Entry Database : PDB / ID : 6nyhTitle Structure of human RIPK1 kinase domain in complex with GNE684 Receptor-interacting serine/threonine-protein kinase 1 Keywords / / / / / Function / homology Function Domain/homology Component
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / Biological species Homo sapiens (human)Method / / / Resolution : 2.1 Å Authors Fong, R. / Lupardus, P.J. Journal : Cell Death Differ. / Year : 2020Title : RIP1 inhibition blocks inflammatory diseases but not tumor growth or metastases.Authors: Patel, S. / Webster, J.D. / Varfolomeev, E. / Kwon, Y.C. / Cheng, J.H. / Zhang, J. / Dugger, D.L. / Wickliffe, K.E. / Maltzman, A. / Sujatha-Bhaskar, S. / Bir Kohli, P. / Ramaswamy, S. / ... Authors : Patel, S. / Webster, J.D. / Varfolomeev, E. / Kwon, Y.C. / Cheng, J.H. / Zhang, J. / Dugger, D.L. / Wickliffe, K.E. / Maltzman, A. / Sujatha-Bhaskar, S. / Bir Kohli, P. / Ramaswamy, S. / Deshmukh, G. / Liederer, B.M. / Fong, R. / Hamilton, G. / Lupardus, P. / Caplazi, P. / Lee, W.P. / van Lookeren Campagne, M. / Johnson, A. / McKenzie, B.S. / Junttila, M.R. / Newton, K. / Vucic, D. History Deposition Feb 11, 2019 Deposition site / Processing site Revision 1.0 May 29, 2019 Provider / Type Revision 1.1 Jan 1, 2020 Group / Category / citation_authorItem _citation.journal_volume / _citation.page_first ... _citation.journal_volume / _citation.page_first / _citation.page_last / _citation.year / _citation_author.identifier_ORCID Revision 1.2 Oct 11, 2023 Group / Database references / Refinement descriptionCategory chem_comp_atom / chem_comp_bond ... chem_comp_atom / chem_comp_bond / database_2 / pdbx_initial_refinement_model Item / _database_2.pdbx_database_accession
Show all Show less
 Open data
Open data Basic information
Basic information Components
Components Keywords
Keywords Function and homology information
Function and homology information Homo sapiens (human)
Homo sapiens (human) X-RAY DIFFRACTION /
X-RAY DIFFRACTION /  SYNCHROTRON /
SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.1 Å
MOLECULAR REPLACEMENT / Resolution: 2.1 Å  Authors
Authors Citation
Citation Journal: Cell Death Differ. / Year: 2020
Journal: Cell Death Differ. / Year: 2020 Structure visualization
Structure visualization Molmil
Molmil Jmol/JSmol
Jmol/JSmol Downloads & links
Downloads & links Download
Download 6nyh.cif.gz
6nyh.cif.gz PDBx/mmCIF format
PDBx/mmCIF format pdb6nyh.ent.gz
pdb6nyh.ent.gz PDB format
PDB format 6nyh.json.gz
6nyh.json.gz PDBx/mmJSON format
PDBx/mmJSON format Other downloads
Other downloads https://data.pdbj.org/pub/pdb/validation_reports/ny/6nyh
https://data.pdbj.org/pub/pdb/validation_reports/ny/6nyh ftp://data.pdbj.org/pub/pdb/validation_reports/ny/6nyh
ftp://data.pdbj.org/pub/pdb/validation_reports/ny/6nyh
 Links
Links Assembly
Assembly
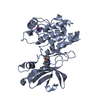

 Components
Components Homo sapiens (human) / Gene: RIPK1, RIP, RIP1 / Production host:
Homo sapiens (human) / Gene: RIPK1, RIP, RIP1 / Production host: 
 X-RAY DIFFRACTION / Number of used crystals: 1
X-RAY DIFFRACTION / Number of used crystals: 1  Sample preparation
Sample preparation SYNCHROTRON / Site:
SYNCHROTRON / Site:  APS
APS  / Beamline: 21-ID-D / Wavelength: 1 Å
/ Beamline: 21-ID-D / Wavelength: 1 Å Processing
Processing MOLECULAR REPLACEMENT
MOLECULAR REPLACEMENT Movie
Movie Controller
Controller













 PDBj
PDBj













