[English] 日本語
 Yorodumi
Yorodumi- PDB-5m8q: Crystal structure of human tyrosinase related protein 1 mutant (T... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 5m8q | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Crystal structure of human tyrosinase related protein 1 mutant (T391V-R374S-Y362F) in complex with kojic acid | |||||||||
 Components Components | 5,6-dihydroxyindole-2-carboxylic acid oxidase | |||||||||
 Keywords Keywords | UNKNOWN FUNCTION / human tyrosinase related protein 1 / melanin biosynthesis / tyrosinase / oxidoreductase | |||||||||
| Function / homology |  Function and homology information Function and homology information: / Melanin biosynthesis / Oxidoreductases; Acting on paired donors, with incorporation or reduction of molecular oxygen; With another compound as one donor, and incorporation of one atom of oxygen into the other donor / tyrosinase activity / catechol oxidase activity / positive regulation of melanin biosynthetic process / melanin biosynthetic process / melanocyte differentiation / melanosome membrane / melanosome organization ...: / Melanin biosynthesis / Oxidoreductases; Acting on paired donors, with incorporation or reduction of molecular oxygen; With another compound as one donor, and incorporation of one atom of oxygen into the other donor / tyrosinase activity / catechol oxidase activity / positive regulation of melanin biosynthetic process / melanin biosynthetic process / melanocyte differentiation / melanosome membrane / melanosome organization / intracellular vesicle / Regulation of MITF-M-dependent genes involved in pigmentation / clathrin-coated endocytic vesicle membrane / melanosome / endosome membrane / protein homodimerization activity / metal ion binding / cytoplasm Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / Resolution: 2.85 Å SYNCHROTRON / Resolution: 2.85 Å | |||||||||
 Authors Authors | Lai, X. / Soler-Lopez, M. / Wichers, H.J. / Dijkstra, B.W. | |||||||||
 Citation Citation |  Journal: Angew. Chem. Int. Ed. Engl. / Year: 2017 Journal: Angew. Chem. Int. Ed. Engl. / Year: 2017Title: Structure of Human Tyrosinase Related Protein 1 Reveals a Binuclear Zinc Active Site Important for Melanogenesis. Authors: Lai, X. / Wichers, H.J. / Soler-Lopez, M. / Dijkstra, B.W. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  5m8q.cif.gz 5m8q.cif.gz | 369 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb5m8q.ent.gz pdb5m8q.ent.gz | 300.9 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  5m8q.json.gz 5m8q.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/m8/5m8q https://data.pdbj.org/pub/pdb/validation_reports/m8/5m8q ftp://data.pdbj.org/pub/pdb/validation_reports/m8/5m8q ftp://data.pdbj.org/pub/pdb/validation_reports/m8/5m8q | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  5m8lC  5m8mC  5m8nC  5m8oC  5m8pC  5m8rC  5m8tC C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||
| 2 | 
| ||||||||
| 3 | 
| ||||||||
| 4 | 
| ||||||||
| Unit cell |
|
- Components
Components
-Protein , 1 types, 4 molecules ABCD
| #1: Protein | Mass: 50531.160 Da / Num. of mol.: 4 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: TYRP1, CAS2, TYRP, TYRRP / Production host: Homo sapiens (human) / Gene: TYRP1, CAS2, TYRP, TYRRP / Production host:  References: UniProt: P17643, Oxidoreductases; Acting on paired donors, with incorporation or reduction of molecular oxygen; With another compound as one donor, and incorporation of one atom of oxygen into the other donor |
|---|
-Sugars , 6 types, 18 molecules 
| #2: Polysaccharide | 2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-[alpha-L-fucopyranose-(1-6)]2-acetamido-2-deoxy-beta- ...2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-[alpha-L-fucopyranose-(1-6)]2-acetamido-2-deoxy-beta-D-glucopyranose Source method: isolated from a genetically manipulated source | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| #3: Polysaccharide | 2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose Source method: isolated from a genetically manipulated source #4: Polysaccharide | alpha-D-mannopyranose-(1-3)-[alpha-D-mannopyranose-(1-6)]alpha-D-mannopyranose-(1-4)-2-acetamido-2- ...alpha-D-mannopyranose-(1-3)-[alpha-D-mannopyranose-(1-6)]alpha-D-mannopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose | Source method: isolated from a genetically manipulated source #5: Polysaccharide | alpha-L-fucopyranose-(1-6)-2-acetamido-2-deoxy-beta-D-glucopyranose Source method: isolated from a genetically manipulated source #6: Polysaccharide | alpha-D-mannopyranose-(1-3)-[alpha-D-mannopyranose-(1-6)]alpha-D-mannopyranose-(1-3)-2-acetamido-2- ...alpha-D-mannopyranose-(1-3)-[alpha-D-mannopyranose-(1-6)]alpha-D-mannopyranose-(1-3)-2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose | Source method: isolated from a genetically manipulated source #7: Sugar | ChemComp-NAG / |
-Non-polymers , 3 types, 17 molecules 
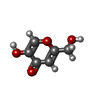



| #8: Chemical | ChemComp-ZN / #9: Chemical | ChemComp-KOJ / #10: Water | ChemComp-HOH / | |
|---|
-Details
| Has protein modification | Y |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.98 Å3/Da / Density % sol: 58.76 % |
|---|---|
| Crystal grow | Temperature: 290 K / Method: vapor diffusion, sitting drop Details: 0.1 M Tris (pH 7.0), 0.2 M NaCl and 30% (w/v) PEG 3000 |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  ESRF ESRF  / Beamline: MASSIF-1 / Wavelength: 0.976 Å / Beamline: MASSIF-1 / Wavelength: 0.976 Å |
| Detector | Type: DECTRIS PILATUS 6M-F / Detector: PIXEL / Date: Oct 1, 2015 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.976 Å / Relative weight: 1 |
| Reflection | Resolution: 2.85→48.77 Å / Num. obs: 57320 / % possible obs: 100 % / Redundancy: 6.7 % / Net I/σ(I): 9.9 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Resolution: 2.85→47.875 Å / SU ML: 0.44 / Cross valid method: FREE R-VALUE / σ(F): 1.33 / Phase error: 28.99
| ||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å | ||||||||||||||||||||||||
| Displacement parameters | Biso max: 112 Å2 / Biso mean: 57.1239 Å2 / Biso min: 25.52 Å2 | ||||||||||||||||||||||||
| Refinement step | Cycle: final / Resolution: 2.85→47.875 Å
|
 Movie
Movie Controller
Controller












 PDBj
PDBj


