[English] 日本語
 Yorodumi
Yorodumi- EMDB-70919: Composite map of GluA1/A2 in the activated state, in complex with... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | ||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Composite map of GluA1/A2 in the activated state, in complex with positive allosteric modulator (R,R)-2b and agonist glutamate (ATD-LBD-TMD) | ||||||||||||||||||||||||||||||
 Map data Map data | A1A2 rr2b glu composite map (LBD-TMD) | ||||||||||||||||||||||||||||||
 Sample Sample |
| ||||||||||||||||||||||||||||||
 Keywords Keywords | GluA1A2 heterotetramer active iGluR / MEMBRANE PROTEIN | ||||||||||||||||||||||||||||||
| Function / homology |  Function and homology information Function and homology informationCargo concentration in the ER / axonal spine / positive regulation of locomotion involved in locomotory behavior / COPII-mediated vesicle transport / positive regulation of membrane potential / cellular response to ammonium ion / response to sucrose / myosin V binding / neuron spine / Trafficking of AMPA receptors ...Cargo concentration in the ER / axonal spine / positive regulation of locomotion involved in locomotory behavior / COPII-mediated vesicle transport / positive regulation of membrane potential / cellular response to ammonium ion / response to sucrose / myosin V binding / neuron spine / Trafficking of AMPA receptors / proximal dendrite / response to arsenic-containing substance / regulation of monoatomic ion transmembrane transport / cellular response to L-glutamate / ligand-gated calcium channel activity / cellular response to dsRNA / dendritic spine membrane / beta-2 adrenergic receptor binding / Synaptic adhesion-like molecules / long-term synaptic depression / cellular response to peptide hormone stimulus / spine synapse / dendritic spine neck / dendritic spine cytoplasm / dendritic spine head / cellular response to amine stimulus / peptide hormone receptor binding / response to psychosocial stress / Activation of AMPA receptors / ligand-gated monoatomic cation channel activity / response to morphine / perisynaptic space / spinal cord development / neuronal cell body membrane / protein kinase A binding / Trafficking of GluR2-containing AMPA receptors / response to lithium ion / AMPA glutamate receptor activity / AMPA glutamate receptor clustering / kainate selective glutamate receptor activity / immunoglobulin binding / adenylate cyclase binding / behavioral response to pain / AMPA glutamate receptor complex / regulation of receptor recycling / response to electrical stimulus / extracellularly glutamate-gated ion channel activity / cellular response to glycine / ionotropic glutamate receptor complex / asymmetric synapse / Unblocking of NMDA receptors, glutamate binding and activation / G-protein alpha-subunit binding / conditioned place preference / glutamate receptor binding / positive regulation of synaptic transmission / long-term memory / postsynaptic density, intracellular component / regulation of synaptic transmission, glutamatergic / response to fungicide / neuronal action potential / extracellular ligand-gated monoatomic ion channel activity / cytoskeletal protein binding / glutamate-gated receptor activity / cellular response to brain-derived neurotrophic factor stimulus / regulation of long-term synaptic depression / somatodendritic compartment / glutamate-gated calcium ion channel activity / synapse assembly / presynaptic active zone membrane / excitatory synapse / ionotropic glutamate receptor signaling pathway / ionotropic glutamate receptor binding / dendrite membrane / dendrite cytoplasm / ligand-gated monoatomic ion channel activity involved in regulation of presynaptic membrane potential / positive regulation of excitatory postsynaptic potential / dendritic shaft / SNARE binding / synaptic membrane / PDZ domain binding / response to cocaine / neuromuscular junction / establishment of protein localization / protein tetramerization / cellular response to amino acid stimulus / transmitter-gated monoatomic ion channel activity involved in regulation of postsynaptic membrane potential / synaptic transmission, glutamatergic / response to nutrient levels / cerebral cortex development / regulation of synaptic plasticity / receptor internalization / recycling endosome / postsynaptic density membrane / response to peptide hormone / cellular response to growth factor stimulus / modulation of chemical synaptic transmission / Schaffer collateral - CA1 synapse / response to toxic substance / recycling endosome membrane / small GTPase binding Similarity search - Function | ||||||||||||||||||||||||||||||
