Entry Database : PDB / ID : 6a6aTitle VanYB in complex with D-Alanine D-alanyl-D-alanine carboxypeptidase Keywords / / / Function / homology Function Domain/homology Component
/ / / / / / / / / / / / / / / / Biological species Enterococcus faecalis (bacteria)Method / / / Resolution : 2.26 Å Authors Kim, H.S. / Hahn, H. Funding support Organization Grant number Country Ministry of Science, ICT and Future Planning 2013R1A2A1A05067303 Ministry of Science, ICT and Future Planning NRF-2011-0030001 Ministry of Science, ICT and Future Planning NRF-2013M3A6A4043695 Ministry of Science, ICT and Future Planning NRF-2017R1C1B2012225
Journal : Int. J. Biol. Macromol. / Year : 2018Title : Structural basis for the substrate recognition of peptidoglycan pentapeptides by Enterococcus faecalis VanYB.Authors : Kim, H.S. / Hahn, H. / Kim, J. / Jang, D.M. / Lee, J.Y. / Back, J.M. / Im, H.N. / Kim, H. / Han, B.W. / Suh, S.W. History Deposition Jun 27, 2018 Deposition site / Processing site Revision 1.0 Sep 5, 2018 Provider / Type Revision 1.1 Nov 22, 2023 Group Data collection / Database references ... Data collection / Database references / Derived calculations / Refinement description Category chem_comp_atom / chem_comp_bond ... chem_comp_atom / chem_comp_bond / database_2 / pdbx_initial_refinement_model / struct_conn Item _database_2.pdbx_DOI / _database_2.pdbx_database_accession ... _database_2.pdbx_DOI / _database_2.pdbx_database_accession / _struct_conn.pdbx_dist_value / _struct_conn.ptnr1_auth_asym_id / _struct_conn.ptnr1_auth_comp_id / _struct_conn.ptnr1_auth_seq_id / _struct_conn.ptnr1_label_asym_id / _struct_conn.ptnr1_label_atom_id / _struct_conn.ptnr1_label_comp_id / _struct_conn.ptnr1_label_seq_id / _struct_conn.ptnr2_auth_asym_id / _struct_conn.ptnr2_label_asym_id
Show all Show less
 Open data
Open data Basic information
Basic information Components
Components Keywords
Keywords Function and homology information
Function and homology information
 X-RAY DIFFRACTION /
X-RAY DIFFRACTION /  SYNCHROTRON /
SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.26 Å
MOLECULAR REPLACEMENT / Resolution: 2.26 Å  Authors
Authors Korea, Republic Of, 4items
Korea, Republic Of, 4items  Citation
Citation Journal: Int. J. Biol. Macromol. / Year: 2018
Journal: Int. J. Biol. Macromol. / Year: 2018 Structure visualization
Structure visualization Molmil
Molmil Jmol/JSmol
Jmol/JSmol Downloads & links
Downloads & links Download
Download 6a6a.cif.gz
6a6a.cif.gz PDBx/mmCIF format
PDBx/mmCIF format pdb6a6a.ent.gz
pdb6a6a.ent.gz PDB format
PDB format 6a6a.json.gz
6a6a.json.gz PDBx/mmJSON format
PDBx/mmJSON format Other downloads
Other downloads https://data.pdbj.org/pub/pdb/validation_reports/a6/6a6a
https://data.pdbj.org/pub/pdb/validation_reports/a6/6a6a ftp://data.pdbj.org/pub/pdb/validation_reports/a6/6a6a
ftp://data.pdbj.org/pub/pdb/validation_reports/a6/6a6a

 Links
Links Assembly
Assembly
 Components
Components

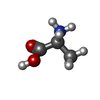










 X-RAY DIFFRACTION / Number of used crystals: 1
X-RAY DIFFRACTION / Number of used crystals: 1  Sample preparation
Sample preparation SYNCHROTRON / Site: PAL/PLS
SYNCHROTRON / Site: PAL/PLS  / Beamline: 7A (6B, 6C1) / Wavelength: 0.97934 Å
/ Beamline: 7A (6B, 6C1) / Wavelength: 0.97934 Å Processing
Processing MOLECULAR REPLACEMENT
MOLECULAR REPLACEMENT Movie
Movie Controller
Controller













 PDBj
PDBj




