[English] 日本語
 Yorodumi
Yorodumi- PDB-4cc2: Complex of human Tuba C-terminal SH3 domain with human N-WASP pro... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 4cc2 | ||||||
|---|---|---|---|---|---|---|---|
| Title | Complex of human Tuba C-terminal SH3 domain with human N-WASP proline- rich peptide - P212121 | ||||||
 Components Components |
| ||||||
 Keywords Keywords | STRUCTURAL PROTEIN / SRC HOMOLOGY 3 / SH3 DOMAIN / PROLINE-RICH PEPTIDE / ACTIN CYTOSKELETON / CORTICAL TENSION | ||||||
| Function / homology |  Function and homology information Function and homology informationnegative regulation of membrane tubulation / spindle localization / positive regulation of clathrin-dependent endocytosis / negative regulation of lymphocyte migration / vesicle transport along actin filament / GTPase regulator activity / NOSTRIN mediated eNOS trafficking / actin cap / vesicle organization / vesicle budding from membrane ...negative regulation of membrane tubulation / spindle localization / positive regulation of clathrin-dependent endocytosis / negative regulation of lymphocyte migration / vesicle transport along actin filament / GTPase regulator activity / NOSTRIN mediated eNOS trafficking / actin cap / vesicle organization / vesicle budding from membrane / dendritic spine morphogenesis / actin polymerization or depolymerization / protein-containing complex localization / Golgi stack / Nephrin family interactions / DCC mediated attractive signaling / regulation of postsynapse organization / regulation of small GTPase mediated signal transduction / positive regulation of filopodium assembly / RHOV GTPase cycle / RHOJ GTPase cycle / RHOQ GTPase cycle / CDC42 GTPase cycle / cilium assembly / RHO GTPases Activate WASPs and WAVEs / actin filament polymerization / RAC1 GTPase cycle / EPHB-mediated forward signaling / guanyl-nucleotide exchange factor activity / FCGR3A-mediated phagocytosis / response to bacterium / Regulation of actin dynamics for phagocytic cup formation / endocytic vesicle membrane / cell-cell junction / presynapse / regulation of cell shape / actin cytoskeleton / lamellipodium / Clathrin-mediated endocytosis / regulation of protein localization / actin binding / actin cytoskeleton organization / protein-containing complex assembly / cytoplasmic vesicle / cytoskeleton / intracellular signal transduction / nuclear body / cell division / synapse / endoplasmic reticulum membrane / nucleolus / glutamatergic synapse / Golgi apparatus / positive regulation of transcription by RNA polymerase II / extracellular exosome / nucleoplasm / nucleus / plasma membrane / cytoplasm / cytosol Similarity search - Function | ||||||
| Biological species |  HOMO SAPIENS (human) HOMO SAPIENS (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.55 Å MOLECULAR REPLACEMENT / Resolution: 1.55 Å | ||||||
 Authors Authors | Polle, L. / Rigano, L. / Julian, R. / Ireton, K. / Schubert, W.-D. | ||||||
 Citation Citation |  Journal: Structure / Year: 2014 Journal: Structure / Year: 2014Title: Structural Details of Human Tuba Recruitment by Inlc of Listeria Monocytogenes Elucidate Bacterial Cell-Cell Spreading. Authors: Polle, L. / Rigano, L.A. / Julian, R. / Ireton, K. / Schubert, W. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  4cc2.cif.gz 4cc2.cif.gz | 79.1 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb4cc2.ent.gz pdb4cc2.ent.gz | 60.1 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  4cc2.json.gz 4cc2.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Summary document |  4cc2_validation.pdf.gz 4cc2_validation.pdf.gz | 465.1 KB | Display |  wwPDB validaton report wwPDB validaton report |
|---|---|---|---|---|
| Full document |  4cc2_full_validation.pdf.gz 4cc2_full_validation.pdf.gz | 469.1 KB | Display | |
| Data in XML |  4cc2_validation.xml.gz 4cc2_validation.xml.gz | 11.3 KB | Display | |
| Data in CIF |  4cc2_validation.cif.gz 4cc2_validation.cif.gz | 15.5 KB | Display | |
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/cc/4cc2 https://data.pdbj.org/pub/pdb/validation_reports/cc/4cc2 ftp://data.pdbj.org/pub/pdb/validation_reports/cc/4cc2 ftp://data.pdbj.org/pub/pdb/validation_reports/cc/4cc2 | HTTPS FTP |
-Related structure data
| Related structure data |  4cc3C  4cc4C  4cc7C  1zuuS C: citing same article ( S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||
| 2 | 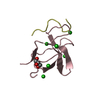
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 7712.617 Da / Num. of mol.: 2 Fragment: C-TERMINAL SH3 DOMAIN OF HUMAN TUBA, RESIDUES 1513-1577 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  HOMO SAPIENS (human) / Production host: HOMO SAPIENS (human) / Production host:  #2: Protein/peptide | Mass: 1077.188 Da / Num. of mol.: 2 / Fragment: N-WASP PROLINE-RICH PEPTIDE, RESIDUES 346-357 / Source method: obtained synthetically / Source: (synth.)  HOMO SAPIENS (human) / References: UniProt: O00401 HOMO SAPIENS (human) / References: UniProt: O00401#3: Chemical | ChemComp-GOL / #4: Chemical | ChemComp-CL / #5: Water | ChemComp-HOH / | Sequence details | FIRST TWO RESIDUES, GP, DERIVE FROM FUSION PROTEIN CLEAVAGE SITE | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 1.81 Å3/Da / Density % sol: 32 % / Description: NONE |
|---|---|
| Crystal grow | pH: 5.5 / Details: 3 M NACL, 0.1 M BIS-TRIS PH 5.5 |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  SOLEIL SOLEIL  / Beamline: PROXIMA 1 / Wavelength: 0.98 / Beamline: PROXIMA 1 / Wavelength: 0.98 |
| Detector | Type: DECTRIS PILATUS 6M / Detector: PIXEL / Date: Dec 18, 2011 / Details: KIRKPATRICK-BAEZ PAIR OF BI-MORPH MIRRORS |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.98 Å / Relative weight: 1 |
| Reflection | Resolution: 1.55→50 Å / Num. obs: 18318 / % possible obs: 93.4 % / Observed criterion σ(I): -3 / Redundancy: 2.8 % / Rmerge(I) obs: 0.04 / Net I/σ(I): 15.5 |
| Reflection shell | Resolution: 1.55→1.64 Å / Redundancy: 2.7 % / Rmerge(I) obs: 0.39 / Mean I/σ(I) obs: 2.7 / % possible all: 95.8 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: PDB ENTRY 1ZUU Resolution: 1.55→40.06 Å / Cor.coef. Fo:Fc: 0.966 / Cor.coef. Fo:Fc free: 0.95 / SU B: 2.624 / SU ML: 0.049 / Cross valid method: THROUGHOUT / ESU R: 0.02 / ESU R Free: 0.021 / Stereochemistry target values: MAXIMUM LIKELIHOOD Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS. U VALUES WITH TLS ADDED.
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.2 Å / Solvent model: BABINET MODEL WITH MASK | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 20.601 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.55→40.06 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller










 PDBj
PDBj










