[English] 日本語
 Yorodumi
Yorodumi- PDB-4c0j: Crystal structure of Drosophila Miro EF hand and cGTPase domains ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 4c0j | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Crystal structure of Drosophila Miro EF hand and cGTPase domains in the apo state (Apo-MiroS) | |||||||||
 Components Components | MITOCHONDRIAL RHO GTPASE | |||||||||
 Keywords Keywords | HYDROLASE / MITOCHONDRIAL TRANSPORT / CALCIUM-BINDING GTPASE / KINESIN / MITOPHAGY / HIDDEN EF HANDS | |||||||||
| Function / homology |  Function and homology information Function and homology informationRHOT2 GTPase cycle / RHOT1 GTPase cycle / anterograde axonal transport of mitochondrion / mitochondrial outer membrane permeabilization / Ub-specific processing proteases / regulation of mitochondrion organization / mitochondrion transport along microtubule / axonal transport of mitochondrion / axon cytoplasm / mitochondrion organization ...RHOT2 GTPase cycle / RHOT1 GTPase cycle / anterograde axonal transport of mitochondrion / mitochondrial outer membrane permeabilization / Ub-specific processing proteases / regulation of mitochondrion organization / mitochondrion transport along microtubule / axonal transport of mitochondrion / axon cytoplasm / mitochondrion organization / Hydrolases; Acting on acid anhydrides; Acting on GTP to facilitate cellular and subcellular movement / mitochondrial outer membrane / GTPase activity / calcium ion binding / GTP binding / magnesium ion binding Similarity search - Function | |||||||||
| Biological species |  | |||||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.82 Å MOLECULAR REPLACEMENT / Resolution: 2.82 Å | |||||||||
 Authors Authors | Klosowiak, J.L. / Focia, P.J. / Wawrzak, Z. / Chakravarthy, S. / Landahl, E.C. / Freymann, D.M. / Rice, S.E. | |||||||||
 Citation Citation |  Journal: Embo Rep. / Year: 2013 Journal: Embo Rep. / Year: 2013Title: Structural Coupling of the EF Hand and C-Terminal Gtpase Domains in the Mitochondrial Protein Miro. Authors: Klosowiak, J.L. / Focia, P.J. / Chakravarthy, S. / Landahl, E.C. / Freymann, D.M. / Rice, S.E. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  4c0j.cif.gz 4c0j.cif.gz | 96.9 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb4c0j.ent.gz pdb4c0j.ent.gz | 72.9 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  4c0j.json.gz 4c0j.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/c0/4c0j https://data.pdbj.org/pub/pdb/validation_reports/c0/4c0j ftp://data.pdbj.org/pub/pdb/validation_reports/c0/4c0j ftp://data.pdbj.org/pub/pdb/validation_reports/c0/4c0j | HTTPS FTP |
|---|
-Related structure data
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
-Protein , 1 types, 1 molecules A
| #1: Protein | Mass: 49118.531 Da / Num. of mol.: 1 / Fragment: ELM1, ELM2, AND CGTPASE, RESIDUES 201-617 Source method: isolated from a genetically manipulated source Details: DROSOPHILA MIRO RESIDUES 201-617 / Source: (gene. exp.)  Description: DROSOPHILA GENOMICS RESOURCE CENTER CLONE RE22983 Production host:  References: UniProt: Q8IMX7, Hydrolases; Acting on acid anhydrides; Acting on GTP to facilitate cellular and subcellular movement |
|---|
-Non-polymers , 5 types, 66 molecules 


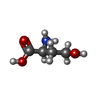





| #2: Chemical | ChemComp-UNX / | ||||||
|---|---|---|---|---|---|---|---|
| #3: Chemical | ChemComp-SO4 / #4: Chemical | ChemComp-NA / | #5: Chemical | ChemComp-HSE / | #6: Water | ChemComp-HOH / | |
-Details
| Nonpolymer details | L-HOMOSERINE |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 3.2 Å3/Da / Density % sol: 61 % Description: MOLECULAR REPLACEMENT MODEL WAS A MIRO STRUCTURE DETERMINED BY SAD PHASING OF SEMET-LABELED PROTEIN. |
|---|---|
| Crystal grow | pH: 7.6 Details: 5.0MG/ML MIROS, 1.7M LISO4, 0.1M BIS-TRIS PH 7.6, 5MM EGTA |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  APS APS  / Beamline: 21-ID-F / Wavelength: 0.97872 / Beamline: 21-ID-F / Wavelength: 0.97872 |
| Detector | Type: MARMOSAIC 225 mm CCD / Detector: CCD / Date: Nov 14, 2012 |
| Radiation | Monochromator: C (111) / Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.97872 Å / Relative weight: 1 |
| Reflection | Resolution: 2.82→41.9 Å / Num. obs: 15216 / % possible obs: 100 % / Observed criterion σ(I): -3 / Redundancy: 14.6 % / Biso Wilson estimate: 61.3 Å2 / Rmerge(I) obs: 0.1 / Net I/σ(I): 16.6 |
| Reflection shell | Resolution: 2.82→2.89 Å / Redundancy: 13.1 % / Rmerge(I) obs: 0.59 / Mean I/σ(I) obs: 3.5 / % possible all: 100 |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: INITIAL MODEL FROM PRIOR DATASET Resolution: 2.82→34.59 Å / SU ML: 0.39 / σ(F): 1.91 / Phase error: 32.38 / Stereochemistry target values: ML Details: REFINEMENT NUMBER OF REFLECTIONS TREATS ANOMALOUS PAIRS SEPARATELY. A LARGE LOOP COMPRISING RESIDUES 434-441 IS POORLY ORDERED, DESPITE THE SIDECHAINS OF I435 AND L437 CONTRIBUTING TO A ...Details: REFINEMENT NUMBER OF REFLECTIONS TREATS ANOMALOUS PAIRS SEPARATELY. A LARGE LOOP COMPRISING RESIDUES 434-441 IS POORLY ORDERED, DESPITE THE SIDECHAINS OF I435 AND L437 CONTRIBUTING TO A HYDROPHOBIC CRYSTAL CONTACT, AND A SECOND LOOP COMPRISING RESIDUES 561-566 IS ALSO POORLY ORDERED. RESIDUES OF BOTH HAVE BEEN BUILT AS ALA OR GLY WHERE SIDECHAIN POSITIONS COULD NOT BE DETERMINED.
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å / Solvent model: FLAT BULK SOLVENT MODEL | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 36.49 Å2 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.82→34.59 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell |
|
 Movie
Movie Controller
Controller














 PDBj
PDBj




