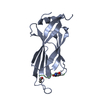
Entry Database : PDB / ID : 5mpvTitle Crystal structure of a Mycobacterium tuberculosis chorismate mutase optimized for high autonomous activity by directed evolution Intracellular chorismate mutase Keywords / / / Function / homology Function Domain/homology Component
/ / / / / / / / / / / / / / / / / / / / / / / / / / / Biological species Mycobacterium tuberculosis (bacteria)Method / / / Resolution : 1.49 Å Authors Thorbjornsrud, H.V. / Kamarauskaite, J. / Kast, P. / Krengel, U. Journal : J.Biol.Chem. / Year : 2020Title : Evolving the naturally compromised chorismate mutase from Mycobacterium tuberculosis to top performance.Authors : Fahrig-Kamarauskait, J. / Wurth-Roderer, K. / Thorbjornsrud, H.V. / Mailand, S. / Krengel, U. / Kast, P. History Deposition Dec 19, 2016 Deposition site / Processing site Revision 1.0 Aug 1, 2018 Provider / Type Revision 1.1 Feb 1, 2023 Group / Category / citation_author / database_2Item _citation.country / _citation.journal_abbrev ... _citation.country / _citation.journal_abbrev / _citation.journal_id_ASTM / _citation.journal_id_CSD / _citation.journal_id_ISSN / _citation.journal_volume / _citation.page_first / _citation.page_last / _citation.pdbx_database_id_DOI / _citation.pdbx_database_id_PubMed / _citation.title / _citation.year / _database_2.pdbx_DOI / _database_2.pdbx_database_accession Revision 1.2 Feb 7, 2024 Group / Refinement descriptionCategory / chem_comp_bond / pdbx_initial_refinement_model
Show all Show less
 Yorodumi
Yorodumi Open data
Open data Basic information
Basic information Components
Components Keywords
Keywords Function and homology information
Function and homology information
 X-RAY DIFFRACTION /
X-RAY DIFFRACTION /  SYNCHROTRON /
SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.49 Å
MOLECULAR REPLACEMENT / Resolution: 1.49 Å  Authors
Authors Citation
Citation Journal: J.Biol.Chem. / Year: 2020
Journal: J.Biol.Chem. / Year: 2020 Structure visualization
Structure visualization Molmil
Molmil Jmol/JSmol
Jmol/JSmol Downloads & links
Downloads & links Download
Download 5mpv.cif.gz
5mpv.cif.gz PDBx/mmCIF format
PDBx/mmCIF format pdb5mpv.ent.gz
pdb5mpv.ent.gz PDB format
PDB format 5mpv.json.gz
5mpv.json.gz PDBx/mmJSON format
PDBx/mmJSON format Other downloads
Other downloads https://data.pdbj.org/pub/pdb/validation_reports/mp/5mpv
https://data.pdbj.org/pub/pdb/validation_reports/mp/5mpv ftp://data.pdbj.org/pub/pdb/validation_reports/mp/5mpv
ftp://data.pdbj.org/pub/pdb/validation_reports/mp/5mpv
 Links
Links Assembly
Assembly

 Components
Components Mycobacterium tuberculosis (strain ATCC 25618 / H37Rv) (bacteria)
Mycobacterium tuberculosis (strain ATCC 25618 / H37Rv) (bacteria)
 X-RAY DIFFRACTION / Number of used crystals: 1
X-RAY DIFFRACTION / Number of used crystals: 1  Sample preparation
Sample preparation SYNCHROTRON / Site:
SYNCHROTRON / Site:  ESRF
ESRF  / Beamline: BM14 / Wavelength: 0.953723 Å
/ Beamline: BM14 / Wavelength: 0.953723 Å Processing
Processing MOLECULAR REPLACEMENT
MOLECULAR REPLACEMENT Movie
Movie Controller
Controller











 PDBj
PDBj