| Biological species |  | ||||||||||||||||||||||||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.2 Å | ||||||||||||||||||||||||||||||
 Authors Authors | Yen LY / Sobolevsky AI / Newton TP / Gangwar SP | ||||||||||||||||||||||||||||||
| Funding support |  United States, 9 items United States, 9 items
| ||||||||||||||||||||||||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2026 Journal: Nat Commun / Year: 2026Title: Auxiliary subunits reshape structural asymmetry and functional plasticity in heterotetrameric GluA1/A2 AMPA receptor core. Authors: Laura Y Yen / Thomas P Newton / Maria V Yelshanskaya / Muhammed Aktolun / Shanti Pal Gangwar / Rasmus P Clausen / Maria G Kurnikova / Alexander I Sobolevsky /   Abstract: AMPA-subtype ionotropic glutamate receptors (AMPARs) mediate the fast component of excitatory neurotransmission. They govern synaptic plasticity that underlies learning and memory, while their ...AMPA-subtype ionotropic glutamate receptors (AMPARs) mediate the fast component of excitatory neurotransmission. They govern synaptic plasticity that underlies learning and memory, while their dysregulation is implicated in numerous neurological disorders. The functional diversity of AMPARs arises from variations in their subunit composition and also their association with auxiliary subunits. While multiple structures of homomeric AMPARs have been reported, structural information for the heteromeric core - particularly in the absence of auxiliary subunits, which would serve as a functional and structural baseline - has been limited. Here, we report cryo-electron microscopy structures of GluA1/A2, the most abundant AMPAR di-heteromer in the brain, in the closed, open, and desensitized states. Using molecular dynamics (MD) simulations and cross-correlating structural and functional information, we find that auxiliary subunits increase the diameter of channel pore, which corresponds to larger conductance. Likewise, we find that recovery from desensitization slows with greater disruption of two-fold rotational symmetry of the ligand-binding domain dimer in the desensitized state. Both receptor activation and desensitization vary with the type and number of associated auxiliary proteins. These structures offer a foundation for uncovering how auxiliary subunits reshape structural asymmetry and functional plasticity in heterotetrameric AMPARs. | ||||||||||||||||||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_70919.map.gz emd_70919.map.gz | 237.6 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-70919-v30.xml emd-70919-v30.xml emd-70919.xml emd-70919.xml | 22.7 KB 22.7 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_70919.png emd_70919.png | 1.4 MB | ||
| Filedesc metadata |  emd-70919.cif.gz emd-70919.cif.gz | 7.7 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-70919 http://ftp.pdbj.org/pub/emdb/structures/EMD-70919 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-70919 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-70919 | HTTPS FTP |
-Related structure data
| Related structure data |  9ovuMC  9ovtC  9ovvC  9ovwC C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_70919.map.gz / Format: CCP4 / Size: 274.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_70919.map.gz / Format: CCP4 / Size: 274.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | A1A2 rr2b glu composite map (LBD-TMD) | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.83 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
- Sample components
Sample components
-Entire : Heteromeric GluA1A2 with positive allosteric modulator (R,R)-2b a...
| Entire | Name: Heteromeric GluA1A2 with positive allosteric modulator (R,R)-2b and agonist glutamate (Glu) |
|---|---|
| Components |
|
-Supramolecule #1: Heteromeric GluA1A2 with positive allosteric modulator (R,R)-2b a...
