+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 6pp9 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Crystal structure of BRAF:MEK1 complex | |||||||||
 Components Components |
| |||||||||
 Keywords Keywords | TRANSFERASE / BRAF / MEK1 | |||||||||
| Function / homology |  Function and homology information Function and homology informationnegative regulation of homotypic cell-cell adhesion / epithelial cell proliferation involved in lung morphogenesis / positive regulation of endodermal cell differentiation / negative regulation of hypoxia-induced intrinsic apoptotic signaling pathway / regulation of vascular associated smooth muscle contraction / CD4-positive, alpha-beta T cell differentiation / positive regulation of axon regeneration / CD4-positive or CD8-positive, alpha-beta T cell lineage commitment / negative regulation of synaptic vesicle exocytosis / mitogen-activated protein kinase kinase ...negative regulation of homotypic cell-cell adhesion / epithelial cell proliferation involved in lung morphogenesis / positive regulation of endodermal cell differentiation / negative regulation of hypoxia-induced intrinsic apoptotic signaling pathway / regulation of vascular associated smooth muscle contraction / CD4-positive, alpha-beta T cell differentiation / positive regulation of axon regeneration / CD4-positive or CD8-positive, alpha-beta T cell lineage commitment / negative regulation of synaptic vesicle exocytosis / mitogen-activated protein kinase kinase / positive regulation of muscle contraction / Golgi inheritance / placenta blood vessel development / Signalling to p38 via RIT and RIN / MAP kinase scaffold activity / regulation of axon regeneration / cerebellar cortex formation / labyrinthine layer development / head morphogenesis / ARMS-mediated activation / myeloid progenitor cell differentiation / melanosome transport / endothelial cell apoptotic process / Signaling by MAP2K mutants / SHOC2 M1731 mutant abolishes MRAS complex function / Gain-of-function MRAS complexes activate RAF signaling / negative regulation of fibroblast migration / positive regulation of D-glucose transmembrane transport / establishment of protein localization to membrane / type B pancreatic cell proliferation / central nervous system neuron differentiation / vesicle transport along microtubule / positive regulation of Ras protein signal transduction / regulation of Golgi inheritance / mitogen-activated protein kinase kinase kinase binding / regulation of T cell differentiation / positive regulation of axonogenesis / trachea formation / triglyceride homeostasis / regulation of early endosome to late endosome transport / Negative feedback regulation of MAPK pathway / regulation of stress-activated MAPK cascade / Frs2-mediated activation / stress fiber assembly / MAPK3 (ERK1) activation / ERBB2-ERBB3 signaling pathway / face development / MAP kinase kinase activity / endodermal cell differentiation / regulation of neurotransmitter receptor localization to postsynaptic specialization membrane / Bergmann glial cell differentiation / positive regulation of protein serine/threonine kinase activity / thyroid gland development / positive regulation of ATP biosynthetic process / Uptake and function of anthrax toxins / somatic stem cell population maintenance / positive regulation of peptidyl-serine phosphorylation / synaptic vesicle exocytosis / MAP kinase kinase kinase activity / protein kinase activator activity / negative regulation of endothelial cell apoptotic process / response to axon injury / Schwann cell development / postsynaptic modulation of chemical synaptic transmission / keratinocyte differentiation / neuron projection morphogenesis / ERK1 and ERK2 cascade / positive regulation of stress fiber assembly / myelination / positive regulation of substrate adhesion-dependent cell spreading / protein serine/threonine/tyrosine kinase activity / substrate adhesion-dependent cell spreading / insulin-like growth factor receptor signaling pathway / positive regulation of autophagy / cellular response to calcium ion / response to glucocorticoid / dendrite cytoplasm / thymus development / animal organ morphogenesis / MAP3K8 (TPL2)-dependent MAPK1/3 activation / Signal transduction by L1 / protein serine/threonine kinase activator activity / cell motility / positive regulation of transcription elongation by RNA polymerase II / sperm end piece / RAF activation / Spry regulation of FGF signaling / Signaling by high-kinase activity BRAF mutants / MAP2K and MAPK activation / visual learning / cellular response to xenobiotic stimulus / small GTPase binding / epidermal growth factor receptor signaling pathway / centriolar satellite / chemotaxis / neuron differentiation / Signaling by RAF1 mutants / Signaling by moderate kinase activity BRAF mutants / Paradoxical activation of RAF signaling by kinase inactive BRAF / Signaling downstream of RAS mutants Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.59 Å MOLECULAR REPLACEMENT / Resolution: 2.59 Å | |||||||||
 Authors Authors | Li, K. / Gonzalez Del-Pino, G. / Park, E. / Eck, M.J. | |||||||||
| Funding support |  United States, 2items United States, 2items
| |||||||||
 Citation Citation |  Journal: Nature / Year: 2019 Journal: Nature / Year: 2019Title: Architecture of autoinhibited and active BRAF-MEK1-14-3-3 complexes. Authors: Eunyoung Park / Shaun Rawson / Kunhua Li / Byeong-Won Kim / Scott B Ficarro / Gonzalo Gonzalez-Del Pino / Humayun Sharif / Jarrod A Marto / Hyesung Jeon / Michael J Eck /  Abstract: RAF family kinases are RAS-activated switches that initiate signalling through the MAP kinase cascade to control cellular proliferation, differentiation and survival. RAF activity is tightly ...RAF family kinases are RAS-activated switches that initiate signalling through the MAP kinase cascade to control cellular proliferation, differentiation and survival. RAF activity is tightly regulated and inappropriate activation is a frequent cause of cancer; however, the structural basis for RAF regulation is poorly understood at present. Here we use cryo-electron microscopy to determine autoinhibited and active-state structures of full-length BRAF in complexes with MEK1 and a 14-3-3 dimer. The reconstruction reveals an inactive BRAF-MEK1 complex restrained in a cradle formed by the 14-3-3 dimer, which binds the phosphorylated S365 and S729 sites that flank the BRAF kinase domain. The BRAF cysteine-rich domain occupies a central position that stabilizes this assembly, but the adjacent RAS-binding domain is poorly ordered and peripheral. The 14-3-3 cradle maintains autoinhibition by sequestering the membrane-binding cysteine-rich domain and blocking dimerization of the BRAF kinase domain. In the active state, these inhibitory interactions are released and a single 14-3-3 dimer rearranges to bridge the C-terminal pS729 binding sites of two BRAFs, which drives the formation of an active, back-to-back BRAF dimer. Our structural snapshots provide a foundation for understanding normal RAF regulation and its mutational disruption in cancer and developmental syndromes. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  6pp9.cif.gz 6pp9.cif.gz | 141.1 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb6pp9.ent.gz pdb6pp9.ent.gz | 105.2 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  6pp9.json.gz 6pp9.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/pp/6pp9 https://data.pdbj.org/pub/pdb/validation_reports/pp/6pp9 ftp://data.pdbj.org/pub/pdb/validation_reports/pp/6pp9 ftp://data.pdbj.org/pub/pdb/validation_reports/pp/6pp9 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  0541C  6nybSC  6q0jC  6q0kC  6q0tC S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
-Protein , 2 types, 2 molecules AB
| #1: Protein | Mass: 32098.104 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: BRAF, BRAF1, RAFB1 / Plasmid: pFASTBac Dual / Cell line (production host): Sf9 / Production host: Homo sapiens (human) / Gene: BRAF, BRAF1, RAFB1 / Plasmid: pFASTBac Dual / Cell line (production host): Sf9 / Production host:  References: UniProt: P15056, non-specific serine/threonine protein kinase |
|---|---|
| #2: Protein | Mass: 43790.281 Da / Num. of mol.: 1 / Mutation: S218A, S222A Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: MAP2K1, MEK1, PRKMK1 / Plasmid: PAC8 / Cell line (production host): Sf9 / Production host: Homo sapiens (human) / Gene: MAP2K1, MEK1, PRKMK1 / Plasmid: PAC8 / Cell line (production host): Sf9 / Production host:  References: UniProt: Q02750, mitogen-activated protein kinase kinase |
-Non-polymers , 7 types, 82 molecules 


