+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 | データベース: PDB / ID: 6ezn | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
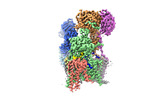
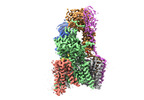
| タイトル | Cryo-EM structure of the yeast oligosaccharyltransferase (OST) complex | |||||||||
 要素 要素 | (Dolichyl-diphosphooligosaccharide--protein glycosyltransferase subunit ...) x 8 | |||||||||
 キーワード キーワード | MEMBRANE PROTEIN / OST complex / oligosaccharyltransferase / N-linked glycosylation / yeast | |||||||||
| 機能・相同性 |  機能・相同性情報 機能・相同性情報Miscellaneous transport and binding events / oligosaccharyltransferase I complex / protein O-linked mannosylation / oligosaccharyltransferase complex / dolichyl-diphosphooligosaccharide-protein glycotransferase / dolichyl-diphosphooligosaccharide-protein glycotransferase activity / protein N-linked glycosylation via asparagine / protein N-linked glycosylation / glycosyltransferase activity / protein glycosylation ...Miscellaneous transport and binding events / oligosaccharyltransferase I complex / protein O-linked mannosylation / oligosaccharyltransferase complex / dolichyl-diphosphooligosaccharide-protein glycotransferase / dolichyl-diphosphooligosaccharide-protein glycotransferase activity / protein N-linked glycosylation via asparagine / protein N-linked glycosylation / glycosyltransferase activity / protein glycosylation / protein-disulfide reductase activity / Neutrophil degranulation / post-translational protein modification / protein-macromolecule adaptor activity / nuclear envelope / protein-containing complex assembly / endoplasmic reticulum membrane / endoplasmic reticulum / mitochondrion / membrane / metal ion binding 類似検索 - 分子機能 | |||||||||
| 生物種 |  | |||||||||
| 手法 | 電子顕微鏡法 / 単粒子再構成法 / クライオ電子顕微鏡法 / 解像度: 3.3 Å | |||||||||
 データ登録者 データ登録者 | Wild, R. / Kowal, J. / Eyring, J. / Ngwa, E.M. / Aebi, M. / Locher, K.P. | |||||||||
| 資金援助 |  スイス, 2件 スイス, 2件
| |||||||||
 引用 引用 |  ジャーナル: Nat Commun / 年: 2022 ジャーナル: Nat Commun / 年: 2022タイトル: Molecular basis for glycan recognition and reaction priming of eukaryotic oligosaccharyltransferase. 著者: Ana S Ramírez / Mario de Capitani / Giorgio Pesciullesi / Julia Kowal / Joël S Bloch / Rossitza N Irobalieva / Jean-Louis Reymond / Markus Aebi / Kaspar P Locher /  要旨: Oligosaccharyltransferase (OST) is the central enzyme of N-linked protein glycosylation. It catalyzes the transfer of a pre-assembled glycan, GlcNAcManGlc, from a dolichyl-pyrophosphate donor to ...Oligosaccharyltransferase (OST) is the central enzyme of N-linked protein glycosylation. It catalyzes the transfer of a pre-assembled glycan, GlcNAcManGlc, from a dolichyl-pyrophosphate donor to acceptor sites in secretory proteins in the lumen of the endoplasmic reticulum. Precise recognition of the fully assembled glycan by OST is essential for the subsequent quality control steps of glycoprotein biosynthesis. However, the molecular basis of the OST-donor glycan interaction is unknown. Here we present cryo-EM structures of S. cerevisiae OST in distinct functional states. Our findings reveal that the terminal glucoses (Glc) of a chemo-enzymatically generated donor glycan analog bind to a pocket formed by the non-catalytic subunits WBP1 and OST2. We further find that binding either donor or acceptor substrate leads to distinct primed states of OST, where subsequent binding of the other substrate triggers conformational changes required for catalysis. This alternate priming allows OST to efficiently process closely spaced N-glycosylation sites. #1:  ジャーナル: Science / 年: 2018 ジャーナル: Science / 年: 2018タイトル: Structure of the yeast oligosaccharyltransferase complex gives insight into eukaryotic N-glycosylation. 