+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 4eak | ||||||
|---|---|---|---|---|---|---|---|
| Title | Co-crystal structure of an AMPK core with ATP | ||||||
 Components Components |
| ||||||
 Keywords Keywords | TRANSFERASE / AMPK | ||||||
| Function / homology |  Function and homology information Function and homology informationeukaryotic elongation factor-2 kinase activator activity / Energy dependent regulation of mTOR by LKB1-AMPK / Regulation of TP53 Activity through Phosphorylation / Macroautophagy / TP53 Regulates Metabolic Genes / nail development / positive regulation of mitochondrial transcription / [hydroxymethylglutaryl-CoA reductase (NADPH)] kinase / [hydroxymethylglutaryl-CoA reductase (NADPH)] kinase activity / regulation of stress granule assembly ...eukaryotic elongation factor-2 kinase activator activity / Energy dependent regulation of mTOR by LKB1-AMPK / Regulation of TP53 Activity through Phosphorylation / Macroautophagy / TP53 Regulates Metabolic Genes / nail development / positive regulation of mitochondrial transcription / [hydroxymethylglutaryl-CoA reductase (NADPH)] kinase / [hydroxymethylglutaryl-CoA reductase (NADPH)] kinase activity / regulation of stress granule assembly / negative regulation of tubulin deacetylation / histone H2BS36 kinase activity / cold acclimation / AMP-activated protein kinase activity / lipid droplet disassembly / phosphatidylethanolamine biosynthetic process / regulation of carbon utilization / positive regulation of skeletal muscle tissue development / CAMKK-AMPK signaling cascade / import into nucleus / phosphatidylcholine biosynthetic process / regulation of vesicle-mediated transport / protein localization to lysosome / negative regulation of hepatocyte apoptotic process / positive regulation of fatty acid oxidation / negative regulation of T cell mediated immune response to tumor cell / positive regulation of T cell mediated immune response to tumor cell / tau-protein kinase / nucleotide-activated protein kinase complex / protein kinase regulator activity / regulation of vascular permeability / negative regulation of TOR signaling / protein K6-linked ubiquitination / response to caffeine / regulation of glycolytic process / : / negative regulation of protein localization to nucleus / protein localization to lipid droplet / tau-protein kinase activity / lysosome organization / cholesterol biosynthetic process / negative regulation of T cell activation / lipid biosynthetic process / AMP binding / cellular response to stress / hepatocyte apoptotic process / SCF-dependent proteasomal ubiquitin-dependent protein catabolic process / fatty acid oxidation / positive regulation of release of cytochrome c from mitochondria / motor behavior / TORC1 signaling / positive regulation of translational initiation / cellular response to ethanol / fatty acid homeostasis / negative regulation of lipid catabolic process / response to UV / autophagosome assembly / cellular response to glucose starvation / positive regulation of protein localization / energy homeostasis / negative regulation of TORC1 signaling / negative regulation of insulin receptor signaling pathway / positive regulation of adipose tissue development / positive regulation of TORC1 signaling / negative regulation of translational initiation / positive regulation of autophagy / cellular response to calcium ion / positive regulation of gluconeogenesis / cellular response to nutrient levels / positive regulation of glycolytic process / cellular response to amino acid starvation / cellular response to starvation / regulation of microtubule cytoskeleton organization / response to activity / positive regulation of D-glucose import across plasma membrane / response to gamma radiation / protein localization to plasma membrane / circadian regulation of gene expression / response to hydrogen peroxide / cellular response to glucose stimulus / neuron cellular homeostasis / ADP binding / regulation of circadian rhythm / response to estrogen / protein destabilization / cellular response to xenobiotic stimulus / autophagy / glucose metabolic process / positive regulation of T cell activation / Wnt signaling pathway / cellular response to hydrogen peroxide / fatty acid biosynthetic process / cellular response to prostaglandin E stimulus / glucose homeostasis / positive regulation of cold-induced thermogenesis / cellular response to oxidative stress / protein-containing complex assembly / cellular response to hypoxia / protein phosphorylation / proteasome-mediated ubiquitin-dependent protein catabolic process Similarity search - Function | ||||||
