhesion-like molecules / long-term synaptic depression / cellular response to peptide hormone stimulus / spine synapse / dendritic spine neck / dendritic spine cytoplasm / dendritic spine head / cellular response to amine stimulus / peptide hormone receptor binding / response to psychosocial stress / Activation of AMPA receptors / ligand-gated monoatomic cation channel activity / response to morphine / perisynaptic space / spinal cord development / neuronal cell body membrane / protein kinase A binding / Trafficking of GluR2-containing AMPA receptors / response to lithium ion / AMPA glutamate receptor activity / AMPA glutamate receptor clustering / kainate selective glutamate receptor activity / immunoglobulin binding / adenylate cyclase binding / behavioral response to pain / AMPA glutamate receptor complex / regulation of receptor recycling / response to electrical stimulus / extracellularly glutamate-gated ion channel activity / cellular response to glycine / ionotropic glutamate receptor complex / asymmetric synapse / Unblocking of NMDA receptors, glutamate binding and activation / G-protein alpha-subunit binding / glutamate receptor binding / conditioned place preference / positive regulation of synaptic transmission / long-term memory / postsynaptic density, intracellular component / response to fungicide / regulation of synaptic transmission, glutamatergic / neuronal action potential / extracellular ligand-gated monoatomic ion channel activity / cytoskeletal protein binding / glutamate-gated receptor activity / cellular response to brain-derived neurotrophic factor stimulus / regulation of long-term synaptic depression / somatodendritic compartment / glutamate-gated calcium ion channel activity / presynaptic active zone membrane / synapse assembly / excitatory synapse / ionotropic glutamate receptor signaling pathway / ionotropic glutamate receptor binding / dendrite membrane / dendrite cytoplasm / ligand-gated monoatomic ion channel activity involved in regulation of presynaptic membrane potential / positive regulation of excitatory postsynaptic potential / dendritic shaft / SNARE binding / synaptic membrane / PDZ domain binding / response to cocaine / neuromuscular junction / establishment of protein localization / protein tetramerization / cellular response to amino acid stimulus / transmitter-gated monoatomic ion channel activity involved in regulation of postsynaptic membrane potential / synaptic transmission, glutamatergic / response to nutrient levels / cerebral cortex development / receptor internalization / regulation of synaptic plasticity / recycling endosome / postsynaptic density membrane / cellular response to growth factor stimulus / response to peptide hormone / modulation of chemical synaptic transmission / Schaffer collateral - CA1 synapse / response to toxic substance / recycling endosome membrane / small GTPase binding Similarity search - Function | ||||||||||||||||||||||||||||||
| Biological species |  | ||||||||||||||||||||||||||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 3.43 Å | ||||||||||||||||||||||||||||||
 Authors Authors | Yen, L.Y. / Sobolevsky, A.I. / Newton, T.P. / Gangwar, S.P. | ||||||||||||||||||||||||||||||
| Funding support |  United States, 9items United States, 9items
| ||||||||||||||||||||||||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2026 Journal: Nat Commun / Year: 2026Title: Auxiliary subunits reshape structural asymmetry and functional plasticity in heterotetrameric GluA1/A2 AMPA receptor core. Authors: Laura Y Yen / Thomas P Newton / Maria V Yelshanskaya / Muhammed Aktolun / Shanti Pal Gangwar / Rasmus P Clausen / Maria G Kurnikova / Alexander I Sobolevsky /   Abstract: AMPA-subtype ionotropic glutamate receptors (AMPARs) mediate the fast component of excitatory neurotransmission. They govern synaptic plasticity that underlies learning and memory, while their ...AMPA-subtype ionotropic glutamate receptors (AMPARs) mediate the fast component of excitatory neurotransmission. They govern synaptic plasticity that underlies learning and memory, while their dysregulation is implicated in numerous neurological disorders. The functional diversity of AMPARs arises from variations in their subunit composition and also their association with auxiliary subunits. While multiple structures of homomeric AMPARs have been reported, structural information for the heteromeric core - particularly in the absence of auxiliary subunits, which would serve as a functional and structural baseline - has been limited. Here, we report cryo-electron microscopy structures of