著者: Rebekka Wild / Julia Kowal / Jillianne Eyring / Elsy M Ngwa / Markus Aebi / Kaspar P Locher /  要旨: Oligosaccharyltransferase (OST) is an essential membrane protein complex in the endoplasmic reticulum, where it transfers an oligosaccharide from a dolichol-pyrophosphate-activated donor to ...Oligosaccharyltransferase (OST) is an essential membrane protein complex in the endoplasmic reticulum, where it transfers an oligosaccharide from a dolichol-pyrophosphate-activated donor to glycosylation sites of secretory proteins. Here we describe the atomic structure of yeast OST determined by cryo-electron microscopy, revealing a conserved subunit arrangement. The active site of the catalytic STT3 subunit points away from the center of the complex, allowing unhindered access to substrates. The dolichol-pyrophosphate moiety binds to a lipid-exposed groove of STT3, whereas two noncatalytic subunits and an ordered N-glycan form a membrane-proximal pocket for the oligosaccharide. The acceptor polypeptide site faces an oxidoreductase domain in stand-alone OST complexes or is immediately adjacent to the translocon, suggesting how eukaryotic OSTs efficiently glycosylate a large number of polypeptides before their folding. | |||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| ムービー |
 ムービービューア ムービービューア |
|---|---|
| 構造ビューア | 分子:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
- ダウンロードとリンク
ダウンロードとリンク
- ダウンロード
ダウンロード
| PDBx/mmCIF形式 |  6ezn.cif.gz 6ezn.cif.gz | 390 KB | 表示 |  PDBx/mmCIF形式 PDBx/mmCIF形式 |
|---|---|---|---|---|
| PDB形式 |  pdb6ezn.ent.gz pdb6ezn.ent.gz | 314.5 KB | 表示 |  PDB形式 PDB形式 |
| PDBx/mmJSON形式 |  6ezn.json.gz 6ezn.json.gz | ツリー表示 |  PDBx/mmJSON形式 PDBx/mmJSON形式 | |
| その他 |  その他のダウンロード その他のダウンロード |
-検証レポート
| 文書・要旨 |  6ezn_validation.pdf.gz 6ezn_validation.pdf.gz | 1.5 MB | 表示 |  wwPDB検証レポート wwPDB検証レポート |
|---|---|---|---|---|
| 文書・詳細版 |  6ezn_full_validation.pdf.gz 6ezn_full_validation.pdf.gz | 1.5 MB | 表示 | |
| XML形式データ |  6ezn_validation.xml.gz 6ezn_validation.xml.gz | 69 KB | 表示 | |
| CIF形式データ |  6ezn_validation.cif.gz 6ezn_validation.cif.gz | 100.8 KB | 表示 | |
| アーカイブディレクトリ |  https://data.pdbj.org/pub/pdb/validation_reports/ez/6ezn https://data.pdbj.org/pub/pdb/validation_reports/ez/6ezn ftp://data.pdbj.org/pub/pdb/validation_reports/ez/6ezn ftp://data.pdbj.org/pub/pdb/validation_reports/ez/6ezn | HTTPS FTP |
-関連構造データ
- リンク
リンク
- 集合体
集合体
| 登録構造単位 | 
|
|---|---|
| 1 |
|
- 要素
要素
-Dolichyl-diphosphooligosaccharide--protein glycosyltransferase subunit ... , 8種, 8分子 ABCDEFGH
| #1: タンパク質 | 分子量: 54116.477 Da / 分子数: 1 / 由来タイプ: 天然 由来: (天然)  株: ATCC 204508 / S288c 参照: UniProt: P41543, dolichyl-diphosphooligosaccharide-protein glycotransferase |
|---|---|
| #2: タンパク質 | 分子量: 14712.531 Da / 分子数: 1 / 由来タイプ: 天然 由来: (天然)  株: ATCC 204508 / S288c 参照: UniProt: P46964, dolichyl-diphosphooligosaccharide-protein glycotransferase |
| #3: タンパク質 | 分子量: 39518.160 Da / 分子数: 1 / 由来タイプ: 天然 由来: (天然)  株: ATCC 204508 / S288c 参照: UniProt: P48439, dolichyl-diphosphooligosaccharide-protein glycotransferase |
| #4: タンパク質・ペプチド | 分子量: 3986.696 Da / 分子数: 1 / 由来タイプ: 天然 由来: (天然)  株: ATCC 204508 / S288c 参照: UniProt: Q99380, dolichyl-diphosphooligosaccharide-protein glycotransferase |
| #5: タンパク質 | 分子量: 9525.090 Da / 分子数: 1 / 由来タイプ: 天然 由来: (天然)  株: ATCC 204508 / S288c 参照: UniProt: Q92316, dolichyl-diphosphooligosaccharide-protein glycotransferase |
| #6: タンパク質 | 分子量: 81604.539 Da / 分子数: 1 / 由来タイプ: 天然 由来: (天然)  株: ATCC 204508 / S288c 参照: UniProt: P39007, dolichyl-diphosphooligosaccharide-protein glycotransferase |
