Entry Database : PDB / ID : 5z0bTitle Crystal structure of plasma-derived human serum albumin Serum albumin Keywords / Function / homology Function Domain/homology Component
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / Biological species Homo sapiens (human)Method / / / Resolution : 2.17 Å Authors Park, J. / Kim, M.-S. / Shin, D.H. Funding support Organization Grant number Country National Research Foundation (NRF, Korea) 2015R1D1A1A01058942
Journal : To Be Published Title : The crystal structure of plasma-derived human serum albuminAuthors : Park, J. / Kim, M.S. / Shin, D.H. History Deposition Dec 19, 2017 Deposition site / Processing site Revision 1.0 Dec 19, 2018 Provider / Type Revision 2.0 Jun 24, 2020 Group Advisory / Atomic model ... Advisory / Atomic model / Author supporting evidence / Data collection / Derived calculations / Non-polymer description / Other / Refinement description / Structure summary Category atom_site / atom_site_anisotrop ... atom_site / atom_site_anisotrop / cell / chem_comp / computing / diffrn / entity / pdbx_audit_support / pdbx_entity_instance_feature / pdbx_entity_nonpoly / pdbx_entry_details / pdbx_nonpoly_scheme / pdbx_refine_tls / pdbx_refine_tls_group / pdbx_struct_assembly_gen / pdbx_struct_assembly_prop / pdbx_validate_close_contact / pdbx_validate_symm_contact / pdbx_validate_torsion / refine / refine_hist / refine_ls_restr / refine_ls_shell / reflns / software / struct_asym / struct_conf / struct_conn / struct_mon_prot_cis / struct_site / struct_site_gen / symmetry Item _cell.angle_beta / _cell.volume ... _cell.angle_beta / _cell.volume / _chem_comp.formula / _chem_comp.formula_weight / _chem_comp.id / _chem_comp.mon_nstd_flag / _chem_comp.name / _chem_comp.pdbx_synonyms / _chem_comp.type / _diffrn.pdbx_serial_crystal_experiment / _pdbx_audit_support.funding_organization / _pdbx_struct_assembly_gen.asym_id_list / _pdbx_struct_assembly_prop.value / _pdbx_validate_symm_contact.auth_asym_id_1 / _pdbx_validate_symm_contact.auth_asym_id_2 / _pdbx_validate_symm_contact.auth_atom_id_1 / _pdbx_validate_symm_contact.auth_atom_id_2 / _pdbx_validate_symm_contact.auth_comp_id_1 / _pdbx_validate_symm_contact.auth_comp_id_2 / _pdbx_validate_symm_contact.auth_seq_id_1 / _pdbx_validate_symm_contact.auth_seq_id_2 / _pdbx_validate_symm_contact.dist / _pdbx_validate_symm_contact.site_symmetry_2 / _refine.B_iso_mean / _refine.ls_R_factor_R_free / _refine.ls_R_factor_R_work / _refine.ls_R_factor_obs / _refine.ls_d_res_high / _refine.ls_number_reflns_R_work / _refine.ls_number_reflns_all / _refine.overall_SU_ML / _refine.pdbx_overall_phase_error / _refine.pdbx_stereochemistry_target_values / _refine.solvent_model_details / _refine_hist.d_res_high / _refine_hist.number_atoms_solvent / _refine_hist.number_atoms_total / _refine_hist.pdbx_number_atoms_ligand / _refine_ls_restr.dev_ideal / _refine_ls_restr.number / _refine_ls_restr.type / _refine_ls_shell.R_factor_R_free / _refine_ls_shell.R_factor_R_work / _refine_ls_shell.d_res_high / _refine_ls_shell.d_res_low / _refine_ls_shell.number_reflns_R_free / _refine_ls_shell.number_reflns_R_work / _refine_ls_shell.percent_reflns_obs / _reflns.B_iso_Wilson_estimate / _software.version / _struct_mon_prot_cis.pdbx_omega_angle / _symmetry.space_group_name_Hall Description Details Provider / Type Revision 2.1 Nov 22, 2023 Group / Database references / Refinement descriptionCategory chem_comp_atom / chem_comp_bond ... chem_comp_atom / chem_comp_bond / database_2 / pdbx_initial_refinement_model Item / _database_2.pdbx_database_accessionRevision 2.2 Oct 23, 2024 Group / Category / pdbx_modification_feature / Item
Show all Show less
 Open data
Open data Basic information
Basic information Components
Components Keywords
Keywords Function and homology information
Function and homology information Homo sapiens (human)
Homo sapiens (human) X-RAY DIFFRACTION /
X-RAY DIFFRACTION /  SYNCHROTRON /
SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.17 Å
MOLECULAR REPLACEMENT / Resolution: 2.17 Å  Authors
Authors Korea, Republic Of, 1items
Korea, Republic Of, 1items  Citation
Citation Journal: To Be Published
Journal: To Be Published Structure visualization
Structure visualization Molmil
Molmil Jmol/JSmol
Jmol/JSmol Downloads & links
Downloads & links Download
Download 5z0b.cif.gz
5z0b.cif.gz PDBx/mmCIF format
PDBx/mmCIF format pdb5z0b.ent.gz
pdb5z0b.ent.gz PDB format
PDB format 5z0b.json.gz
5z0b.json.gz PDBx/mmJSON format
PDBx/mmJSON format Other downloads
Other downloads https://data.pdbj.org/pub/pdb/validation_reports/z0/5z0b
https://data.pdbj.org/pub/pdb/validation_reports/z0/5z0b ftp://data.pdbj.org/pub/pdb/validation_reports/z0/5z0b
ftp://data.pdbj.org/pub/pdb/validation_reports/z0/5z0b
 Links
Links Assembly
Assembly
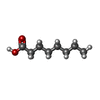
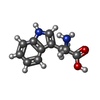
 Components
Components Homo sapiens (human) / References: UniProt: P02768
Homo sapiens (human) / References: UniProt: P02768
















 X-RAY DIFFRACTION / Number of used crystals: 1
X-RAY DIFFRACTION / Number of used crystals: 1  Sample preparation
Sample preparation SYNCHROTRON / Site:
SYNCHROTRON / Site:  Australian Synchrotron
Australian Synchrotron  / Beamline: MX1 / Wavelength: 0.9536 Å
/ Beamline: MX1 / Wavelength: 0.9536 Å Processing
Processing MOLECULAR REPLACEMENT
MOLECULAR REPLACEMENT Movie
Movie Controller
Controller











 PDBj
PDBj


























