| Entry | Database: PDB / ID: 6myk
|
|---|
| Title | Pleurotus ostreatus OstreolysinA mutant E69A with Bis-Tris |
|---|
 Components Components | Ostreolysin A6 |
|---|
 Keywords Keywords | MEMBRANE PROTEIN / BETA-SANDWICH FOLD / MEMBRANE BINDING PROTEIN |
|---|
| Function / homology | Mutm (Fpg) Protein; Chain: A, domain 2 - #50 / Hemolysin, aegerolysin type / Aegerolysin / Mutm (Fpg) Protein; Chain: A, domain 2 / symbiont-mediated hemolysis of host erythrocyte / Sandwich / Mainly Beta / Ostreolysin A6 Function and homology information Function and homology information |
|---|
| Biological species |  Pleurotus ostreatus (oyster mushroom) Pleurotus ostreatus (oyster mushroom) |
|---|
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.8 Å MOLECULAR REPLACEMENT / Resolution: 1.8 Å |
|---|
 Authors Authors | Tomchick, D.R. / Radhakrishnan, A. / Endapally, S. |
|---|
| Funding support |  United States, 3items United States, 3items | Organization | Grant number | Country |
|---|
| National Institutes of Health/National Heart, Lung, and Blood Institute (NIH/NHLBI) | HL20948 |  United States United States | | American Heart Association | 12SDG12040267 |  United States United States | | Welch Foundation | I-1793 |  United States United States |
|
|---|
 Citation Citation |  Journal: Cell / Year: 2019 Journal: Cell / Year: 2019
Title: Molecular Discrimination between Two Conformations of Sphingomyelin in Plasma Membranes.
Authors: Endapally, S. / Frias, D. / Grzemska, M. / Gay, A. / Tomchick, D.R. / Radhakrishnan, A. |
|---|
| History | | Deposition | Nov 1, 2018 | Deposition site: RCSB / Processing site: RCSB |
|---|
| Revision 1.0 | Feb 13, 2019 | Provider: repository / Type: Initial release |
|---|
| Revision 1.1 | Mar 6, 2019 | Group: Data collection / Database references
Category: citation / database_PDB_rev ...citation / database_PDB_rev / database_PDB_rev_record / pdbx_database_proc
Item: _citation.journal_volume / _citation.page_first |
|---|
| Revision 1.2 | Dec 4, 2019 | Group: Author supporting evidence / Category: pdbx_audit_support / Item: _pdbx_audit_support.funding_organization |
|---|
| Revision 1.3 | Oct 11, 2023 | Group: Data collection / Database references ...Data collection / Database references / Derived calculations / Refinement description
Category: chem_comp_atom / chem_comp_bond ...chem_comp_atom / chem_comp_bond / database_2 / pdbx_initial_refinement_model / struct_conn
Item: _database_2.pdbx_DOI / _database_2.pdbx_database_accession ..._database_2.pdbx_DOI / _database_2.pdbx_database_accession / _struct_conn.pdbx_dist_value / _struct_conn.ptnr1_auth_asym_id / _struct_conn.ptnr1_auth_comp_id / _struct_conn.ptnr1_auth_seq_id / _struct_conn.ptnr1_label_asym_id / _struct_conn.ptnr1_label_atom_id / _struct_conn.ptnr1_label_comp_id / _struct_conn.ptnr1_label_seq_id / _struct_conn.ptnr2_auth_asym_id / _struct_conn.ptnr2_auth_comp_id / _struct_conn.ptnr2_auth_seq_id / _struct_conn.ptnr2_label_asym_id / _struct_conn.ptnr2_label_atom_id / _struct_conn.ptnr2_label_comp_id |
|---|
|
|---|
 Open data
Open data Basic information
Basic information Components
Components Keywords
Keywords Function and homology information
Function and homology information Pleurotus ostreatus (oyster mushroom)
Pleurotus ostreatus (oyster mushroom) X-RAY DIFFRACTION /
X-RAY DIFFRACTION /  SYNCHROTRON /
SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.8 Å
MOLECULAR REPLACEMENT / Resolution: 1.8 Å  Authors
Authors United States, 3items
United States, 3items  Citation
Citation Journal: Cell / Year: 2019
Journal: Cell / Year: 2019 Structure visualization
Structure visualization Molmil
Molmil Jmol/JSmol
Jmol/JSmol Downloads & links
Downloads & links Download
Download 6myk.cif.gz
6myk.cif.gz PDBx/mmCIF format
PDBx/mmCIF format pdb6myk.ent.gz
pdb6myk.ent.gz PDB format
PDB format 6myk.json.gz
6myk.json.gz PDBx/mmJSON format
PDBx/mmJSON format Other downloads
Other downloads https://data.pdbj.org/pub/pdb/validation_reports/my/6myk
https://data.pdbj.org/pub/pdb/validation_reports/my/6myk ftp://data.pdbj.org/pub/pdb/validation_reports/my/6myk
ftp://data.pdbj.org/pub/pdb/validation_reports/my/6myk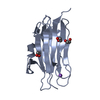


 Links
Links Assembly
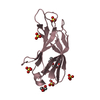
Assembly
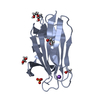

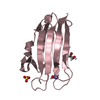

 Components
Components Pleurotus ostreatus (oyster mushroom) / Gene: OlyA6 / Plasmid: pLysS / Production host:
Pleurotus ostreatus (oyster mushroom) / Gene: OlyA6 / Plasmid: pLysS / Production host: 









 X-RAY DIFFRACTION / Number of used crystals: 1
X-RAY DIFFRACTION / Number of used crystals: 1  Sample preparation
Sample preparation SYNCHROTRON / Site:
SYNCHROTRON / Site:  APS
APS  / Beamline: 19-ID / Wavelength: 0.97903 Å
/ Beamline: 19-ID / Wavelength: 0.97903 Å Processing
Processing MOLECULAR REPLACEMENT
MOLECULAR REPLACEMENT Movie
Movie Controller
Controller












 PDBj
PDBj


