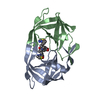
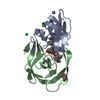
Entry Database : PDB / ID : 4ll3Title Structure of wild-type HIV protease in complex with darunavir Protease Keywords / / / / Function / homology Function Domain/homology Component
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / Biological species Method / / / Resolution : 1.95 Å Authors Grantz Saskova, K. / Rezacova, P. / Brynda, J. / Kozisek, M. / Konvalinka, J. Journal : Febs J. / Year : 2014Title : Thermodynamic and structural analysis of HIV protease resistance to darunavir - analysis of heavily mutated patient-derived HIV-1 proteases.Authors : Kozisek, M. / Lepsik, M. / Grantz Saskova, K. / Brynda, J. / Konvalinka, J. / Rezacova, P. History Deposition Jul 9, 2013 Deposition site / Processing site Supersession Apr 16, 2014 ID 3QOZ Revision 1.0 Apr 16, 2014 Provider / Type Revision 1.1 Sep 24, 2014 Group Revision 1.2 Feb 28, 2024 Group Data collection / Database references ... Data collection / Database references / Derived calculations / Refinement description / Structure summary Category chem_comp / chem_comp_atom ... chem_comp / chem_comp_atom / chem_comp_bond / database_2 / diffrn_source / struct_ncs_dom_lim / struct_site Item _chem_comp.pdbx_synonyms / _database_2.pdbx_DOI ... _chem_comp.pdbx_synonyms / _database_2.pdbx_DOI / _database_2.pdbx_database_accession / _diffrn_source.pdbx_synchrotron_site / _struct_ncs_dom_lim.beg_auth_comp_id / _struct_ncs_dom_lim.beg_label_alt_id / _struct_ncs_dom_lim.beg_label_asym_id / _struct_ncs_dom_lim.beg_label_comp_id / _struct_ncs_dom_lim.beg_label_seq_id / _struct_ncs_dom_lim.end_auth_comp_id / _struct_ncs_dom_lim.end_label_alt_id / _struct_ncs_dom_lim.end_label_asym_id / _struct_ncs_dom_lim.end_label_comp_id / _struct_ncs_dom_lim.end_label_seq_id / _struct_site.pdbx_auth_asym_id / _struct_site.pdbx_auth_comp_id / _struct_site.pdbx_auth_seq_id
Show all Show less
 Open data
Open data Basic information
Basic information Components
Components Keywords
Keywords Function and homology information
Function and homology information
 Human immunodeficiency virus 1
Human immunodeficiency virus 1 X-RAY DIFFRACTION /
X-RAY DIFFRACTION /  SYNCHROTRON /
SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.95 Å
MOLECULAR REPLACEMENT / Resolution: 1.95 Å  Authors
Authors Citation
Citation Journal: Febs J. / Year: 2014
Journal: Febs J. / Year: 2014 Structure visualization
Structure visualization Molmil
Molmil Jmol/JSmol
Jmol/JSmol Downloads & links
Downloads & links Download
Download 4ll3.cif.gz
4ll3.cif.gz PDBx/mmCIF format
PDBx/mmCIF format pdb4ll3.ent.gz
pdb4ll3.ent.gz PDB format
PDB format 4ll3.json.gz
4ll3.json.gz PDBx/mmJSON format
PDBx/mmJSON format Other downloads
Other downloads https://data.pdbj.org/pub/pdb/validation_reports/ll/4ll3
https://data.pdbj.org/pub/pdb/validation_reports/ll/4ll3 ftp://data.pdbj.org/pub/pdb/validation_reports/ll/4ll3
ftp://data.pdbj.org/pub/pdb/validation_reports/ll/4ll3 Links
Links Assembly
Assembly
 Components
Components
 Human immunodeficiency virus 1 / References: UniProt: Q9WFL7
Human immunodeficiency virus 1 / References: UniProt: Q9WFL7 X-RAY DIFFRACTION / Number of used crystals: 1
X-RAY DIFFRACTION / Number of used crystals: 1  Sample preparation
Sample preparation SYNCHROTRON / Site:
SYNCHROTRON / Site:  EMBL/DESY, HAMBURG
EMBL/DESY, HAMBURG  / Beamline: X12 / Wavelength: 0.953
/ Beamline: X12 / Wavelength: 0.953  Processing
Processing MOLECULAR REPLACEMENT / Resolution: 1.95→27.09 Å / Cor.coef. Fo:Fc: 0.966 / Cor.coef. Fo:Fc free: 0.946 / SU B: 9.638 / SU ML: 0.145 / Cross valid method: THROUGHOUT / ESU R: 0.21 / ESU R Free: 0.173 / Stereochemistry target values: MAXIMUM LIKELIHOOD / Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS
MOLECULAR REPLACEMENT / Resolution: 1.95→27.09 Å / Cor.coef. Fo:Fc: 0.966 / Cor.coef. Fo:Fc free: 0.946 / SU B: 9.638 / SU ML: 0.145 / Cross valid method: THROUGHOUT / ESU R: 0.21 / ESU R Free: 0.173 / Stereochemistry target values: MAXIMUM LIKELIHOOD / Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS Movie
Movie Controller
Controller






















 PDBj
PDBj