| Supramolecule | Name: Heteromeric GluA1A2 with positive allosteric modulator (R,R)-2b and agonist glutamate (Glu) type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#2 |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 556 KDa |
-Macromolecule #1: Isoform Flip of Glutamate receptor 1
| Macromolecule | Name: Isoform Flip of Glutamate receptor 1 / type: protein_or_peptide / ID: 1 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 94.483922 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MPYIFAFFCT GFLGAVVGAN FPNNIQIGGL FPNQQSQEHA AFRFALSQLT EPPKLLPQID IVNISDSFEM TYRFCSQFSK GVYAIFGFY ERRTVNMLTS FCGALHVCFI TPSFPVDTSN QFVLQLRPEL QEALISIIDH YKWQTFVYIY DADRGLSVLQ R VLDTAAEK ...String: MPYIFAFFCT GFLGAVVGAN FPNNIQIGGL FPNQQSQEHA AFRFALSQLT EPPKLLPQID IVNISDSFEM TYRFCSQFSK GVYAIFGFY ERRTVNMLTS FCGALHVCFI TPSFPVDTSN QFVLQLRPEL QEALISIIDH YKWQTFVYIY DADRGLSVLQ R VLDTAAEK NWQVTAVNIL TTTEEGYRML FQDLEKKKER LVVVDCESER LNAILGQIVK LEKNGIGYHY ILANLGFMDI DL NKFKESG ANVTGFQLVN YTDTIPARIM QQWRTSDSRD HTRVDWKRPK YTSALTYDGV KVMAEAFQSL RRQRIDISRR GNA GDCLAN PAVPWGQGID IQRALQQVRF EGLTGNVQFN EKGRRTNYTL HVIEMKHDGI RKIGYWNEDD KFVPAGGDNS SVQN RTYIV TTILEDPYVM LKKNANQFEG NDRYEGYCVE LAAEIAKHVG YSYRLEIVSD GKYGARDPDT KAWNGMVGEL VYGRA DVAV APLTITLVRE EVIDFSKPFM SLGISIMIKK PQKSKPGVFS FLDPLAYEIW MCIVFAYIGV SVVLFLVSRF SPYEWH SEE FEEGRDQTTS DQSNEFGIFN SLWFSLGAFM QQGCDISPRS LSGRIVGGVW WFFTLIIISS YTANLAAFLT VERMVSP IE SAEDLAKQTE IAYGTLEAGS TKEFFRRSKI AVFEKMWTYM KSAEPSVFVR TTEEGMIRVR KSKGKYAYLL ESTMNEYI E QRKPCDTMKV GGNLDSKGYG IATPKGSALR GPVNLAVLKL SEQGVLDKLK SKWWYDKGEC GSKDSGSKDK TSALSLSNV AGVFYILIGG LGLAMLVALI EFCYKSRSES KRMKG UniProtKB: Glutamate receptor 1 |
-Macromolecule #2: Isoform Flip of Glutamate receptor 2
| Macromolecule | Name: Isoform Flip of Glutamate receptor 2 / type: protein_or_peptide / ID: 2 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 94.369961 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MGKIMHISVL LSPVLWGLIF GVSSNSIQIG GLFPRGADQE YSAFRVGMVQ FSTSEFRLTP HIDNLEVANS FAVTNAFCSQ FSRGVYAIF GFYDKKSVNT ITSFCGTLHV SFITPSFPTD GTHPFVIQMR PDLKGALLSL IEYYQWDKFA YLYDSDRGLS T LQAVLDSA ...String: MGKIMHISVL LSPVLWGLIF GVSSNSIQIG GLFPRGADQE YSAFRVGMVQ FSTSEFRLTP HIDNLEVANS FAVTNAFCSQ FSRGVYAIF GFYDKKSVNT ITSFCGTLHV SFITPSFPTD GTHPFVIQMR PDLKGALLSL IEYYQWDKFA YLYDSDRGLS T LQAVLDSA AEKKWQVTAI NVGNINNDKK DETYRSLFQD LELKKERRVI LDCERDKVND IVDQVITIGK HVKGYHYIIA NL GFTDGDL LKIQFGGAEV SGFQIVDYDD SLVSKFIERW STLEEKEYPG AHTATIKYTS ALTYDAVQVM TEAFRNLRKQ RIE ISRRGN AGDCLANPAV PWGQGVEIER ALKQVQVEGL SGNIKFDQNG KRINYTINIM ELKTNGPRKI GYWSEVDKMV LTED DTSGL EQKTVVVTTI LESPYVMMKK NHEMLEGNER YEGYCVDLAA EIAKHCGFKY KLTIVGDGKY GARDADTKIW NGMVG ELVY GKADIAIAPL TITLVREEVI DFSKPFMSLG ISIMIKKPQK SKPGVFSFLD PLAYEIWMCI VFAYIGVSVV LFLVSR FSP YEWHTEEFED GRETQSSEST NEFGIFNSLW FSLGAFMRQG CDISPRSLSG RIVGGVWWFF TLIIISSYTA NLAAFLT VE RMVSPIESAE DLSKQTEIAY GTLDSGSTKE FFRRSKIAVF DKMWTYMRSA EPSVFVRTTA EGVARVRKSK GKYAYLLE S TMNEYIEQRK PCDTMKVGGN LDSKGYGIAT PKGSSLGTPV NLAVLKLSEQ GVLDKLKNKW WYDKGECGAK DSGSKEKTS ALSLSNVAGV FYILVGGLGL AMLVALIEFC YKSRAEAKRM KG UniProtKB: Glutamate receptor 2 |