| #7: タンパク質 | 分子量: 49444.438 Da / 分子数: 1 / 由来タイプ: 天然 由来: (天然)  株: ATCC 204508 / S288c 参照: UniProt: P33767, dolichyl-diphosphooligosaccharide-protein glycotransferase |
| #8: タンパク質 | 分子量: 31682.832 Da / 分子数: 1 / 由来タイプ: 天然 由来: (天然)  株: ATCC 204508 / S288c 参照: UniProt: Q02795, dolichyl-diphosphooligosaccharide-protein glycotransferase |
-糖 , 4種, 5分子 
| #9: 多糖 | beta-D-mannopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-2-acetamido-2-deoxy-beta- ...beta-D-mannopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose | ||||
|---|---|---|---|---|---|
| #10: 多糖 | | #11: 多糖 | alpha-D-mannopyranose-(1-2)-alpha-D-mannopyranose-(1-2)-alpha-D-mannopyranose-(1-3)-[alpha-D- ...alpha-D-mannopyranose-(1-2)-alpha-D-mannopyranose-(1-2)-alpha-D-mannopyranose-(1-3)-[alpha-D-mannopyranose-(1-3)-alpha-D-mannopyranose-(1-6)]beta-D-mannopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose | #14: 糖 | ChemComp-NAG / | |
-非ポリマー , 2種, 5分子 


| #12: 化合物 | ChemComp-CPL / #13: 化合物 | ChemComp-PTY / | |
|---|
-詳細
| 構成要素の詳細 | Amino acid residues H22 to H65 in Swp1 were built as PolyA chain and renamed to specific amino ...Amino acid residues H22 to H65 in Swp1 were built as PolyA chain and renamed to specific amino acids according to residue numbers on request by wwPDB |
|---|
-実験情報
-実験
| 実験 | 手法: 電子顕微鏡法 |
|---|---|
| EM実験 | 試料の集合状態: PARTICLE / 3次元再構成法: 単粒子再構成法 |
- 試料調製
試料調製
| 構成要素 | 名称: yeast oligosaccharyltransferase (OST) complex / タイプ: COMPLEX / Entity ID: #1-#8 / 由来: NATURAL |
|---|---|
| 由来(天然) | 生物種:  |
| 緩衝液 | pH: 7.5 / 詳細: 20 mM HEPES, pH 7.5, 150 mM NaCl |
| 試料 | 濃度: 0.5 mg/ml / 包埋: NO / シャドウイング: NO / 染色: NO / 凍結: YES / 詳細: OST complex reconstituted into nanodisc |
| 試料支持 | グリッドの材料: COPPER / グリッドのサイズ: 300 divisions/in. / グリッドのタイプ: Quantifoil R1.2/1.3 |
| 急速凍結 | 装置: FEI VITROBOT MARK IV / 凍結剤: ETHANE-PROPANE / 湿度: 95 % / 凍結前の試料温度: 277 K 詳細: Quantifoil carbon grids (300 mesh, copper) were glow discharged at 25 mA for 40 s. 3.5 ul of nanodisc-reconstituted OST sample at concentration of 0.5 mg/ml was applied onto the grid and ...詳細: Quantifoil carbon grids (300 mesh, copper) were glow discharged at 25 mA for 40 s. 3.5 ul of nanodisc-reconstituted OST sample at concentration of 0.5 mg/ml was applied onto the grid and grids were blotted for 3 s and flash-frozen in a mixture of liquid ethane and propane cooled by liquid nitrogen. |
- 電子顕微鏡撮影
電子顕微鏡撮影
| 実験機器 |  モデル: Titan Krios / 画像提供: FEI Company |
|---|---|
| 顕微鏡 | モデル: FEI TITAN KRIOS |
| 電子銃 | 電子線源:  FIELD EMISSION GUN / 加速電圧: 300 kV / 照射モード: FLOOD BEAM FIELD EMISSION GUN / 加速電圧: 300 kV / 照射モード: FLOOD BEAM |
| 電子レンズ | モード: BRIGHT FIELD / Cs: 2.7 mm / C2レンズ絞り径: 100 µm / アライメント法: COMA FREE |
| 試料ホルダ | 凍結剤: NITROGEN 試料ホルダーモデル: FEI TITAN KRIOS AUTOGRID HOLDER |
| 撮影 | 平均露光時間: 0.25 sec. / 電子線照射量: 2 e/Å2 フィルム・検出器のモデル: GATAN K2 SUMMIT (4k x 4k) 撮影したグリッド数: 2 / 実像数: 3915 |
| 電子光学装置 | エネルギーフィルター名称: GIF Quantum LS |
- 解析
解析
| ソフトウェア | 名称: PHENIX / バージョン: 1.11.1_2575: / 分類: 精密化 | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| EMソフトウェア |
| ||||||||||||||||||||||||||||||||
| CTF補正 | タイプ: PHASE FLIPPING AND AMPLITUDE CORRECTION | ||||||||||||||||||||||||||||||||
| 粒子像の選択 | 選択した粒子像数: 486361 | ||||||||||||||||||||||||||||||||
| 対称性 | 点対称性: C1 (非対称) | ||||||||||||||||||||||||||||||||
| 3次元再構成 | 解像度: 3.3 Å / 解像度の算出法: FSC 0.143 CUT-OFF / 粒子像の数: 110091 / 対称性のタイプ: POINT | ||||||||||||||||||||||||||||||||
| 原子モデル構築 | プロトコル: AB INITIO MODEL | ||||||||||||||||||||||||||||||||
| 拘束条件 |
|
 ムービー
ムービー コントローラー
コントローラー

















 PDBj
PDBj