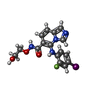









| #3: Chemical | | #4: Chemical | #5: Chemical | #6: Chemical | ChemComp-LCJ / | #7: Chemical | ChemComp-SO4 / | #8: Chemical | #9: Water | ChemComp-HOH / | |
|---|
-Details
| Has ligand of interest | Y |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 3.39 Å3/Da / Density % sol: 67.29 % |
|---|---|
| Crystal grow | Temperature: 293 K / Method: vapor diffusion, hanging drop / pH: 6.4 Details: 100 mM Bis-Tris pH 6.4, 200 mM Ammonium Sulfate, and 22% PEG3350 PH range: 6.0-6.8 / Temp details: RT |
-Data collection
| Diffraction | Mean temperature: 100 K / Serial crystal experiment: N |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  APS APS  / Beamline: 24-ID-C / Wavelength: 0.978 Å / Beamline: 24-ID-C / Wavelength: 0.978 Å |
| Detector | Type: DECTRIS PILATUS 6M / Detector: PIXEL / Date: Jun 11, 2019 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.978 Å / Relative weight: 1 |
| Reflection | Resolution: 2.58→43.648 Å / Num. obs: 32549 / % possible obs: 99.95 % / Redundancy: 13.3 % / Biso Wilson estimate: 52.17 Å2 / CC1/2: 0.999 / Rmerge(I) obs: 0.215 / Rpim(I) all: 0.06127 / Rrim(I) all: 0.2237 / Net I/σ(I): 10.86 |
| Reflection shell | Resolution: 2.58→2.683 Å / Redundancy: 13.9 % / Mean I/σ(I) obs: 2 / Num. unique obs: 3190 / CC1/2: 0.888 / Rpim(I) all: 0.4253 / % possible all: 100 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 6NYB Resolution: 2.59→43.648 Å / SU ML: 0.31 / Cross valid method: FREE R-VALUE / σ(F): 1.34 / Phase error: 24.7
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso max: 122.96 Å2 / Biso mean: 53.5009 Å2 / Biso min: 27.5 Å2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: final / Resolution: 2.59→43.648 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Refine-ID: X-RAY DIFFRACTION / Rfactor Rfree error: 0 / % reflection obs: 100 %
|
 Movie
Movie Controller
Controller
















 PDBj
PDBj
























