[English] 日本語
 Yorodumi
Yorodumi- PDB-7new: Fucosylated heterochiral linear peptide Fdln69 bound to the fucos... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 7new | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Fucosylated heterochiral linear peptide Fdln69 bound to the fucose binding lectin LecB PA-IIL from Pseudomonas aeruginosa at 2.0 Angstrom resolution | |||||||||
 Components Components |
| |||||||||
 Keywords Keywords | ANTIBIOTIC / Antimicrobial peptide / Lectin / ANTIMICROBIAL PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationsingle-species biofilm formation / carbohydrate binding / metal ion binding Similarity search - Function | |||||||||
| Biological species |  synthetic construct (others) | |||||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / MOLECULAR REPLACEMENT /  molecular replacement / Resolution: 2.02 Å molecular replacement / Resolution: 2.02 Å | |||||||||
 Authors Authors | Personne, H. / Baeriswyl, S. / Stocker, A. / Reymond, J.-L. | |||||||||
| Funding support |  Switzerland, 1items Switzerland, 1items
| |||||||||
 Citation Citation |  Journal: Rsc Chem Biol / Year: 2021 Journal: Rsc Chem Biol / Year: 2021Title: A mixed chirality alpha-helix in a stapled bicyclic and a linear antimicrobial peptide revealed by X-ray crystallography. Authors: Baeriswyl, S. / Personne, H. / Di Bonaventura, I. / Kohler, T. / van Delden, C. / Stocker, A. / Javor, S. / Reymond, J.L. #1:  Journal: Rsc Chem Biol / Year: 2021 Journal: Rsc Chem Biol / Year: 2021Title: A mixed chirality alpha-helix in a stapled bicyclic and a linear antimicrobial peptide revealed by X-ray crystallography. Authors: Baeriswyl, S. / Personne, H. / Di Bonaventura, I. / Kohler, T. / van Delden, C. / Stocker, A. / Javor, S. / Reymond, J.L. #2:  Journal: Rsc Chem Biol / Year: 2021 Journal: Rsc Chem Biol / Year: 2021Title: A mixed chirality alpha-helix in a stapled bicyclic and a linear antimicrobial peptide revealed by X-ray crystallography. Authors: Baeriswyl, S. / Personne, H. / Di Bonaventura, I. / Kohler, T. / van Delden, C. / Stocker, A. / Javor, S. / Reymond, J.L. #3:  Journal: Chemrxiv / Year: 2021 Journal: Chemrxiv / Year: 2021Title: Mixed chirality alpha-helix in a stapled bicyclic and a linear antimicrobial peptide revealed by X-ray crystallography Authors: Personne, H. / Baeriswyl, S. / Di Bonaventura, I. / Kohler, T. / van Delden, C. / Stocker, A. / Javor, S. / Reymond, J.-L. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  7new.cif.gz 7new.cif.gz | 195.5 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb7new.ent.gz pdb7new.ent.gz | 154.8 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  7new.json.gz 7new.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/ne/7new https://data.pdbj.org/pub/pdb/validation_reports/ne/7new ftp://data.pdbj.org/pub/pdb/validation_reports/ne/7new ftp://data.pdbj.org/pub/pdb/validation_reports/ne/7new | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  6y0uC  6y0vC  6y13C  6y14C  6y1sC  7nefC  1oxcS S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||
| 2 | 
| ||||||||
| Unit cell |
|
- Components
Components
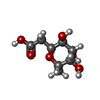
| #1: Protein | Mass: 11865.905 Da / Num. of mol.: 4 Source method: isolated from a genetically manipulated source Details: Fucose-binding Lectin LecB PA-IIL from Pseudomonas aeruginosa Source: (gene. exp.)  Gene: lecB, C0044_25260, CAZ10_21840, DT376_00595, DY979_15445, ECC04_10105, EFK27_13700, EGV95_09240, EGY23_15550, IPC669_23070, PA5486_01888, PAERUG_E15_London_28_01_14_00983, PAMH19_1713, RW109_RW109_02453 Production host:  #2: Polypeptide(D) | Mass: 1324.847 Da / Num. of mol.: 4 / Source method: obtained synthetically / Details: Fucosylated heterochiral linear peptide Fdln69 / Source: (synth.) synthetic construct (others) #3: Chemical | ChemComp-CA / #4: Sugar | ChemComp-ZDC / #5: Water | ChemComp-HOH / | Has ligand of interest | N | Has protein modification | Y | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.81 Å3/Da / Density % sol: 56.17 % |
|---|---|
| Crystal grow | Temperature: 291 K / Method: vapor diffusion, sitting drop / pH: 6.5 Details: 0.2M Sodium citrate tribasic dihydrate, 0.1M Sodium cacodylate trihydrate pH 6.5, 30% v/v 2-Propanol |
-Data collection
| Diffraction | Mean temperature: 100 K / Serial crystal experiment: N |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  SLS SLS  / Beamline: X06DA / Wavelength: 0.976437 Å / Beamline: X06DA / Wavelength: 0.976437 Å |
| Detector | Type: DECTRIS PILATUS 2M-F / Detector: PIXEL / Date: Nov 2, 2020 |
| Radiation | Protocol: MAD / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.976437 Å / Relative weight: 1 |
| Reflection | Resolution: 2.017→48.288 Å / Num. obs: 71291 / % possible obs: 97.5 % / Redundancy: 3.55 % / CC1/2: 0.995 / Rrim(I) all: 0.167 / Net I/σ(I): 6.89 |
| Reflection shell | Resolution: 2.02→2.14 Å / Redundancy: 3.48 % / Mean I/σ(I) obs: 1.01 / Num. unique obs: 11367 / CC1/2: 0.512 / % possible all: 96.1 |
-Phasing
| Phasing | Method:  molecular replacement molecular replacement |
|---|
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 1OXC Resolution: 2.02→40.03 Å / SU ML: 0.28 / Cross valid method: THROUGHOUT / σ(F): 1.32 / Phase error: 27.17 / Stereochemistry target values: ML
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å / Solvent model: FLAT BULK SOLVENT MODEL | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso max: 161.23 Å2 / Biso mean: 43.0425 Å2 / Biso min: 25.46 Å2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: final / Resolution: 2.02→40.03 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Refine-ID: X-RAY DIFFRACTION / Rfactor Rfree error: 0 / Total num. of bins used: 25
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS params. | Method: refined / Origin x: 26.2953 Å / Origin y: 8.6788 Å / Origin z: 17.1692 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS group |
|
 Movie
Movie Controller
Controller












 PDBj
PDBj