+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 6sip | ||||||
|---|---|---|---|---|---|---|---|
| Title | Fragment AZ-011 binding at the p53pT387/14-3-3 sigma interface | ||||||
 Components Components |
| ||||||
 Keywords Keywords | PEPTIDE BINDING PROTEIN / protein protein interaction / fragment soaking / stabilization | ||||||
| Function / homology |  Function and homology information Function and homology informationnegative regulation of helicase activity / Loss of function of TP53 in cancer due to loss of tetramerization ability / Regulation of TP53 Expression / signal transduction by p53 class mediator / negative regulation of G1 to G0 transition / negative regulation of glucose catabolic process to lactate via pyruvate / Transcriptional activation of cell cycle inhibitor p21 / regulation of intrinsic apoptotic signaling pathway by p53 class mediator / negative regulation of pentose-phosphate shunt / ATP-dependent DNA/DNA annealing activity ...negative regulation of helicase activity / Loss of function of TP53 in cancer due to loss of tetramerization ability / Regulation of TP53 Expression / signal transduction by p53 class mediator / negative regulation of G1 to G0 transition / negative regulation of glucose catabolic process to lactate via pyruvate / Transcriptional activation of cell cycle inhibitor p21 / regulation of intrinsic apoptotic signaling pathway by p53 class mediator / negative regulation of pentose-phosphate shunt / ATP-dependent DNA/DNA annealing activity / Activation of NOXA and translocation to mitochondria / regulation of cell cycle G2/M phase transition / regulation of fibroblast apoptotic process / oligodendrocyte apoptotic process / negative regulation of miRNA processing / intrinsic apoptotic signaling pathway in response to hypoxia / positive regulation of thymocyte apoptotic process / oxidative stress-induced premature senescence / regulation of tissue remodeling / positive regulation of mitochondrial membrane permeability / mRNA transcription / bone marrow development / positive regulation of programmed necrotic cell death / circadian behavior / T cell proliferation involved in immune response / regulation of mitochondrial membrane permeability involved in apoptotic process / germ cell nucleus / RUNX3 regulates CDKN1A transcription / glucose catabolic process to lactate via pyruvate / TP53 Regulates Transcription of Death Receptors and Ligands / Activation of PUMA and translocation to mitochondria / TP53 regulates transcription of additional cell cycle genes whose exact role in the p53 pathway remain uncertain / regulation of DNA damage response, signal transduction by p53 class mediator / histone deacetylase regulator activity / negative regulation of glial cell proliferation / Regulation of TP53 Activity through Association with Co-factors / negative regulation of neuroblast proliferation / mitochondrial DNA repair / T cell lineage commitment / Formation of Senescence-Associated Heterochromatin Foci (SAHF) / ER overload response / thymocyte apoptotic process / B cell lineage commitment / TP53 Regulates Transcription of Caspase Activators and Caspases / negative regulation of mitophagy / cardiac septum morphogenesis / regulation of epidermal cell division / protein kinase C inhibitor activity / negative regulation of DNA replication / entrainment of circadian clock by photoperiod / positive regulation of epidermal cell differentiation / keratinocyte development / keratinization / negative regulation of telomere maintenance via telomerase / Zygotic genome activation (ZGA) / positive regulation of release of cytochrome c from mitochondria / PI5P Regulates TP53 Acetylation / necroptotic process / Association of TriC/CCT with target proteins during biosynthesis / regulation of cell-cell adhesion / TP53 Regulates Transcription of Genes Involved in Cytochrome C Release / TFIID-class transcription factor complex binding / SUMOylation of transcription factors / TP53 regulates transcription of several additional cell death genes whose specific roles in p53-dependent apoptosis remain uncertain / intrinsic apoptotic signaling pathway by p53 class mediator / negative regulation of reactive oxygen species metabolic process / rRNA transcription / Transcriptional Regulation by VENTX / cAMP/PKA signal transduction / replicative senescence / Regulation of localization of FOXO transcription factors / cellular response to UV-C / general transcription initiation factor binding / keratinocyte proliferation / cellular response to actinomycin D / intrinsic apoptotic signaling pathway in response to endoplasmic reticulum stress / positive regulation of RNA polymerase II transcription preinitiation complex assembly / positive regulation of execution phase of apoptosis / intrinsic apoptotic signaling pathway in response to DNA damage by p53 class mediator / neuroblast proliferation / Pyroptosis / phosphoserine residue binding / hematopoietic stem cell differentiation / Activation of BAD and translocation to mitochondria / viral process / negative regulation of keratinocyte proliferation / response to X-ray / embryonic organ development / establishment of skin barrier / chromosome organization / type II interferon-mediated signaling pathway / negative regulation of protein localization to plasma membrane / somitogenesis / TP53 Regulates Transcription of Genes Involved in G1 Cell Cycle Arrest / hematopoietic progenitor cell differentiation / glial cell proliferation / Chk1/Chk2(Cds1) mediated inactivation of Cyclin B:Cdk1 complex / SARS-CoV-2 targets host intracellular signalling and regulatory pathways / negative regulation of fibroblast proliferation / core promoter sequence-specific DNA binding Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  MOLECULAR REPLACEMENT / Resolution: 1.6004470265 Å MOLECULAR REPLACEMENT / Resolution: 1.6004470265 Å | ||||||
 Authors Authors | Leysen, S. / Wolter, M. / Guillory, X. / Genet, S. / Somsen, B. / Patel, J. / Castaldi, P. / Ottmann, C. | ||||||
| Funding support |  Netherlands, 1items Netherlands, 1items
| ||||||
 Citation Citation |  Journal: J.Med.Chem. / Year: 2020 Journal: J.Med.Chem. / Year: 2020Title: Fragment-based Differential Targeting of PPI Stabilizer Interfaces. Authors: Guillory, X. / Wolter, M. / Leysen, S. / Neves, J.F. / Kuusk, A. / Genet, S. / Somsen, B. / Morrow, J.K. / Rivers, E. / van Beek, L. / Patel, J. / Goodnow, R. / Schoenherr, H. / Fuller, N. / ...Authors: Guillory, X. / Wolter, M. / Leysen, S. / Neves, J.F. / Kuusk, A. / Genet, S. / Somsen, B. / Morrow, J.K. / Rivers, E. / van Beek, L. / Patel, J. / Goodnow, R. / Schoenherr, H. / Fuller, N. / Cao, Q. / Doveston, R.G. / Brunsveld, L. / Arkin, M.R. / Castaldi, P. / Boyd, H. / Landrieu, I. / Chen, H. / Ottmann, C. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  6sip.cif.gz 6sip.cif.gz | 89.8 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb6sip.ent.gz pdb6sip.ent.gz | 53.1 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  6sip.json.gz 6sip.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Summary document |  6sip_validation.pdf.gz 6sip_validation.pdf.gz | 965.5 KB | Display |  wwPDB validaton report wwPDB validaton report |
|---|---|---|---|---|
| Full document |  6sip_full_validation.pdf.gz 6sip_full_validation.pdf.gz | 967 KB | Display | |
| Data in XML |  6sip_validation.xml.gz 6sip_validation.xml.gz | 15.2 KB | Display | |
| Data in CIF |  6sip_validation.cif.gz 6sip_validation.cif.gz | 23 KB | Display | |
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/si/6sip https://data.pdbj.org/pub/pdb/validation_reports/si/6sip ftp://data.pdbj.org/pub/pdb/validation_reports/si/6sip ftp://data.pdbj.org/pub/pdb/validation_reports/si/6sip | HTTPS FTP |
-Related structure data
| Related structure data |  6r5lC  6rhcC  6rjlC  6rjqC  6rjzC  6rk8C  6rkiC  6rkkC  6rkmC  6rl3C  6rl4C  6rl6C  6rm5C  6rm7C  6rp6C  6rwhC  6rwiC  6rwsC  6rwuC  6rx2C  6s39C  6s3cC  6s40C  6s9qC  6sinC  6sioC  6siqC  6slvC  6slwC  6slxC  5mocS C: citing same article ( S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||||||
| Unit cell |
|
- Components
Components
-Protein / Protein/peptide , 2 types, 2 molecules AP
| #1: Protein | Mass: 26542.914 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: SFN, HME1 / Production host: Homo sapiens (human) / Gene: SFN, HME1 / Production host:  |
|---|---|
| #2: Protein/peptide | Mass: 1449.520 Da / Num. of mol.: 1 / Source method: obtained synthetically / Source: (synth.)  Homo sapiens (human) / References: UniProt: P04637 Homo sapiens (human) / References: UniProt: P04637 |
-Non-polymers , 4 types, 344 molecules 

