Entry Database : PDB / ID : 4e1gTitle X-ray crystal structure of alpha-linolenic acid bound to the cyclooxygenase channel of cyclooxygenase-2 Prostaglandin G/H synthase 2 Keywords / / / / / Function / homology Function Domain/homology Component
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / Biological species Mus musculus (house mouse)Method / / / Resolution : 2.1 Å Authors Vecchio, A.J. / Malkowski, M.G. Journal : J.Biol.Chem. / Year : 2012Title : Investigating Substrate Promiscuity in Cyclooxygenase-2: THE ROLE OF ARG-120 AND RESIDUES LINING THE HYDROPHOBIC GROOVE.Authors : Vecchio, A.J. / Orlando, B.J. / Nandagiri, R. / Malkowski, M.G. History Deposition Mar 6, 2012 Deposition site / Processing site Revision 1.0 Apr 25, 2012 Provider / Type Revision 1.1 Jun 13, 2012 Group Revision 1.2 Aug 1, 2012 Group Revision 1.3 Nov 15, 2017 Group / Category / Item / _software.nameRevision 2.0 Jul 29, 2020 Group Advisory / Atomic model ... Advisory / Atomic model / Data collection / Database references / Derived calculations / Structure summary Category atom_site / chem_comp ... atom_site / chem_comp / entity / pdbx_branch_scheme / pdbx_chem_comp_identifier / pdbx_entity_branch / pdbx_entity_branch_descriptor / pdbx_entity_branch_link / pdbx_entity_branch_list / pdbx_entity_nonpoly / pdbx_nonpoly_scheme / pdbx_struct_assembly_gen / pdbx_struct_conn_angle / pdbx_validate_close_contact / struct_asym / struct_conn / struct_ref_seq_dif / struct_site / struct_site_gen Item _atom_site.B_iso_or_equiv / _atom_site.Cartn_x ... _atom_site.B_iso_or_equiv / _atom_site.Cartn_x / _atom_site.Cartn_y / _atom_site.Cartn_z / _atom_site.auth_asym_id / _atom_site.auth_atom_id / _atom_site.auth_comp_id / _atom_site.auth_seq_id / _atom_site.label_asym_id / _atom_site.label_atom_id / _atom_site.label_comp_id / _atom_site.label_entity_id / _atom_site.type_symbol / _chem_comp.mon_nstd_flag / _chem_comp.name / _chem_comp.type / _pdbx_struct_assembly_gen.asym_id_list / _pdbx_struct_conn_angle.ptnr1_auth_asym_id / _pdbx_struct_conn_angle.ptnr1_label_alt_id / _pdbx_struct_conn_angle.ptnr1_label_asym_id / _pdbx_struct_conn_angle.ptnr2_auth_asym_id / _pdbx_struct_conn_angle.ptnr2_label_asym_id / _pdbx_struct_conn_angle.ptnr3_auth_asym_id / _pdbx_struct_conn_angle.ptnr3_label_asym_id / _pdbx_struct_conn_angle.value / _pdbx_validate_close_contact.auth_asym_id_2 / _pdbx_validate_close_contact.auth_seq_id_2 / _struct_conn.pdbx_dist_value / _struct_conn.pdbx_leaving_atom_flag / _struct_conn.pdbx_ptnr1_label_alt_id / _struct_conn.pdbx_role / _struct_conn.ptnr1_auth_asym_id / _struct_conn.ptnr1_auth_comp_id / _struct_conn.ptnr1_auth_seq_id / _struct_conn.ptnr1_label_asym_id / _struct_conn.ptnr1_label_atom_id / _struct_conn.ptnr1_label_comp_id / _struct_conn.ptnr1_label_seq_id / _struct_conn.ptnr2_auth_asym_id / _struct_conn.ptnr2_auth_comp_id / _struct_conn.ptnr2_auth_seq_id / _struct_conn.ptnr2_label_asym_id / _struct_conn.ptnr2_label_comp_id / _struct_ref_seq_dif.details Description / Provider / Type Revision 2.1 Sep 13, 2023 Group Data collection / Database references ... Data collection / Database references / Refinement description / Structure summary Category chem_comp / chem_comp_atom ... chem_comp / chem_comp_atom / chem_comp_bond / database_2 / pdbx_initial_refinement_model Item / _database_2.pdbx_DOI / _database_2.pdbx_database_accessionRevision 2.2 Nov 27, 2024 Group / Structure summaryCategory chem_comp_atom / chem_comp_bond ... chem_comp_atom / chem_comp_bond / pdbx_entry_details / pdbx_modification_feature Item / _chem_comp_atom.pdbx_stereo_config / _chem_comp_bond.pdbx_aromatic_flag
Show all Show less
 Yorodumi
Yorodumi Open data
Open data Basic information
Basic information Components
Components Keywords
Keywords Function and homology information
Function and homology information
 X-RAY DIFFRACTION /
X-RAY DIFFRACTION /  SYNCHROTRON /
SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.1 Å
MOLECULAR REPLACEMENT / Resolution: 2.1 Å  Authors
Authors Citation
Citation Journal: J.Biol.Chem. / Year: 2012
Journal: J.Biol.Chem. / Year: 2012 Structure visualization
Structure visualization Molmil
Molmil Jmol/JSmol
Jmol/JSmol Downloads & links
Downloads & links Download
Download 4e1g.cif.gz
4e1g.cif.gz PDBx/mmCIF format
PDBx/mmCIF format pdb4e1g.ent.gz
pdb4e1g.ent.gz PDB format
PDB format 4e1g.json.gz
4e1g.json.gz PDBx/mmJSON format
PDBx/mmJSON format Other downloads
Other downloads 4e1g_validation.pdf.gz
4e1g_validation.pdf.gz wwPDB validaton report
wwPDB validaton report 4e1g_full_validation.pdf.gz
4e1g_full_validation.pdf.gz 4e1g_validation.xml.gz
4e1g_validation.xml.gz 4e1g_validation.cif.gz
4e1g_validation.cif.gz https://data.pdbj.org/pub/pdb/validation_reports/e1/4e1g
https://data.pdbj.org/pub/pdb/validation_reports/e1/4e1g ftp://data.pdbj.org/pub/pdb/validation_reports/e1/4e1g
ftp://data.pdbj.org/pub/pdb/validation_reports/e1/4e1g

 Links
Links Assembly
Assembly
 Components
Components




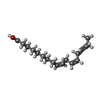
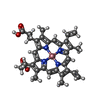

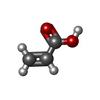





 X-RAY DIFFRACTION / Number of used crystals: 1
X-RAY DIFFRACTION / Number of used crystals: 1  Sample preparation
Sample preparation SYNCHROTRON / Site:
SYNCHROTRON / Site:  CHESS
CHESS  / Beamline: A1 / Wavelength: 0.9767 Å
/ Beamline: A1 / Wavelength: 0.9767 Å Processing
Processing MOLECULAR REPLACEMENT
MOLECULAR REPLACEMENT Movie
Movie Controller
Controller

















 PDBj
PDBj