GluA1/A2, the most abundant AMPAR di-heteromer in the brain, in the closed, open, and desensitized states. Using molecular dynamics (MD) simulations and cross-correlating structural and functional information, we find that auxiliary subunits increase the diameter of channel pore, which corresponds to larger conductance. Likewise, we find that recovery from desensitization slows with greater disruption of two-fold rotational symmetry of the ligand-binding domain dimer in the desensitized state. Both receptor activation and desensitization vary with the type and number of associated auxiliary proteins. These structures offer a foundation for uncovering how auxiliary subunits reshape structural asymmetry and functional plasticity in heterotetrameric AMPARs. | ||||||||||||||||||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  9ovt.cif.gz 9ovt.cif.gz | 299.1 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb9ovt.ent.gz pdb9ovt.ent.gz | 237.9 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  9ovt.json.gz 9ovt.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/ov/9ovt https://data.pdbj.org/pub/pdb/validation_reports/ov/9ovt ftp://data.pdbj.org/pub/pdb/validation_reports/ov/9ovt ftp://data.pdbj.org/pub/pdb/validation_reports/ov/9ovt | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  70912MC  9ovuC  9ovvC  9ovwC C: citing same article ( M: map data used to model this data |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
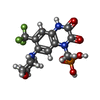
| #1: Protein | Mass: 48183.172 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Homo sapiens (human) / Tissue (production host): kidney / References: UniProt: P19490 Homo sapiens (human) / Tissue (production host): kidney / References: UniProt: P19490#2: Protein | Mass: 47751.934 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Homo sapiens (human) / Tissue (production host): kidney / References: UniProt: P19491 Homo sapiens (human) / Tissue (production host): kidney / References: UniProt: P19491#3: Chemical | ChemComp-ZK1 / {[ #4: Chemical | #5: Water | ChemComp-HOH / | Has ligand of interest | Y | Has protein modification | Y | |
|---|
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: Heteromeric GluA1A2 with competitive antagonist ZK / Type: COMPLEX / Entity ID: #1-#2 / Source: RECOMBINANT | |||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Molecular weight | Value: 0.556 MDa / Experimental value: NO | |||||||||||||||||||||||||
| Source (natural) | Organism:  | |||||||||||||||||||||||||
| Source (recombinant) | Organism:  Homo sapiens (human) / Strain: HEK 293S GntI- / Plasmid: pEG BacMam Homo sapiens (human) / Strain: HEK 293S GntI- / Plasmid: pEG BacMam | |||||||||||||||||||||||||
| Buffer solution | pH: 8 Details: 150 mM NaCl, 20 mM Tris-HCl pH 8.0, and 0.05% digitonin, 100 uM ZK-200775 | |||||||||||||||||||||||||
| Buffer component |
| |||||||||||||||||||||||||
| Specimen | Conc.: 4.5 mg/ml / Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES Details: The sample had compositional heterogeneity, with broken particles seen throughout. Otherwise, sample was monodisperse | |||||||||||||||||||||||||
| Specimen support | Details: 15 mA / Grid material: GOLD / Grid mesh size: 300 divisions/in. / Grid type: UltrAuFoil R1.2/1.3 | |||||||||||||||||||||||||
| Vitrification | Instrument: FEI VITROBOT MARK IV / Cryogen name: ETHANE / Humidity: 100 % / Chamber temperature: 298 K |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: TFS KRIOS |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: SPOT SCAN FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: SPOT SCAN |
| Electron lens | Mode: BRIGHT FIELD / Nominal defocus max: 2500 nm / Nominal defocus min: 1000 nm / Cs: 2.7 mm / C2 aperture diameter: 100 µm |
| Image recording | Electron dose: 45.7 e/Å2 / Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) |
- Processing
Processing
| EM software |
| ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION | ||||||||||||||||||||||||||||
| Particle selection | Num. of particles selected: 5527903 | ||||||||||||||||||||||||||||
| Symmetry | Point symmetry: C2 (2 fold cyclic) | ||||||||||||||||||||||||||||
| 3D reconstruction | Resolution: 3.43 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 216104 Details: this is a composite map; where required, consensus refinement parameters are entered Symmetry type: POINT | ||||||||||||||||||||||||||||
| Refinement | Stereochemistry target values: REAL-SPACE (WEIGHTED MAP SUM AT ATOM CENTERS) | ||||||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller



















 PDBj
PDBj