[English] 日本語
 Yorodumi
Yorodumi- PDB-7aup: Yeast Diphosphoinositol Polyphosphate Phosphohydrolase DDP1 in co... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 7aup | ||||||
|---|---|---|---|---|---|---|---|
| Title | Yeast Diphosphoinositol Polyphosphate Phosphohydrolase DDP1 in complex with PCP-InsP7 | ||||||
 Components Components | Diphosphoinositol polyphosphate phosphohydrolase DDP1 | ||||||
 Keywords Keywords | HYDROLASE / Inositol / PP-InsP / Pyrophosphatase / Polyphosphate / Diadenosine polyphosphate / DDP1 / Nudix | ||||||
| Function / homology |  Function and homology information Function and homology informationdiadenosine hexaphosphate hydrolase (AMP-forming) / Synthesis of pyrophosphates in the cytosol / polyphosphate catabolic process / endopolyphosphatase / diadenosine polyphosphate catabolic process / bis(5'-adenosyl)-hexaphosphatase activity / diadenosine pentaphosphate catabolic process / diadenosine hexaphosphate catabolic process / adenosine 5'-(hexahydrogen pentaphosphate) catabolic process / endopolyphosphatase activity ...diadenosine hexaphosphate hydrolase (AMP-forming) / Synthesis of pyrophosphates in the cytosol / polyphosphate catabolic process / endopolyphosphatase / diadenosine polyphosphate catabolic process / bis(5'-adenosyl)-hexaphosphatase activity / diadenosine pentaphosphate catabolic process / diadenosine hexaphosphate catabolic process / adenosine 5'-(hexahydrogen pentaphosphate) catabolic process / endopolyphosphatase activity / diphosphoinositol polyphosphate metabolic process / diphosphoinositol-polyphosphate diphosphatase activity / phosphodiesterase decapping endonuclease activity / bis(5'-adenosyl)-pentaphosphatase activity / diphosphoinositol-polyphosphate diphosphatase / metal ion binding / nucleus / cytoplasm Similarity search - Function | ||||||
| Biological species |  | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  FOURIER SYNTHESIS / Resolution: 1.85 Å FOURIER SYNTHESIS / Resolution: 1.85 Å | ||||||
 Authors Authors | Marquez-Monino, M.A. / Gonzalez, B. | ||||||
| Funding support |  Spain, 1items Spain, 1items
| ||||||
 Citation Citation |  Journal: Sci Adv / Year: 2021 Journal: Sci Adv / Year: 2021Title: Multiple substrate recognition by yeast diadenosine and diphosphoinositol polyphosphate phosphohydrolase through phosphate clamping. Authors: Marquez-Monino, M.A. / Ortega-Garcia, R. / Shipton, M.L. / Franco-Echevarria, E. / Riley, A.M. / Sanz-Aparicio, J. / Potter, B.V.L. / Gonzalez, B. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  7aup.cif.gz 7aup.cif.gz | 53.6 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb7aup.ent.gz pdb7aup.ent.gz | 36.9 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  7aup.json.gz 7aup.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/au/7aup https://data.pdbj.org/pub/pdb/validation_reports/au/7aup ftp://data.pdbj.org/pub/pdb/validation_reports/au/7aup ftp://data.pdbj.org/pub/pdb/validation_reports/au/7aup | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  7auiC  7aujC  7aukC  7aulC  7aumC  7aunSC  7auoC 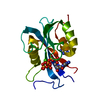 7auqC  7aurC  7ausC  7autC  7auuC C: citing same article ( S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| |||||||||
| Unit cell |
| |||||||||
| Components on special symmetry positions |
|
- Components
Components
| #1: Protein | Mass: 21812.713 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Details: GGS is a rest of purification linker, protein starts in MGK. Source: (gene. exp.)  Strain: ATCC 204508 / S288c / Gene: DDP1, YOR163W, O3575 / Plasmid: pKLSLt / Production host:  References: UniProt: Q99321, diphosphoinositol-polyphosphate diphosphatase, diadenosine hexaphosphate hydrolase (AMP-forming) | ||||
|---|---|---|---|---|---|
| #2: Chemical | ChemComp-VEB / [ | ||||
| #3: Chemical | | #4: Water | ChemComp-HOH / | Has ligand of interest | Y | |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.42 Å3/Da / Density % sol: 49.18 % / Description: Bypiramid |
|---|---|
| Crystal grow | Temperature: 291 K / Method: vapor diffusion, sitting drop / pH: 4.5 Details: 20% PEG 3350, 0.1 M Sodium acetate pH 4.5, 0.1 M NaCl. Protein buffer: 20 mM Tris pH 8.0, 150 mM NaCl, 1 mM DTT, 10 mM PCP-InsP7. |
-Data collection
| Diffraction | Mean temperature: 100 K / Serial crystal experiment: N |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  ALBA ALBA  / Beamline: XALOC / Wavelength: 0.97926 Å / Beamline: XALOC / Wavelength: 0.97926 Å |
| Detector | Type: DECTRIS PILATUS 6M / Detector: PIXEL / Date: Apr 27, 2019 / Details: KB focusing mirrors |
| Radiation | Monochromator: Si(111) channel-cut, cryocooled / Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.97926 Å / Relative weight: 1 |
| Reflection | Resolution: 1.85→46.72 Å / Num. obs: 18625 / % possible obs: 100 % / Redundancy: 18.6 % / Biso Wilson estimate: 32.47 Å2 / CC1/2: 1 / Rmerge(I) obs: 0.052 / Rpim(I) all: 0.013 / Net I/σ(I): 33.2 |
| Reflection shell | Resolution: 1.85→1.89 Å / Redundancy: 17.9 % / Rmerge(I) obs: 0.652 / Mean I/σ(I) obs: 6.2 / Num. unique obs: 1141 / CC1/2: 0.915 / Rpim(I) all: 0.16 / % possible all: 100 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  FOURIER SYNTHESIS FOURIER SYNTHESISStarting model: 7AUN Resolution: 1.85→46.72 Å / Cor.coef. Fo:Fc: 0.956 / Cor.coef. Fo:Fc free: 0.933 / SU B: 3.883 / SU ML: 0.115 / Cross valid method: THROUGHOUT / σ(F): 0 / ESU R: 0.148 / ESU R Free: 0.145 / Stereochemistry target values: MAXIMUM LIKELIHOOD Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS U VALUES : REFINED INDIVIDUALLY
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.2 Å / Solvent model: MASK | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso max: 146.54 Å2 / Biso mean: 46.945 Å2 / Biso min: 24.36 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: final / Resolution: 1.85→46.72 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 1.85→1.898 Å / Rfactor Rfree error: 0 / Total num. of bins used: 20
|
 Movie
Movie Controller
Controller












 PDBj
PDBj