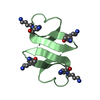
Entry Database : PDB / ID : 6q6cTitle Pore-modulating toxins exploit inherent slow inactivation to block K+ channels Kunitz-type conkunitzin-S1 Keywords Function / homology Function Domain/homology Component
/ / / / / / / / / Biological species Conus striatus (striated cone)Method / / / Resolution : 1.3 Å Authors Karbat, I. / Gueta, H. / Fine, S. / Szanto, T. / Hamer-Rogotner, S. / Dym, O. / Frolow, F. / Gordon, D. / Panyi, G. / Gurevitz, M. / Reuveny, E. Funding support Organization Grant number Country Israel Science Foundation 1248/2015
Journal : Proc.Natl.Acad.Sci.USA / Year : 2019Title : Pore-modulating toxins exploit inherent slow inactivation to block K+channels.Authors : Karbat, I. / Altman-Gueta, H. / Fine, S. / Szanto, T. / Hamer-Rogotner, S. / Dym, O. / Frolow, F. / Gordon, D. / Panyi, G. / Gurevitz, M. / Reuveny, E. History Deposition Dec 10, 2018 Deposition site / Processing site Revision 1.0 Aug 21, 2019 Provider / Type Revision 1.1 Sep 4, 2019 Group / Database references / Category / citation_authorItem _citation.country / _citation.journal_abbrev ... _citation.country / _citation.journal_abbrev / _citation.journal_id_ASTM / _citation.journal_id_CSD / _citation.journal_id_ISSN / _citation.pdbx_database_id_DOI / _citation.pdbx_database_id_PubMed / _citation.title / _citation.year / _citation_author.identifier_ORCID / _citation_author.name Revision 1.2 Sep 18, 2019 Group / Database references / Category Item / _citation.page_first / _citation.page_lastRevision 1.3 Jan 24, 2024 Group / Database references / Refinement descriptionCategory chem_comp_atom / chem_comp_bond ... chem_comp_atom / chem_comp_bond / database_2 / pdbx_initial_refinement_model / refine_hist Item / _database_2.pdbx_database_accession / _refine_hist.d_res_lowRevision 1.4 Nov 13, 2024 Group / Category / pdbx_modification_feature
Show all Show less
 Yorodumi
Yorodumi Open data
Open data Basic information
Basic information Components
Components Keywords
Keywords Function and homology information
Function and homology information Conus striatus (striated cone)
Conus striatus (striated cone) X-RAY DIFFRACTION /
X-RAY DIFFRACTION /  SYNCHROTRON /
SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.3 Å
MOLECULAR REPLACEMENT / Resolution: 1.3 Å  Authors
Authors Israel, 1items
Israel, 1items  Citation
Citation Journal: Proc.Natl.Acad.Sci.USA / Year: 2019
Journal: Proc.Natl.Acad.Sci.USA / Year: 2019 Structure visualization
Structure visualization Molmil
Molmil Jmol/JSmol
Jmol/JSmol Downloads & links
Downloads & links Download
Download 6q6c.cif.gz
6q6c.cif.gz PDBx/mmCIF format
PDBx/mmCIF format pdb6q6c.ent.gz
pdb6q6c.ent.gz PDB format
PDB format 6q6c.json.gz
6q6c.json.gz PDBx/mmJSON format
PDBx/mmJSON format Other downloads
Other downloads https://data.pdbj.org/pub/pdb/validation_reports/q6/6q6c
https://data.pdbj.org/pub/pdb/validation_reports/q6/6q6c ftp://data.pdbj.org/pub/pdb/validation_reports/q6/6q6c
ftp://data.pdbj.org/pub/pdb/validation_reports/q6/6q6c

 Links
Links Assembly
Assembly

 Components
Components Conus striatus (striated cone) / Production host:
Conus striatus (striated cone) / Production host: 
 X-RAY DIFFRACTION / Number of used crystals: 1
X-RAY DIFFRACTION / Number of used crystals: 1  Sample preparation
Sample preparation SYNCHROTRON / Site:
SYNCHROTRON / Site:  ESRF
ESRF  / Beamline: ID14-4 / Wavelength: 0.98 Å
/ Beamline: ID14-4 / Wavelength: 0.98 Å Processing
Processing MOLECULAR REPLACEMENT
MOLECULAR REPLACEMENT Movie
Movie Controller
Controller









 PDBj
PDBj









