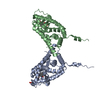
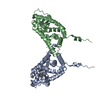
Entry Database : PDB / ID : 5dizTitle Crystal Structure of nuclear proteinaceous RNase P 2 (PRORP2) from A. thaliana Proteinaceous RNase P 2 Keywords / / / / / / / / / Function / homology Function Domain/homology Component
/ / / / / / / / / / / / / / / / / / / / / / / / / / / Biological species Arabidopsis thaliana (thale cress)Method / / / Resolution : 3.2 Å Authors Karasik, A. / Shanmuganathan, A. / Howard, M.J. / Fierke, C.A. / Koutmos, M. Journal : J.Mol.Biol. / Year : 2016Title : Nuclear Protein-Only Ribonuclease P2 Structure and Biochemical Characterization Provide Insight into the Conserved Properties of tRNA 5' End Processing Enzymes.Authors : Karasik, A. / Shanmuganathan, A. / Howard, M.J. / Fierke, C.A. / Koutmos, M. History Deposition Sep 1, 2015 Deposition site / Processing site Revision 1.0 Dec 30, 2015 Provider / Type Revision 1.1 Feb 17, 2016 Group Revision 1.2 Nov 1, 2017 Group / Database references / Derived calculationsCategory / pdbx_struct_assembly_auth_evidence / pdbx_struct_oper_listItem / _pdbx_struct_oper_list.symmetry_operationRevision 1.3 Sep 27, 2023 Group / Database references / Refinement descriptionCategory chem_comp_atom / chem_comp_bond ... chem_comp_atom / chem_comp_bond / database_2 / pdbx_initial_refinement_model Item / _database_2.pdbx_database_accession
Show all Show less
 Yorodumi
Yorodumi Open data
Open data Basic information
Basic information Components
Components Keywords
Keywords Function and homology information
Function and homology information
 X-RAY DIFFRACTION /
X-RAY DIFFRACTION /  SYNCHROTRON /
SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 3.2 Å
MOLECULAR REPLACEMENT / Resolution: 3.2 Å  Authors
Authors Citation
Citation Journal: J.Mol.Biol. / Year: 2016
Journal: J.Mol.Biol. / Year: 2016 Structure visualization
Structure visualization Molmil
Molmil Jmol/JSmol
Jmol/JSmol Downloads & links
Downloads & links Download
Download 5diz.cif.gz
5diz.cif.gz PDBx/mmCIF format
PDBx/mmCIF format pdb5diz.ent.gz
pdb5diz.ent.gz PDB format
PDB format 5diz.json.gz
5diz.json.gz PDBx/mmJSON format
PDBx/mmJSON format Other downloads
Other downloads 5diz_validation.pdf.gz
5diz_validation.pdf.gz wwPDB validaton report
wwPDB validaton report 5diz_full_validation.pdf.gz
5diz_full_validation.pdf.gz 5diz_validation.xml.gz
5diz_validation.xml.gz 5diz_validation.cif.gz
5diz_validation.cif.gz https://data.pdbj.org/pub/pdb/validation_reports/di/5diz
https://data.pdbj.org/pub/pdb/validation_reports/di/5diz ftp://data.pdbj.org/pub/pdb/validation_reports/di/5diz
ftp://data.pdbj.org/pub/pdb/validation_reports/di/5diz
 Links
Links Assembly
Assembly


 Components
Components

 X-RAY DIFFRACTION
X-RAY DIFFRACTION Sample preparation
Sample preparation SYNCHROTRON / Site:
SYNCHROTRON / Site:  APS
APS  / Beamline: 23-ID-B / Wavelength: 1.033 Å
/ Beamline: 23-ID-B / Wavelength: 1.033 Å Processing
Processing MOLECULAR REPLACEMENT
MOLECULAR REPLACEMENT Movie
Movie Controller
Controller










 PDBj
PDBj


