[English] 日本語
 Yorodumi
Yorodumi- PDB-5vu4: Crystal structure of the A/Hong Kong/1/1968 (H3N2) influenza viru... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 5vu4 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Crystal structure of the A/Hong Kong/1/1968 (H3N2) influenza virus hemagglutinin G225Q/L226A mutant in complex with 6'-SLN | |||||||||
 Components Components | (Hemagglutinin ...) x 2 | |||||||||
 Keywords Keywords | VIRAL PROTEIN / Influenza A virus / hemagglutinin / mutant / receptor binding | |||||||||
| Function / homology |  Function and homology information Function and homology informationviral budding from plasma membrane / clathrin-dependent endocytosis of virus by host cell / host cell surface receptor binding / fusion of virus membrane with host plasma membrane / fusion of virus membrane with host endosome membrane / viral envelope / virion attachment to host cell / host cell plasma membrane / virion membrane / membrane Similarity search - Function | |||||||||
| Biological species |   Influenza A virus Influenza A virus | |||||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.25 Å MOLECULAR REPLACEMENT / Resolution: 2.25 Å | |||||||||
 Authors Authors | Wu, N.C. / Wilson, I.A. | |||||||||
| Funding support |  United States, 2items United States, 2items
| |||||||||
 Citation Citation |  Journal: Cell Host Microbe / Year: 2017 Journal: Cell Host Microbe / Year: 2017Title: Diversity of Functionally Permissive Sequences in the Receptor-Binding Site of Influenza Hemagglutinin. Authors: Wu, N.C. / Xie, J. / Zheng, T. / Nycholat, C.M. / Grande, G. / Paulson, J.C. / Lerner, R.A. / Wilson, I.A. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  5vu4.cif.gz 5vu4.cif.gz | 611.6 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb5vu4.ent.gz pdb5vu4.ent.gz | 507.2 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  5vu4.json.gz 5vu4.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/vu/5vu4 https://data.pdbj.org/pub/pdb/validation_reports/vu/5vu4 ftp://data.pdbj.org/pub/pdb/validation_reports/vu/5vu4 ftp://data.pdbj.org/pub/pdb/validation_reports/vu/5vu4 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  5vtqC  5vtrC  5vtuC  5vtvC  5vtwC  5vtxC  5vtyC  5vtzC  4fnkS C: citing same article ( S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 2 | 
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 3 | 
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 4 |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Unit cell |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Noncrystallographic symmetry (NCS) | NCS domain:
NCS domain segments: Component-ID: _ / Refine code: _
NCS ensembles :
|
- Components
Components
-Hemagglutinin ... , 2 types, 6 molecules ACEBDF
| #1: Protein | Mass: 35535.945 Da / Num. of mol.: 3 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Influenza A virus (strain A/Hong Kong/1/1968 H3N2) Influenza A virus (strain A/Hong Kong/1/1968 H3N2)Strain: A/Hong Kong/1/1968 H3N2 / Gene: HA / Production host:  Trichoplusia ni (cabbage looper) / References: UniProt: Q91MA7 Trichoplusia ni (cabbage looper) / References: UniProt: Q91MA7#2: Protein | Mass: 20041.131 Da / Num. of mol.: 3 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Influenza A virus (strain A/Hong Kong/1/1968 H3N2) Influenza A virus (strain A/Hong Kong/1/1968 H3N2)Strain: A/Hong Kong/1/1968 H3N2 / Gene: HA / Production host:  Trichoplusia ni (cabbage looper) / References: UniProt: Q91MA7 Trichoplusia ni (cabbage looper) / References: UniProt: Q91MA7 |
|---|
-Sugars , 7 types, 13 molecules 
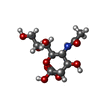

| #3: Polysaccharide | beta-D-mannopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-2-acetamido-2-deoxy-beta- ...beta-D-mannopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose Source method: isolated from a genetically manipulated source | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| #4: Polysaccharide | Source method: isolated from a genetically manipulated source #5: Polysaccharide | N-acetyl-alpha-neuraminic acid-(2-6)-beta-D-galactopyranose | Source method: isolated from a genetically manipulated source #6: Polysaccharide | alpha-D-mannopyranose-(1-3)-[alpha-D-mannopyranose-(1-6)]beta-D-mannopyranose-(1-4)-2-acetamido-2- ...alpha-D-mannopyranose-(1-3)-[alpha-D-mannopyranose-(1-6)]beta-D-mannopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose | Source method: isolated from a genetically manipulated source #7: Polysaccharide | alpha-D-mannopyranose-(1-3)-beta-D-mannopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose-(1- ...alpha-D-mannopyranose-(1-3)-beta-D-mannopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose | Source method: isolated from a genetically manipulated source #8: Sugar | ChemComp-NAG / #9: Sugar | |
-Non-polymers , 1 types, 685 molecules 
| #10: Water | ChemComp-HOH / |
|---|
-Details
| Has protein modification | Y |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.96 Å3/Da / Density % sol: 58.51 % |
|---|---|
| Crystal grow | Temperature: 293 K / Method: vapor diffusion, sitting drop / pH: 6.5 Details: 0.1 M sodium cacodylate pH 6.5, 5% PEG 8000, and 38% 2-methyl-2,4-pentanediol |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  APS APS  / Beamline: 23-ID-D / Wavelength: 1.0331 Å / Beamline: 23-ID-D / Wavelength: 1.0331 Å |
| Detector | Type: DECTRIS PILATUS3 6M / Detector: PIXEL / Date: Oct 15, 2016 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1.0331 Å / Relative weight: 1 |
| Reflection | Resolution: 2.25→50 Å / Num. obs: 91825 / % possible obs: 99.9 % / Redundancy: 6.6 % / CC1/2: 1 / Rpim(I) all: 0.03 / Rsym value: 0.08 / Net I/σ(I): 17.8 |
| Reflection shell | Resolution: 2.25→2.32 Å / Redundancy: 5.6 % / Mean I/σ(I) obs: 1.8 / Num. unique obs: 8243 / Rpim(I) all: 0.27 / Rsym value: 0.6 / % possible all: 99.3 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 4FNK Resolution: 2.25→50 Å / Cor.coef. Fo:Fc: 0.961 / Cor.coef. Fo:Fc free: 0.951 / SU B: 10.835 / SU ML: 0.141 / Cross valid method: THROUGHOUT / ESU R: 0.241 / ESU R Free: 0.181 / Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.2 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 55.453 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: 1 / Resolution: 2.25→50 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller






















 PDBj
PDBj







