Entry Database : PDB / ID : 5b6sTitle Catalytic domain of Coprinopsis cinerea GH62 alpha-L-arabinofuranosidase Glycosyl hydrolase family 62 protein Keywords / / / / / Function / homology Function Domain/homology Component
/ / / / / / / / / / / / / / / / / / / / / / / / / / / Biological species Coprinopsis cinerea (fungus)Method / / / Resolution : 1.7 Å Authors Tonozuka, T. Funding support Organization Grant number Country Japan Society for the Promotion of Science 16K07687
Journal : Appl. Biochem. Biotechnol. / Year : 2017Title : Structure of the Catalytic Domain of alpha-L-Arabinofuranosidase from Coprinopsis cinerea, CcAbf62A, Provides Insights into Structure-Function Relationships in Glycoside Hydrolase Family 62Authors : Tonozuka, T. / Tanaka, Y. / Okuyama, S. / Miyazaki, T. / Nishikawa, A. / Yoshida, M. History Deposition Jun 1, 2016 Deposition site / Processing site Revision 1.0 Sep 7, 2016 Provider / Type Revision 1.1 Sep 14, 2016 Group Revision 1.2 Feb 15, 2017 Group Revision 1.3 Feb 26, 2020 Group / Category / Item Revision 1.4 Nov 8, 2023 Group Data collection / Database references ... Data collection / Database references / Derived calculations / Refinement description Category chem_comp_atom / chem_comp_bond ... chem_comp_atom / chem_comp_bond / database_2 / pdbx_initial_refinement_model / pdbx_struct_conn_angle / struct_conn Item _database_2.pdbx_DOI / _database_2.pdbx_database_accession ... _database_2.pdbx_DOI / _database_2.pdbx_database_accession / _pdbx_struct_conn_angle.ptnr1_auth_seq_id / _pdbx_struct_conn_angle.ptnr3_auth_seq_id / _pdbx_struct_conn_angle.value / _struct_conn.pdbx_dist_value / _struct_conn.ptnr2_auth_seq_id Revision 1.5 Nov 20, 2024 Group / Category / pdbx_modification_feature
Show all Show less
 Yorodumi
Yorodumi Open data
Open data Basic information
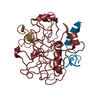
Basic information Components
Components Keywords
Keywords Function and homology information
Function and homology information Coprinopsis cinerea (fungus)
Coprinopsis cinerea (fungus) X-RAY DIFFRACTION /
X-RAY DIFFRACTION /  SYNCHROTRON /
SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.7 Å
MOLECULAR REPLACEMENT / Resolution: 1.7 Å  Authors
Authors Japan, 1items
Japan, 1items  Citation
Citation Journal: Appl. Biochem. Biotechnol. / Year: 2017
Journal: Appl. Biochem. Biotechnol. / Year: 2017 Structure visualization
Structure visualization Molmil
Molmil Jmol/JSmol
Jmol/JSmol Downloads & links
Downloads & links Download
Download 5b6s.cif.gz
5b6s.cif.gz PDBx/mmCIF format
PDBx/mmCIF format pdb5b6s.ent.gz
pdb5b6s.ent.gz PDB format
PDB format 5b6s.json.gz
5b6s.json.gz PDBx/mmJSON format
PDBx/mmJSON format Other downloads
Other downloads https://data.pdbj.org/pub/pdb/validation_reports/b6/5b6s
https://data.pdbj.org/pub/pdb/validation_reports/b6/5b6s ftp://data.pdbj.org/pub/pdb/validation_reports/b6/5b6s
ftp://data.pdbj.org/pub/pdb/validation_reports/b6/5b6s
 Links
Links Assembly
Assembly


 Components
Components Coprinopsis cinerea (strain Okayama-7 / 130 / ATCC MYA-4618 / FGSC 9003) (fungus)
Coprinopsis cinerea (strain Okayama-7 / 130 / ATCC MYA-4618 / FGSC 9003) (fungus)
 X-RAY DIFFRACTION / Number of used crystals: 1
X-RAY DIFFRACTION / Number of used crystals: 1  Sample preparation
Sample preparation SYNCHROTRON / Site:
SYNCHROTRON / Site:  Photon Factory
Photon Factory  / Beamline: AR-NW12A / Wavelength: 1 Å
/ Beamline: AR-NW12A / Wavelength: 1 Å Processing
Processing MOLECULAR REPLACEMENT
MOLECULAR REPLACEMENT Movie
Movie Controller
Controller











 PDBj
PDBj