-Macromolecule #5: GLUTAMIC ACID
| Macromolecule | Name: GLUTAMIC ACID / type: ligand / ID: 5 / Number of copies: 4 / Formula: GLU |
|---|---|
| Molecular weight | Theoretical: 147.129 Da |
| Chemical component information |  ChemComp-GLU: |
-Macromolecule #6: SODIUM ION
| Macromolecule | Name: SODIUM ION / type: ligand / ID: 6 / Number of copies: 1 |
|---|---|
| Molecular weight | Theoretical: 22.99 Da |
-Macromolecule #7: N,N'-[biphenyl-4,4'-diyldi(2R)propane-2,1-diyl]dipropane-2-sulfonamide
| Macromolecule | Name: N,N'-[biphenyl-4,4'-diyldi(2R)propane-2,1-diyl]dipropane-2-sulfonamide type: ligand / ID: 7 / Number of copies: 2 / Formula: FWF |
|---|---|
| Molecular weight | Theoretical: 480.684 Da |
| Chemical component information | 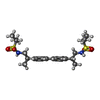 ChemComp-FWF: |
-Macromolecule #8: water
| Macromolecule | Name: water / type: ligand / ID: 8 / Number of copies: 4 / Formula: HOH |
|---|---|
| Molecular weight | Theoretical: 18.015 Da |
| Chemical component information |  ChemComp-HOH: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 4.5 mg/mL | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 8 Component:
Details: 150 mM NaCl, 20 mM Tris-HCl pH 8.0, and 0.05% digitonin, 500 uM (R,R)-2b, 1 mM glutamate | ||||||||||||||||||
| Grid | Model: UltrAuFoil R1.2/1.3 / Material: GOLD / Mesh: 300 / Support film - Material: GOLD / Support film - topology: HOLEY / Support film - Film thickness: 500 / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 25 sec. / Pretreatment - Atmosphere: AIR / Details: 15 mA | ||||||||||||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 298 K / Instrument: FEI VITROBOT MARK IV | ||||||||||||||||||
| Details | The sample had compositional heterogeneity, with broken particles seen throughout. Otherwise, sample was monodisperse |
- Electron microscopy
Electron microscopy
| Microscope | TFS KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Average electron dose: 47.03 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 100.0 µm / Illumination mode: SPOT SCAN / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 2.5 µm / Nominal defocus min: 1.0 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller

























 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)




