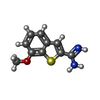




| #3: Chemical | | #4: Chemical | ChemComp-CL / | #5: Chemical | ChemComp-LFB / | #6: Water | ChemComp-HOH / | |
|---|
-Details
| Has ligand of interest | Y |
|---|---|
| Has protein modification | Y |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.64 Å3/Da / Density % sol: 53.34 % |
|---|---|
| Crystal grow | Temperature: 277 K / Method: vapor diffusion, hanging drop Details: 0.1M Hepes, pH7.5, 27%PEG, 5% Glycerol, 0.2M Calcium Chloride, 2mM DTT |
-Data collection
| Diffraction | Mean temperature: 100 K / Serial crystal experiment: N |
|---|---|
| Diffraction source | Source:  ROTATING ANODE / Type: RIGAKU FR-E+ SUPERBRIGHT / Wavelength: 1.54 Å ROTATING ANODE / Type: RIGAKU FR-E+ SUPERBRIGHT / Wavelength: 1.54 Å |
| Detector | Type: RIGAKU SATURN 944+ / Detector: CCD / Date: May 2, 2016 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1.54 Å / Relative weight: 1 |
| Reflection | Resolution: 1.6→62.68 Å / Num. obs: 29038 / % possible obs: 74.8 % / Redundancy: 3.1 % / Biso Wilson estimate: 12.2248090387 Å2 / CC1/2: 0.999 / Rmerge(I) obs: 0.026 / Net I/σ(I): 34.3 |
| Reflection shell | Resolution: 1.6→1.69 Å / Redundancy: 1.2 % / Mean I/σ(I) obs: 6.7 / Num. unique obs: 1460 / CC1/2: 0.971 / % possible all: 26.1 |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 5MOC Resolution: 1.6004470265→19.9296097901 Å / SU ML: 0.128115032053 / Cross valid method: FREE R-VALUE / σ(F): 1.34879926691 / Phase error: 19.3000125847 Stereochemistry target values: GeoStd + Monomer Library + CDL v1.2
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å / Solvent model: FLAT BULK SOLVENT MODEL | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 15.4714960146 Å2 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.6004470265→19.9296097901 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell |
|
 Movie
Movie Controller
Controller























 PDBj
PDBj





















