[English] 日本語
 Yorodumi
Yorodumi- PDB-6z32: Human cation-independent mannose 6-phosphate/IGF2 receptor domain... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 6z32 | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Human cation-independent mannose 6-phosphate/IGF2 receptor domains 7-11 | |||||||||||||||
 Components Components | Cation-independent mannose-6-phosphate receptor | |||||||||||||||
 Keywords Keywords | SUGAR BINDING PROTEIN / Cation-independent mannose 6-phosphate receptor / Insulin-like growth factor 2 receptor / Mannose 6-phosphate | |||||||||||||||
| Function / homology |  Function and homology information Function and homology informationRetrograde transport at the Trans-Golgi-Network / response to tetrachloromethane / insulin-like growth factor receptor activity / clathrin coat / retromer complex binding / insulin-like growth factor binding / insulin-like growth factor II binding / trans-Golgi network transport vesicle / host-mediated activation of viral process / retinoic acid binding ...Retrograde transport at the Trans-Golgi-Network / response to tetrachloromethane / insulin-like growth factor receptor activity / clathrin coat / retromer complex binding / insulin-like growth factor binding / insulin-like growth factor II binding / trans-Golgi network transport vesicle / host-mediated activation of viral process / retinoic acid binding / lysosomal transport / Golgi Associated Vesicle Biogenesis / nuclear envelope lumen / D-mannose binding / animal organ regeneration / response to retinoic acid / endocytic vesicle / G-protein alpha-subunit binding / transport vesicle / receptor-mediated endocytosis / secretory granule membrane / trans-Golgi network membrane / post-embryonic development / trans-Golgi network / liver development / clathrin-coated endocytic vesicle membrane / phosphoprotein binding / late endosome / Cargo recognition for clathrin-mediated endocytosis / Clathrin-mediated endocytosis / signaling receptor activity / spermatogenesis / early endosome / endosome / endosome membrane / positive regulation of apoptotic process / G protein-coupled receptor signaling pathway / Golgi membrane / focal adhesion / Neutrophil degranulation / perinuclear region of cytoplasm / enzyme binding / cell surface / Golgi apparatus / signal transduction / extracellular exosome / membrane / identical protein binding / plasma membrane Similarity search - Function | |||||||||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 3.47 Å MOLECULAR REPLACEMENT / Resolution: 3.47 Å | |||||||||||||||
 Authors Authors | Bochel, A.J. / Williams, C. / McCoy, A.J. / Hoppe, H. / Winter, A.J. / Nicholls, R.D. / Harlos, K. / Jones, Y.E. / Berger, I. / Hassan, B. / Crump, M.P. | |||||||||||||||
| Funding support |  United Kingdom, 4items United Kingdom, 4items
| |||||||||||||||
 Citation Citation |  Journal: Structure / Year: 2020 Journal: Structure / Year: 2020Title: Structure of the Human Cation-Independent Mannose 6-Phosphate/IGF2 Receptor Domains 7-11 Uncovers the Mannose 6-Phosphate Binding Site of Domain 9. Authors: Bochel, A.J. / Williams, C. / McCoy, A.J. / Hoppe, H.J. / Winter, A.J. / Nicholls, R.D. / Harlos, K. / Jones, E.Y. / Berger, I. / Hassan, A.B. / Crump, M.P. | |||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  6z32.cif.gz 6z32.cif.gz | 549.2 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb6z32.ent.gz pdb6z32.ent.gz | 456.6 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  6z32.json.gz 6z32.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/z3/6z32 https://data.pdbj.org/pub/pdb/validation_reports/z3/6z32 ftp://data.pdbj.org/pub/pdb/validation_reports/z3/6z32 ftp://data.pdbj.org/pub/pdb/validation_reports/z3/6z32 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  6z30C 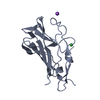 6z31C  1gp0S S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data | |
| Other databases |
|
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||||||||||||
| 2 | 
| ||||||||||||||||||
| Unit cell |
| ||||||||||||||||||
| Noncrystallographic symmetry (NCS) | NCS domain:
NCS domain segments: Component-ID: _ / Ens-ID: 1 / Beg auth comp-ID: CYS / Beg label comp-ID: CYS / End auth comp-ID: CYS / End label comp-ID: CYS / Refine code: _ / Auth seq-ID: 934 - 1646 / Label seq-ID: 22 - 734
|
- Components
Components
| #1: Protein | Mass: 80832.938 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Details: ETGQLKHHHHHHEFTTTDTDQ are vector derived / Source: (gene. exp.)  Homo sapiens (human) / Gene: IGF2R, MPRI / Cell line (production host): HEK293S / Production host: Homo sapiens (human) / Gene: IGF2R, MPRI / Cell line (production host): HEK293S / Production host:  Homo sapiens (human) / References: UniProt: P11717 Homo sapiens (human) / References: UniProt: P11717#2: Polysaccharide | Source method: isolated from a genetically manipulated source #3: Chemical | ChemComp-SO4 / | #4: Sugar | Has ligand of interest | N | Has protein modification | Y | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 3.75 Å3/Da / Density % sol: 67.2 % |
|---|---|
| Crystal grow | Temperature: 298 K / Method: vapor diffusion, sitting drop / pH: 6.5 Details: 0.1 M MES pH 6.5, 1.6 M MgSO4, 10 mM mannose 6-phosphate |
-Data collection
| Diffraction | Mean temperature: 80 K / Serial crystal experiment: N |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  Diamond Diamond  / Beamline: I03 / Wavelength: 1.05811 Å / Beamline: I03 / Wavelength: 1.05811 Å |
| Detector | Type: DECTRIS EIGER2 XE 16M / Detector: PIXEL / Date: Mar 7, 2013 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1.05811 Å / Relative weight: 1 |
| Reflection | Resolution: 3.47→89.72 Å / Num. obs: 30224 / % possible obs: 98.55 % / Redundancy: 14.5 % / CC1/2: 0.997 / Net I/σ(I): 6.03 |
| Reflection shell | Resolution: 3.47→3.59 Å / Redundancy: 14.9 % / Mean I/σ(I) obs: 0.84 / Num. unique obs: 2944 / CC1/2: 0.127 / % possible all: 98.43 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: Homology models of D7, 8, 9, 10, Crystal structure of D11 (1GP0) Resolution: 3.47→89.72 Å / Cor.coef. Fo:Fc: 0.92 / Cor.coef. Fo:Fc free: 0.913 / SU B: 105.945 / SU ML: 0.706 / Cross valid method: THROUGHOUT / ESU R Free: 0.628 / Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.2 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 197.426 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: 1 / Resolution: 3.47→89.72 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller








 PDBj
PDBj