| Biological species |  | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.5 Å MOLECULAR REPLACEMENT / Resolution: 2.5 Å | ||||||
 Authors Authors | Chen, L. / Wang, J. / Zhang, Y.-Y. / Yan, S.F. / Neumann, D. / Schlattner, U. / Wang, Z.-X. / Wu, J.-W. | ||||||
 Citation Citation |  Journal: Nat.Struct.Mol.Biol. / Year: 2012 Journal: Nat.Struct.Mol.Biol. / Year: 2012Title: AMP-activated protein kinase undergoes nucleotide-dependent conformational changes Authors: Chen, L. / Wang, J. / Zhang, Y.-Y. / Yan, S.F. / Neumann, D. / Schlattner, U. / Wang, Z.-X. / Wu, J.-W. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  4eak.cif.gz 4eak.cif.gz | 193.1 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb4eak.ent.gz pdb4eak.ent.gz | 153.7 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  4eak.json.gz 4eak.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/ea/4eak https://data.pdbj.org/pub/pdb/validation_reports/ea/4eak ftp://data.pdbj.org/pub/pdb/validation_reports/ea/4eak ftp://data.pdbj.org/pub/pdb/validation_reports/ea/4eak | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  4eagC  4eaiC  4eajC  4ealC  2v8qS C: citing same article ( S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
-Protein , 1 types, 1 molecules A
| #1: Protein | Mass: 12097.888 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Details: Chimera protein of residues 405-479 and residues 540-559 from 5'-AMP-activated protein kinase catalytic subunit alpha-1 (Uniprot P54645), linked by linker GGGGGG Source: (gene. exp.)   References: UniProt: P54645, non-specific serine/threonine protein kinase, EC: 2.7.11.27, [hydroxymethylglutaryl-CoA reductase (NADPH)] kinase, tau-protein kinase |
|---|
-5'-AMP-activated protein kinase subunit ... , 2 types, 2 molecules BC
| #2: Protein | Mass: 8133.786 Da / Num. of mol.: 1 / Fragment: UNP residues 200-270 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   |
|---|---|
| #3: Protein | Mass: 37434.094 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   |
-Non-polymers , 3 types, 77 molecules 
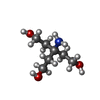



| #4: Chemical | | #5: Chemical | ChemComp-TAM / | #6: Water | ChemComp-HOH / | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.32 Å3/Da / Density % sol: 47.05 % |
|---|---|
| Crystal grow | Temperature: 298 K / Method: vapor diffusion, hanging drop / pH: 5.9 Details: 0.1M MES pH 5.9, 18% IPP, VAPOR DIFFUSION, HANGING DROP, temperature 298 K |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  SSRF SSRF  / Beamline: BL17U / Beamline: BL17U |
| Detector | Type: MARMOSAIC 225 mm CCD / Detector: CCD / Date: Mar 15, 2010 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Relative weight: 1 |
| Reflection | Resolution: 2.5→50 Å / Num. all: 18796 / Num. obs: 18796 / % possible obs: 100 % / Observed criterion σ(F): 0 / Observed criterion σ(I): 0 / Redundancy: 4.2 % / Biso Wilson estimate: 57.2 Å2 / Rmerge(I) obs: 0.089 / Net I/σ(I): 13.11 |
| Reflection shell | Resolution: 2.5→2.54 Å / Redundancy: 4.1 % / Rmerge(I) obs: 0.581 / Mean I/σ(I) obs: 2.2 / % possible all: 100 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: PDB ENTRY 2V8Q Resolution: 2.5→28.631 Å / Occupancy max: 1 / Occupancy min: 0.3 / SU ML: 0.39 / σ(F): 0.13 / Phase error: 26.47 / Stereochemistry target values: ML
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.8 Å / VDW probe radii: 1 Å / Solvent model: FLAT BULK SOLVENT MODEL / Bsol: 70.49 Å2 / ksol: 0.342 e/Å3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso max: 169.36 Å2 / Biso mean: 72.9033 Å2 / Biso min: 25.54 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.5→28.631 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Refine-ID: X-RAY DIFFRACTION / Total num. of bins used: 6
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS params. | Method: refined / Refine-ID: X-RAY DIFFRACTION
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS group |
|
 Movie
Movie Controller
Controller













 PDBj
PDBj

